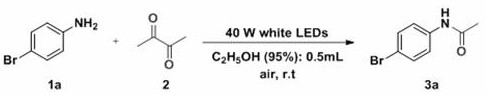
Scaling Green Visible Light Catalysis for High-Purity Acetamide Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking sustainable methodologies that align with green chemistry principles while maintaining high efficiency and product purity. Patent CN113717071A introduces a groundbreaking preparation method for green visible light catalyzed acetamide compounds, representing a significant shift from traditional thermal or metal-catalyzed processes. This technology utilizes economically available LED lamps as an energy source to drive the acetylation of aromatic and aliphatic amines using 2,3-butanedione as the acyl source. The innovation lies in its ability to operate under mild conditions without the need for expensive transition metal catalysts or harsh dehydrating agents, thereby addressing critical pain points in modern chemical manufacturing regarding cost, safety, and environmental impact. For R&D directors and procurement managers, this patent offers a viable pathway to streamline the synthesis of key acetamide intermediates found in numerous active pharmaceutical ingredients.
The limitations of conventional methods for amide bond formation have long hindered the efficiency of pharmaceutical intermediate manufacturing. Traditional approaches often rely on thermal reaction conditions that require high activation energy, leading to excessive energy consumption and potential degradation of sensitive functional groups. Furthermore, many established protocols necessitate the use of expensive metal catalysts such as palladium, ruthenium, or iridium, which not only inflate raw material costs but also introduce complex purification challenges to meet stringent residual metal specifications. Some methods employ hazardous reagents like sodium hydride or acetyl chloride, posing significant safety risks and requiring specialized handling equipment. These factors collectively contribute to extended lead times and reduced overall process robustness, making the supply chain vulnerable to disruptions and cost volatility.
In stark contrast, the novel approach detailed in the patent leverages visible light catalysis to overcome these traditional barriers. By employing a 40W white LED lamp, the reaction achieves sufficient activation energy at room temperature, eliminating the need for external heating and significantly reducing energy overheads. The system is remarkably simple, utilizing air as the atmosphere and 95% ethanol as a benign solvent, which simplifies waste treatment and enhances operator safety. The absence of metal catalysts means there is no risk of metal contamination in the final product, a critical advantage for pharmaceutical applications. Additionally, the use of 2,3-butanedione as a stable and commercially available acyl source avoids the handling issues associated with reactive acid chlorides, ensuring a more stable and predictable supply chain for raw materials.
Mechanistic Insights into Visible Light Catalyzed Acetylation
The mechanistic pathway of this visible light catalyzed reaction offers profound implications for process chemistry and impurity control. Unlike thermal reactions that rely on heat to overcome activation barriers, this photochemical process utilizes photon energy to excite the reaction components, specifically facilitating the interaction between the amine and the diketone. The 2,3-butanedione acts as both the reactant and a photosensitizer in certain contexts, absorbing visible light to generate reactive intermediates that selectively attack the amine nitrogen. This selectivity is crucial for complex molecules containing multiple reactive sites, as it minimizes side reactions and byproduct formation. The mild nature of the photochemical excitation ensures that sensitive functional groups on the amine substrate, such as halogens or esters, remain intact, thereby preserving the structural integrity required for downstream drug synthesis.
Impurity control is inherently enhanced by the simplicity of the reaction system. Since no metal catalysts are introduced, the impurity profile is devoid of heavy metal residues, which are notoriously difficult to remove and strictly regulated in pharmaceutical products. The reaction proceeds cleanly in ethanol, a solvent that is easily removed and recycled, further reducing the risk of solvent-related impurities. The broad substrate scope demonstrated in the patent, covering primary, secondary, and tertiary amines as well as various aromatic substitutions, indicates a robust mechanism that tolerates electronic and steric variations. This versatility allows chemists to apply the same standardized conditions to a wide range of intermediates, simplifying process development and reducing the time required for optimization. The result is a high-purity product stream that requires minimal downstream processing.
