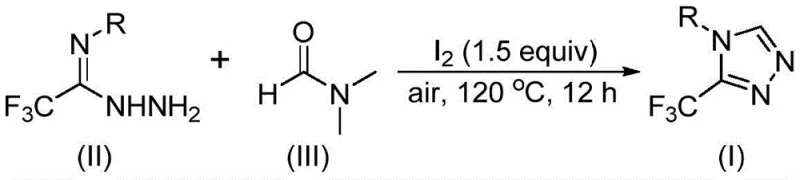
Advanced Iodine-Catalyzed Process Delivers High-Purity Triazole Intermediates with Unmatched Supply Chain Resilience and Cost Efficiency
The recently granted Chinese patent CN114920707B introduces a groundbreaking methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds that serve as critical building blocks in pharmaceutical development. This innovative approach leverages dimethylformamide (DMF) not merely as a solvent but as an essential carbon source within a tandem cyclization reaction promoted by molecular iodine under ambient air conditions. Operating at elevated temperatures between 110°C and 130°C for durations of 10 to 15 hours without requiring anhydrous or anaerobic environments represents a significant departure from conventional heterocyclic synthesis protocols. The methodology demonstrates remarkable versatility through substrate design flexibility, enabling the production of diverse triazole derivatives with varying functional groups at the critical position while maintaining consistent reaction efficiency across multiple aromatic systems. This advancement addresses fundamental limitations in current manufacturing approaches by eliminating specialized equipment requirements and reducing operational complexity throughout the production chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for trifluoromethyl-substituted triazoles typically require stringent anhydrous and anaerobic conditions that significantly increase operational complexity and cost while limiting scalability potential in commercial manufacturing environments. These methods often depend on transition metal catalysts that introduce challenging purification steps to remove heavy metal residues below acceptable thresholds for pharmaceutical applications. The narrow substrate scope observed in conventional approaches restricts structural diversity and functional group tolerance, thereby limiting the ability to tailor molecular properties for specific therapeutic applications. Additionally, many existing protocols demand specialized equipment for inert atmosphere maintenance and cryogenic temperature control that substantially increases capital expenditure and operational costs without corresponding yield improvements. The multi-step nature of conventional syntheses also generates significant waste streams that complicate environmental compliance and increase disposal costs across the production lifecycle.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-promoted tandem cyclization process that operates effectively under ambient air conditions without requiring moisture-sensitive handling procedures or specialized equipment. By utilizing DMF as both solvent and carbon source within a single reaction vessel, the process eliminates multiple unit operations while maintaining high functional group tolerance across diverse aromatic systems including halogenated and alkyl-substituted variants. The molecular iodine catalyst activates DMF through dual carbon-source pathways that enable consistent product formation without transition metal contamination concerns that plague conventional approaches. This streamlined protocol reduces processing steps from multi-stage sequences to a single operation while maintaining compatibility with standard manufacturing equipment found across global chemical production facilities. The inherent simplicity translates directly to enhanced operational flexibility and reduced capital requirements without compromising product quality or structural diversity.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Mechanistic studies reveal a sophisticated dual-pathway process where DMF contributes carbon atoms through two distinct routes during the iodine-promoted cyclization reaction. When the formyl group acts as the carbon source, it undergoes condensation with trifluoroethyliminohydrazide to form a hydrazone intermediate followed by intramolecular cyclization that eliminates dimethylamine to yield the triazole product with high regioselectivity. Alternatively, when the N-methyl group serves as the carbon source, DMF first reacts with iodine to generate an amine salt that eliminates hydrogen iodide before nucleophilic addition occurs with the hydrazide substrate; this pathway proceeds through elimination of N-methylformamide to create an azadiene intermediate which then undergoes intramolecular nucleophilic addition and oxidative aromatization to form the final product. The molecular iodine catalyst plays a crucial role in activating DMF for both pathways while maintaining reaction efficiency under mild atmospheric conditions without requiring additional oxidants or promoters.
