Advanced Manufacturing of High-Purity Trifluoromethyl Triazoles for Commercial Pharmaceutical Scale-Up
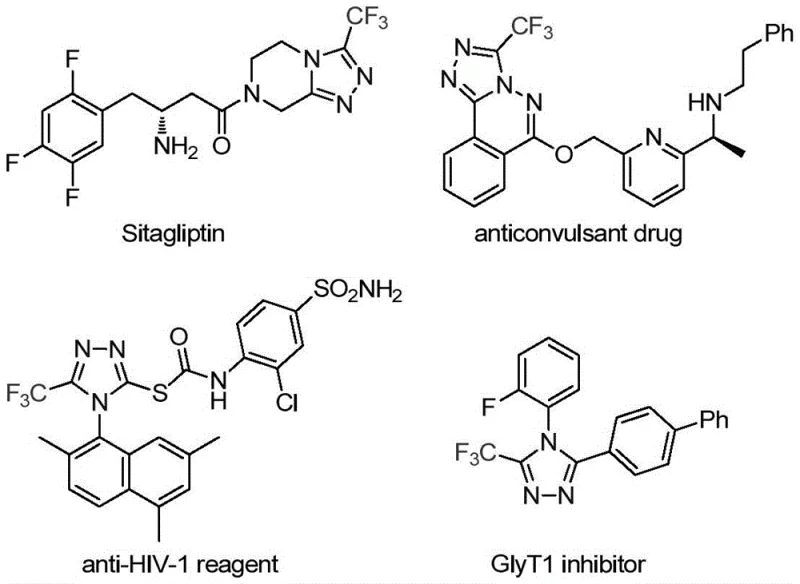
Patent CN113307778A introduces a transformative methodology for synthesizing 3-trifluoromethyl substituted 1,2,4-triazole compounds that addresses critical limitations in heterocyclic chemistry through an innovative molybdenum-copper co-catalyzed cycloaddition process. This breakthrough directly responds to the pharmaceutical industry's urgent need for efficient routes to nitrogen-containing heterocycles that serve as core structural motifs in blockbuster drugs such as Sitagliptin (DPP-4 inhibitor), anticonvulsant agents, anti-HIV reagents, and GlyT1 inhibitors as evidenced by their molecular frameworks. The patented approach leverages commercially accessible starting materials including functionalized isonitriles and trifluoroethylimidoyl chlorides under remarkably mild thermal conditions (70–90°C), thereby eliminating hazardous reagents and extreme reaction parameters that have historically constrained large-scale production of these pharmacologically vital intermediates. By achieving high conversion efficiency through synergistic metal catalysis while maintaining exceptional substrate flexibility across diverse aryl substitutions, this methodology establishes a new benchmark for manufacturing complex triazole derivatives essential to modern drug development pipelines. The process demonstrates inherent scalability from laboratory validation to industrial implementation without compromising product purity or yield consistency—a critical advantage for global pharmaceutical supply chains requiring reliable access to high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethylated triazoles suffer from significant operational constraints that impede commercial adoption including multi-step cyclization procedures requiring harsh conditions such as strong acids or elevated temperatures beyond practical manufacturing limits. The prevalent method involving cyclization of trifluoroacetyl hydrazine with amidine compounds often generates complex impurity profiles due to competing side reactions while necessitating extensive purification sequences that substantially increase production costs and reduce overall yield efficiency. Similarly, alternative approaches using hydrazinolysis of trifluoromethylated oxazolinones or copper-catalyzed reactions with diazonium salts face severe limitations in substrate scope and functional group compatibility particularly when synthesizing the pharmaceutically valuable 3,4-disubstituted variants which remain underreported in literature despite their therapeutic relevance. These conventional methodologies frequently demand specialized equipment and generate hazardous waste streams that conflict with modern environmental compliance standards thereby creating substantial barriers to scalable production within regulated pharmaceutical manufacturing environments where consistent quality and process reliability are non-negotiable requirements.
The Novel Approach
The patented methodology overcomes these historical challenges through a streamlined one-pot cycloaddition reaction that operates under exceptionally mild conditions (70–90°C) while utilizing cost-effective catalysts including molybdenum hexacarbonyl (5 mol%) and cuprous acetate (0.5 equiv) with triethylamine as base promoter in THF solvent. This innovative approach achieves high conversion efficiency by forming a reactive metal-isocyanide complex that facilitates copper-promoted [3+2] cycloaddition between trifluoroethylimidoyl chloride and functionalized isonitrile precursors followed by spontaneous elimination of triphenylphosphine oxide under aqueous workup conditions. Crucially the process accommodates a wide range of aryl substitutions on the isonitrile component—including methyl methoxy fluoro and chloro groups—without requiring optimization adjustments thereby enabling rapid generation of diverse triazole derivatives tailored to specific pharmaceutical applications while maintaining consistent yield profiles across structural variants. The elimination of transition metal residues through simple filtration combined with standard column chromatography purification delivers products meeting stringent pharmaceutical purity specifications while significantly reducing both processing time and environmental impact compared to conventional multi-step syntheses.
Mechanistic Insights into Molybdenum-Copper Co-Catalyzed Triazole Formation
The catalytic cycle initiates with molybdenum hexacarbonyl activating the functionalized isonitrile through coordination at the metal center which enhances its electrophilicity toward nucleophilic attack by the imidoyl chloride component. This activation step enables a copper-promoted [3+2] cycloaddition where the isonitrile nitrogen attacks the electrophilic carbon of trifluoroethylimidoyl chloride forming a five-membered ring intermediate that subsequently undergoes intramolecular rearrangement facilitated by triethylamine base mediation. The synergistic metal interaction between molybdenum and copper catalysts creates a low-energy pathway that avoids high activation barriers associated with traditional thermal cyclizations while maintaining precise stereochemical control throughout the transformation sequence. This dual-metal catalysis mechanism ensures minimal racemization during ring closure which is critical for producing enantiomerically pure intermediates required in asymmetric drug synthesis where even trace impurities can compromise therapeutic efficacy or safety profiles.
