Advanced Rhodium-Catalyzed Aminocarbonylation for Scalable Acetamide Production
Advanced Rhodium-Catalyzed Aminocarbonylation for Scalable Acetamide Production
The landscape of organic synthesis is undergoing a significant transformation driven by the urgent need for greener, more sustainable methodologies that do not compromise on efficiency or yield. A pivotal development in this arena is detailed in patent CN112812032A, which discloses a novel preparation method for acetamide compounds utilizing a rhodium-catalyzed aminocarbonylation strategy. This technology represents a paradigm shift by employing dimethyl carbonate (DMC) not merely as a benign solvent but as a critical C1 building block, thereby addressing multiple pain points associated with traditional carbonylation and methylation reactions. For R&D directors and process chemists, this approach offers a robust pathway to construct valuable amide bonds while adhering to strict environmental standards. The protocol leverages a sophisticated catalytic system involving dirhodium tetracarbonyl dichloride and specific phosphine ligands to activate nitro compounds, transforming them into high-value acetamide derivatives with exceptional selectivity. This innovation is particularly relevant for the production of pharmaceutical intermediates where purity and regulatory compliance are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acetamide compounds and related carbonyl-containing structures has relied heavily on reagents that pose significant safety and environmental hazards. Traditional methylating agents such as diazomethane, dimethyl sulfate, and methyl iodide are frequently employed, yet they suffer from severe drawbacks including explosiveness, high toxicity, and corrosiveness. Furthermore, these conventional pathways often necessitate the use of stoichiometric amounts of strong bases to drive the reaction forward, which inevitably leads to the generation of substantial quantities of useless inorganic salts that require complex and costly disposal procedures. In the context of industrial carbonylation, processes like the Monsanto acetic acid synthesis utilize hydrogen iodide as a cocatalyst, which creates severe corrosive effects on reactor equipment, driving up maintenance costs and posing safety risks. These legacy methods create a bottleneck for modern chemical manufacturing, where the pressure to reduce carbon footprints and eliminate hazardous waste streams is intensifying. Consequently, there is a critical demand for alternative synthetic routes that can bypass these dangerous reagents while maintaining high reaction efficiency and substrate compatibility.
The Novel Approach
The methodology outlined in the patent data introduces a groundbreaking solution by utilizing dimethyl carbonate as a dual-function reagent, acting simultaneously as a green solvent and the source of the carbonyl and methyl groups. This innovative route circumvents the need for toxic methylating agents and corrosive promoters, offering a much safer operational profile for chemical plants. The reaction proceeds under relatively mild thermal conditions, typically around 120°C, utilizing a rhodium-based catalytic system that demonstrates remarkable versatility. Unlike older methods that might struggle with sensitive functional groups, this new approach exhibits a wide tolerance range, successfully accommodating substrates with alkoxy, trifluoromethyl, halogen, and thiomethyl substituents. The simplicity of the operation is another key advantage; the process involves mixing readily available starting materials in a sealed vessel, reacting for a defined period, and performing straightforward post-treatment steps. This streamlines the workflow significantly, reducing the complexity of unit operations required to isolate the final product. By integrating sustainability with high performance, this method sets a new standard for the synthesis of acetamide compounds in fine chemistry.
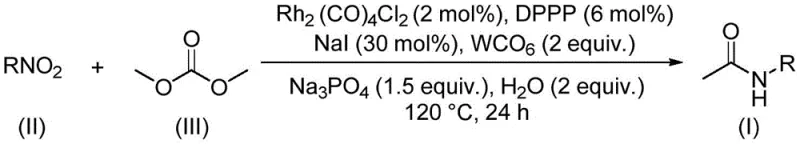
Mechanistic Insights into Rhodium-Catalyzed Aminocarbonylation
At the heart of this transformative synthesis lies a complex catalytic cycle driven by the synergistic interaction between the rhodium precursor and the phosphine ligand. The system employs dirhodium tetracarbonyl dichloride (Rh2(CO)4Cl2) in conjunction with 1,3-bis(diphenylphosphino)propane (DPPP) to generate the active catalytic species in situ. Tungsten hexacarbonyl (W(CO)6) serves as an additional carbonyl source or promoter, ensuring a sufficient concentration of CO species within the reaction medium to facilitate the carbonylation step. Sodium iodide plays a crucial role as a promoter, likely assisting in the activation of the nitro group or the dimethyl carbonate through halide exchange mechanisms. The presence of water and sodium phosphate helps to maintain the necessary pH balance and may participate in the reduction of the nitro group to the corresponding amine intermediate, which then undergoes carbonylation. This multi-component catalytic environment allows for the direct conversion of nitroarenes and nitroalkanes into acetamides without the need for isolating unstable amine intermediates. The mechanistic elegance of this system ensures that the reaction proceeds with high atom economy, minimizing side reactions and maximizing the yield of the desired acetamide product.