The process inherently minimizes impurity formation through its self-regulating mechanism where competing pathways converge to produce consistent product profiles without generating persistent side products that require extensive purification. The absence of transition metal catalysts eliminates concerns about heavy metal contamination that typically necessitates additional purification steps in conventional syntheses; instead, simple post-treatment procedures involving filtration followed by column chromatography yield high-purity products suitable for pharmaceutical applications without specialized removal techniques. The reaction's tolerance to various functional groups including halogens and alkyl substituents prevents unwanted side reactions that could lead to impurity accumulation while maintaining consistent product quality across different substrate variations. This inherent selectivity reduces quality control challenges during scale-up by producing predictable impurity profiles that align with regulatory requirements for pharmaceutical intermediates.
How to Synthesize 3-Trifluoromethyl Substituted 1,2,4-Triazole Efficiently
This patented methodology provides a streamlined approach to producing high-value triazole intermediates through an operationally simple protocol that leverages readily available starting materials under standard manufacturing conditions. The process eliminates complex handling requirements by functioning effectively in ambient air without specialized equipment while maintaining compatibility with common production infrastructure found across chemical manufacturing facilities worldwide. Detailed standardized synthesis steps below outline the precise methodology developed through extensive optimization studies that ensure consistent product quality and yield across multiple production scales from laboratory validation through commercial implementation.
- Prepare the reaction mixture by combining trifluoroethyliminohydrazide with DMF solvent in a Schlenk tube under air atmosphere without requiring anhydrous or anaerobic conditions.
- Add molecular iodine (1.5 equivalents) and heat the mixture to 120°C for 12 hours to facilitate tandem cyclization through dual carbon-source pathways from DMF.
- Perform post-treatment including filtration, silica gel mixing, and column chromatography purification to isolate high-purity triazole products with diverse functional group tolerance.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis route directly addresses critical pain points in pharmaceutical intermediate supply chains by delivering enhanced operational flexibility while maintaining strict quality standards required for active pharmaceutical ingredient production. The elimination of specialized environmental controls reduces facility requirements while increasing production capacity utilization through simplified scheduling and reduced changeover times between different product runs.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes expensive heavy metal removal processes during purification while utilizing DMF as both solvent and carbon source reduces raw material costs; simplified processing steps decrease energy consumption and labor requirements without requiring capital-intensive equipment modifications or specialized operator training.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials with broad supplier options ensures consistent raw material availability while eliminating moisture-sensitive handling procedures reduces production delays; simplified process parameters enable rapid technology transfer between manufacturing sites without extensive revalidation requirements.
- Scalability and Environmental Compliance: The single-vessel operation minimizes waste generation compared to multi-step conventional routes while avoiding hazardous reagents simplifies environmental management; consistent performance across scales from laboratory validation through commercial production ensures reliable capacity expansion without process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address specific technical concerns raised by procurement teams regarding implementation feasibility and quality assurance protocols based on detailed analysis of patent CN114920707B's technical specifications and experimental validation data.
Q: How does this method overcome the need for anhydrous and anaerobic conditions in traditional triazole synthesis?
A: The process utilizes molecular iodine as a promoter that activates DMF under ambient air conditions while maintaining reaction efficiency through dual carbon-source pathways that eliminate moisture sensitivity typically required in conventional heterocyclic syntheses.
Q: What are the key advantages of using DMF as both solvent and carbon source in this process?
A: DMF serves as a dual-function reagent that simplifies process design by eliminating additional carbon sources while providing inherent stability under air atmosphere; its dual reactivity pathways enable broad substrate scope without requiring specialized equipment or handling procedures.
Q: How does the iodine-promoted reaction ensure high purity and scalability for pharmaceutical intermediates?
A: The mechanism avoids transition metal catalysts that create purification challenges while maintaining consistent yields across diverse substrates; this inherent simplicity enables seamless scale-up from laboratory to commercial production without complex process modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl Substituted 1,2,4-Triazole Supplier
This patented technology represents a significant advancement in triazole intermediate manufacturing that aligns perfectly with our company's expertise in complex molecule synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities for comprehensive quality verification.
We invite your technical procurement team to request our Customized Cost-Saving Analysis which details specific COA data and route feasibility assessments tailored to your production requirements; our specialists will work directly with your R&D team to optimize implementation timelines while ensuring seamless integration into your existing supply chain infrastructure.
