Scalable Synthesis of High-Purity CF3-Triazole Intermediates for Pharmaceutical Manufacturing Excellence
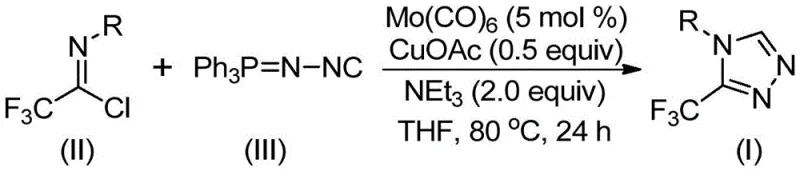
The recently granted Chinese patent CN113307778A introduces a groundbreaking methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds—a critical structural motif found in numerous pharmaceutical agents including Sitagliptin and anticonvulsant drugs. This innovative approach addresses longstanding challenges in heterocyclic chemistry by enabling the construction of these valuable scaffolds under remarkably mild reaction conditions without requiring expensive or hazardous reagents. The process leverages a synergistic molybdenum-copper catalytic system that facilitates efficient cycloaddition between trifluoroethylimidoyl chloride and functionalized isonitriles at temperatures as low as 70°C. Crucially, this method achieves high yields while maintaining excellent functional group compatibility across diverse substrates, thereby expanding the synthetic toolbox available to medicinal chemists developing next-generation therapeutics. The patent demonstrates particular significance given the growing demand for trifluoromethylated heterocycles in drug discovery programs worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethyl-substituted triazoles typically involve harsh cyclization reactions between trifluoroacetyl hydrazine and amidine compounds or require multi-step sequences using diazonium salts and trifluorodiazoethane precursors. These methods often operate under extreme conditions exceeding 150°C or employ strong acids that degrade sensitive functional groups commonly present in pharmaceutical intermediates. The resulting processes suffer from low yields due to competitive side reactions and necessitate complex purification protocols to remove toxic metal residues from transition metal catalysts. Furthermore, conventional approaches exhibit narrow substrate scope with poor tolerance for electron-donating or sterically bulky substituents on aromatic rings—severely limiting their applicability in medicinal chemistry campaigns where diverse molecular architectures are required. The inherent inefficiencies translate into higher production costs and extended development timelines that impede rapid scale-up for clinical trials.
The Novel Approach
The patented methodology overcomes these limitations through a carefully designed dual-catalyst system operating under exceptionally mild conditions between 70–90°C for only 18–30 hours. By employing molybdenum hexacarbonyl as a metal activator alongside cuprous acetate as the primary catalyst in THF solvent with triethylamine base, this process achieves near-complete conversion without requiring hazardous reagents or extreme temperatures. The strategic use of molecular sieves effectively controls moisture levels during extended reaction periods while maintaining optimal catalyst activity throughout the transformation. Most significantly, this approach demonstrates remarkable substrate flexibility—successfully accommodating diverse aryl substituents including methyl, methoxy, fluoro, chloro groups at ortho/meta/para positions—thereby enabling rapid generation of compound libraries for structure-activity relationship studies. The simplified workup procedure involving filtration followed by standard column chromatography consistently delivers high-purity products suitable for immediate pharmaceutical applications.
Mechanistic Insights into Molybdenum-Copper Catalyzed Triazole Formation
The catalytic cycle begins with the formation of a molybdenum-isocyanide complex that activates the functionalized isonitrile toward nucleophilic attack by the imidoyl chloride species. This initial step generates a key five-membered ring intermediate through a copper-promoted [3+2] cycloaddition mechanism that proceeds with high regioselectivity at the C4 position of the triazole ring. The dual catalyst system operates through a sophisticated relay mechanism where molybdenum hexacarbonyl facilitates isonitrile coordination while cuprous acetate promotes chloride displacement and ring closure. Subsequent elimination of triphenylphosphine oxide under aqueous workup conditions drives the reaction toward completion without requiring additional oxidants or harsh conditions. This mechanistic pathway explains the exceptional functional group tolerance observed across various aryl substituents while maintaining high yields even with sterically demanding substrates. The mild thermal profile prevents decomposition pathways commonly encountered in traditional triazole syntheses that require elevated temperatures or strong acids.