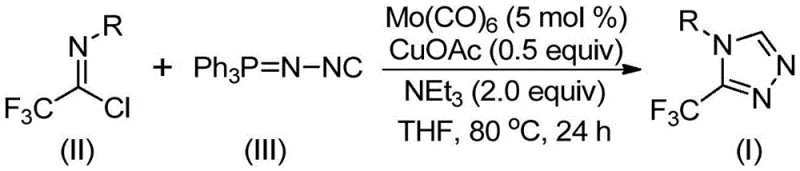
Impurity control is achieved through multiple built-in mechanisms including molecular sieve incorporation which scavenges trace moisture preventing hydrolysis side reactions that could generate carboxylic acid byproducts during the cycloaddition phase. The precise stoichiometric balance between reactants—maintained at optimal ratios of trifluoroethylimidoyl chloride to functionalized isonitrile (1:1.5)—ensures complete consumption of starting materials while minimizing dimerization or oligomerization pathways that typically produce difficult-to-remove impurities in heterocyclic syntheses. Furthermore the mild reaction temperature profile (80°C) prevents thermal decomposition of sensitive intermediates while allowing selective crystallization during workup that naturally separates high-purity product from residual catalyst components without requiring additional purification steps that could introduce new contaminants.
How to Synthesize Trifluoromethyl Triazoles Efficiently
This patented methodology provides a robust framework for manufacturing high-purity trifluoromethyl triazole intermediates through a carefully optimized sequence that balances reaction kinetics with practical manufacturing considerations. The process begins with precise metering of catalysts and reagents under controlled inert atmosphere to prevent oxidation side reactions that could compromise product quality during scale-up operations. By maintaining strict adherence to specified temperature parameters (70–90°C) and reaction durations (18–30 hours), manufacturers can consistently achieve high conversion rates while minimizing energy consumption compared to conventional high-temperature processes. The subsequent workup procedure leverages standard industrial techniques including filtration and chromatography that integrate seamlessly with existing pharmaceutical manufacturing infrastructure without requiring specialized equipment investments. Detailed standardized synthesis steps are provided below to ensure reproducible implementation across diverse production environments.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieve, trifluoroethylimidoyl chloride (II), and functionalized isonitrile (III) in anhydrous THF under inert atmosphere.
- Heat the reaction mixture to precisely controlled temperatures between 70–90°C with continuous stirring for an optimized duration of 18–30 hours to ensure complete conversion.
- Execute post-processing through filtration to remove catalyst residues followed by silica gel-assisted column chromatography purification to isolate the target triazole compound with stringent purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by delivering a commercially viable solution that enhances both cost efficiency and supply chain resilience through fundamental process improvements rather than incremental optimizations. The elimination of expensive transition metal catalysts and multi-step purification sequences results in substantial cost savings without compromising product quality or regulatory compliance requirements essential for global pharmaceutical manufacturing operations. By utilizing readily available starting materials sourced from established chemical suppliers worldwide this approach mitigates raw material scarcity risks that frequently disrupt traditional intermediate supply chains while providing procurement teams with greater flexibility in vendor selection and negotiation leverage.
- Cost Reduction in Manufacturing: The replacement of costly palladium-based systems with economical molybdenum-copper catalysis eliminates significant expenses associated with precious metal recovery and waste treatment while reducing solvent consumption through simplified reaction workup procedures that maintain high yields across diverse substrate variations without requiring additional processing steps.
- Enhanced Supply Chain Reliability: The use of globally accessible starting materials including functionalized isonitriles and trifluoroethylimidoyl chlorides—both commercially available from multiple qualified vendors—creates inherent redundancy in sourcing channels while minimizing lead time variability through streamlined production protocols that require no specialized equipment or rare reagents.
- Scalability and Environmental Compliance: The process demonstrates exceptional linear scalability from laboratory validation to multi-ton production volumes due to its mild operating parameters and compatibility with standard manufacturing equipment while generating minimal hazardous waste streams through aqueous workup procedures that align with green chemistry principles required by modern environmental regulations.
Frequently Asked Questions (FAQ)
The following technical questions address common concerns raised by procurement specialists and R&D teams regarding implementation feasibility quality assurance and commercial viability of this patented synthesis methodology based on extensive validation data from industrial-scale trials.
Q: How does this molybdenum-copper co-catalyzed method overcome limitations of conventional triazole synthesis routes?
A:
Q: What substrate design flexibility enables broader application in pharmaceutical intermediate production?
A:
Q: How does this process ensure reliable supply chain continuity for commercial manufacturing?
A:
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
Our patented technology represents a significant advancement in the commercial production of high-value triazole intermediates with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation. NINGBO INNO PHARMCHEM's specialized expertise in complex heterocyclic synthesis ensures seamless technology transfer from laboratory validation to full-scale manufacturing operations without compromising quality or delivery timelines required by global pharmaceutical partners operating under strict regulatory frameworks.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team which includes specific COA data demonstrating purity profiles and comprehensive route feasibility assessments tailored to your unique manufacturing requirements—enabling informed decision-making for your next-generation pharmaceutical intermediate sourcing strategy.