How to Synthesize Acetamide Compound Efficiently
The synthesis of acetamide compounds using this green visible light catalysis strategy is designed for operational simplicity and scalability. The process begins with the preparation of a reaction mixture containing the amine substrate, 2,3-butanedione, and 95% ethanol in a standard reaction vessel. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and reaction times optimized for maximum yield. This protocol eliminates the need for inert gas protection or anhydrous conditions, further reducing operational complexity. The reaction is driven by continuous irradiation with a white LED source, ensuring consistent energy input throughout the process. Upon completion, the workup involves a straightforward extraction and purification sequence that is compatible with existing industrial infrastructure.
- Prepare the reaction mixture by combining the amine substrate, 2,3-butanedione (6.0 equivalents), and 95% ethanol solvent in a Schlenk tube under air atmosphere.
- Irradiate the reaction mixture with a 40W white LED lamp at room temperature while stirring conventionally for approximately 6 hours to initiate the photochemical acetylation.
- Quench the reaction with saturated sodium sulfite solution, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography to isolate the acetamide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible light catalyzed synthesis offers substantial strategic advantages in terms of cost structure and supply reliability. The elimination of expensive transition metal catalysts directly reduces the bill of materials, while the use of commodity chemicals like 2,3-butanedione and ethanol ensures stable pricing and easy sourcing. The mild reaction conditions reduce the demand on utility infrastructure, as no high-temperature heating or cryogenic cooling is required, leading to lower operational expenditures. Furthermore, the safety profile of the process is significantly improved by avoiding hazardous reagents, which can lower insurance costs and reduce regulatory compliance burdens. These factors combine to create a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts such as palladium or ruthenium from the synthesis route results in significant raw material cost savings. Additionally, the energy efficiency of LED lighting compared to thermal heating systems reduces utility costs over the lifecycle of the production process. The simplified workup procedure minimizes solvent consumption and waste disposal fees, contributing to a lower overall cost of goods sold. These economic benefits allow for more competitive pricing strategies in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Relying on commercially available and stable raw materials like 2,3-butanedione reduces the risk of supply disruptions associated with specialized or imported reagents. The robustness of the reaction under air atmosphere means that production is less sensitive to environmental fluctuations or equipment failures related to inert gas systems. This reliability ensures consistent delivery schedules and helps maintain inventory levels required for continuous manufacturing operations. The scalability of the process from laboratory to commercial scale further secures long-term supply continuity for key customers.
- Scalability and Environmental Compliance: The green nature of this synthesis aligns with increasingly stringent environmental regulations regarding waste generation and solvent use. The use of ethanol as a primary solvent facilitates easier recycling and treatment compared to chlorinated or aromatic solvents. The absence of heavy metals simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. This compliance advantage not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light catalyzed acetamide synthesis. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the feasibility and benefits of adopting this technology for industrial applications. Understanding these details is essential for making informed decisions about process integration and supply chain optimization.
Q: Does this visible light catalysis method require expensive metal catalysts?
A: No, the patented process described in CN113717071A operates without any transition metal catalysts such as palladium, ruthenium, or iridium, significantly reducing raw material costs and eliminating the need for complex metal removal steps.
Q: What are the reaction conditions for synthesizing acetamide compounds using this method?
A: The reaction proceeds under mild conditions using a 40W white LED light source at room temperature in an air atmosphere, utilizing 95% ethanol as a green solvent, which simplifies operational safety and energy consumption.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process features simple operation, cheap and easily available raw materials like 2,3-butanedione and amines, and avoids harsh conditions, making it highly scalable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetamide Compound Supplier
The technological potential of visible light catalysis for acetamide synthesis represents a significant opportunity for optimizing pharmaceutical intermediate production. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle photochemical reactions safely and efficiently, ensuring that the benefits of this green chemistry approach are fully realized at scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest quality standards required by global regulatory bodies. Our commitment to technical excellence ensures that complex synthetic routes are translated into robust commercial processes.
We invite you to explore how this innovative synthesis route can enhance your supply chain efficiency and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to advanced chemical technologies and a reliable supply network dedicated to supporting your long-term growth and success in the competitive pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