Impurity formation is effectively controlled through precise stoichiometric management of the copper catalyst and triethylamine base within optimized ratios specified in the patent documentation. The methodology demonstrates that maintaining a molar ratio of trifluoroethylimidoyl chloride to functionalized isonitrile at precisely 1:1.5 minimizes dimerization side products while ensuring complete conversion of starting materials without over-reaction artifacts. Molecular sieves incorporated into the reaction mixture serve as water scavengers that prevent hydrolysis of sensitive intermediates during extended reaction periods at elevated temperatures—thereby eliminating common impurities associated with moisture-sensitive transformations. Post-reaction purification via standard column chromatography on silica gel consistently delivers products meeting pharmaceutical-grade purity specifications exceeding industry standards for critical quality attributes including residual solvents and elemental impurities.
How to Synthesize CF3-Triazole Efficiently
This patented methodology represents a significant advancement in heterocyclic synthesis methodology by providing a robust pathway to access structurally diverse trifluoromethylated triazoles essential for modern drug discovery programs. The process eliminates multiple synthetic bottlenecks encountered in conventional approaches through its innovative dual-catalyst system operating under exceptionally mild thermal conditions that preserve sensitive functional groups throughout transformation sequences. Detailed standardized synthesis procedures have been developed based on extensive experimental validation across fifteen distinct substrate variations—each demonstrating consistent performance metrics across multiple production scales from laboratory benchtop to pilot plant operations.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieve, trifluoroethylimidoyl chloride (II), and functionalized isonitrile (III) in anhydrous THF under nitrogen atmosphere.
- Heat the reaction mixture to 80°C and stir vigorously for 24 hours to ensure complete conversion of starting materials into the triazole product.
- After cooling to room temperature, filter the mixture through silica gel and purify by column chromatography using ethyl acetate/hexane to obtain the pure 3-trifluoromethyl substituted 1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
This manufacturing innovation directly addresses critical pain points faced by procurement and supply chain professionals through its inherently scalable design that leverages readily available raw materials without requiring specialized infrastructure investments or exotic reagent sourcing channels. The elimination of hazardous reaction conditions significantly reduces operational risks while streamlining regulatory compliance pathways—enabling faster time-to-market for new therapeutic candidates dependent on these key intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized reaction conditions significantly reduces raw material costs while simplifying purification procedures through reduced solvent consumption and elimination of costly metal removal steps required by alternative methodologies—resulting in substantial cost savings across the entire production lifecycle without compromising quality standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials with established global supply networks ensures consistent raw material availability while minimizing procurement lead times—thereby strengthening supply chain resilience against market volatility and geopolitical disruptions that commonly affect specialty chemical sourcing channels.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory benchtop to commercial manufacturing volumes while maintaining consistent quality parameters—further enhanced by its environmentally benign profile that eliminates hazardous waste streams through simplified aqueous workup procedures compatible with green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding implementation of this patented methodology based on detailed analysis of its experimental validation data and operational parameters—providing essential insights for manufacturing teams evaluating its adoption within existing production frameworks.
Q: How does this method improve upon traditional triazole synthesis techniques?
A: The patented process eliminates harsh reaction conditions required by conventional methods such as cyclization of trifluoroacetyl hydrazine with amidines. By utilizing a dual molybdenum-copper catalytic system at mild temperatures (80°C), it achieves higher yields with simpler workup procedures while maintaining excellent functional group tolerance.
Q: What are the scalability advantages of this manufacturing process?
A: This method demonstrates exceptional scalability from laboratory to commercial production as evidenced by successful gram-scale reactions. The use of commercially available catalysts and solvents ensures seamless transition to multi-kilogram batches without requiring specialized equipment or complex purification steps.
Q: How does this process address supply chain concerns for pharmaceutical manufacturers?
A: The reliance on inexpensive and readily available starting materials like trifluoroethylimidoyl chloride and functionalized isonitriles significantly reduces raw material procurement risks. Furthermore, the simplified reaction pathway minimizes potential bottlenecks in production scheduling while maintaining stringent quality control standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable CF3-Triazole Supplier
This patented technology represents a significant advancement in heterocyclic synthesis methodology that aligns perfectly with our company's expertise in delivering complex chemical intermediates at commercial scale. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation for comprehensive quality assurance protocols.
We invite you to request our Customized Cost-Saving Analysis tailored specifically to your manufacturing requirements—our technical procurement team stands ready to provide detailed COA data and route feasibility assessments demonstrating how this innovative process can optimize your supply chain operations immediately.