From an impurity control perspective, the mild nature of this rhodium-catalyzed process is instrumental in achieving high-purity profiles essential for pharmaceutical applications. Traditional harsh conditions often lead to over-alkylation, decomposition of sensitive functional groups, or the formation of polymeric byproducts. In contrast, the use of dimethyl carbonate under these specific catalytic conditions provides a controlled environment where the reactivity is tuned precisely. The functional group tolerance observed across various substrates, including those with electron-withdrawing groups like trifluoromethyl and electron-donating groups like ethoxy, suggests that the catalytic cycle is robust against electronic variations. This stability reduces the formation of structurally related impurities that are difficult to separate. Furthermore, the absence of corrosive acids like HI prevents equipment degradation, which can be a source of metal contamination in the final product. The post-treatment procedure, involving simple filtration and silica gel mixing followed by column chromatography, is highly effective at removing residual catalyst and inorganic salts, ensuring that the final acetamide compound meets stringent quality specifications required for downstream drug synthesis.
How to Synthesize Acetamide Compounds Efficiently
The practical implementation of this synthesis route is designed to be accessible for both laboratory-scale optimization and pilot-plant production. The protocol dictates a precise molar ratio of components to ensure optimal catalytic turnover, specifically utilizing a ratio of rhodium catalyst to ligand to additives that maximizes efficiency while minimizing precious metal usage. The reaction is typically conducted in a sealed tube or autoclave capable of withstanding the pressure generated at 120°C over a 24-hour period. This duration allows for the complete consumption of the nitro starting material, as evidenced by the high yields reported across a broad scope of substrates. Following the reaction, the workup is remarkably uncomplicated; the mixture is filtered to remove insoluble residues, mixed with silica gel to adsorb polar impurities, and purified via standard column chromatography techniques. This straightforward isolation procedure underscores the practicality of the method for generating diverse libraries of acetamide derivatives for biological screening or process development.
- Combine rhodium catalyst, ligand, tungsten carbonyl, sodium phosphate, sodium iodide, water, nitro compound, and dimethyl carbonate in a sealed vessel.
- Heat the reaction mixture to 120°C and maintain stirring for 24 hours to ensure complete conversion.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the target acetamide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this rhodium-catalyzed technology offers compelling economic and logistical benefits that extend beyond simple yield metrics. The primary driver for cost reduction lies in the substitution of hazardous, expensive, and heavily regulated reagents with dimethyl carbonate, which is not only inexpensive but also derived from sustainable sources like carbon dioxide. By eliminating the need for specialized corrosion-resistant equipment required for processes using hydrogen iodide or methyl iodide, capital expenditure for plant infrastructure can be significantly lowered. Additionally, the reduction in hazardous waste generation translates directly into lower disposal costs and reduced regulatory burden, simplifying the compliance landscape for manufacturing sites. The use of cheap and widely available nitro compounds as starting materials further stabilizes the supply chain, reducing dependency on niche intermediates that may be subject to market volatility. This resilience is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the dual functionality of dimethyl carbonate, which removes the necessity for separate solvents and C1 sources, thereby consolidating material costs. The avoidance of stoichiometric bases means that there is no generation of massive amounts of inorganic salt waste, which drastically cuts down on waste treatment expenses. Furthermore, the high reaction efficiency and yield minimize the loss of valuable starting materials, ensuring that the cost per kilogram of the final acetamide product is optimized. The ability to recycle methanol and carbon dioxide released during the reaction back into dimethyl carbonate synthesis adds a layer of circular economy benefit that can further enhance long-term cost savings.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical factor for any commercial chemical operation, and this method excels by relying on commodity chemicals that are produced on a massive global scale. Nitro compounds and dimethyl carbonate are standard inventory items for most chemical suppliers, reducing the risk of supply disruptions that can occur with specialized or custom-synthesized reagents. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain fluctuations. This reliability allows procurement teams to negotiate better terms with vendors and secure long-term contracts with confidence, knowing that the production process is not vulnerable to single-source bottlenecks.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and safety, but the moderate temperature and pressure requirements of this synthesis make it inherently safer and easier to scale up. The green chemistry credentials of using a biodegradable solvent and non-toxic reagents align perfectly with increasingly strict environmental regulations, future-proofing the manufacturing process against tighter emission standards. The simplified post-treatment workflow reduces the number of unit operations required, which shortens the overall production cycle time and increases throughput capacity. This scalability ensures that the technology can grow with market demand, from initial clinical trial batches to full commercial production without the need for fundamental process redesigns.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is essential for stakeholders evaluating its potential integration into existing manufacturing portfolios. The following questions address common inquiries regarding the operational parameters, substrate scope, and commercial viability of the rhodium-catalyzed aminocarbonylation process. These insights are derived directly from the experimental data and technical disclosures found in the patent literature, providing a factual basis for decision-making. Whether you are concerned about the compatibility of specific functional groups or the logistics of raw material sourcing, the answers below aim to clarify the practical implications of adopting this technology.
Q: What are the primary advantages of using dimethyl carbonate in this synthesis?
A: Dimethyl carbonate serves a dual role as both a green solvent and a C1 source, eliminating the need for toxic methylating agents like methyl iodide and reducing inorganic salt waste.
Q: What is the functional group tolerance of this rhodium-catalyzed method?
A: The method exhibits excellent tolerance for various substituents including alkoxy, trifluoromethyl, halogens, and thiomethyl groups on aryl rings, allowing for diverse derivative synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of cheap, commercially available raw materials and simple post-treatment procedures like filtration and chromatography makes it highly scalable for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acetamide Compound Supplier
As the demand for high-quality pharmaceutical intermediates continues to rise, partnering with a manufacturer that possesses both technical expertise and scalable capacity is vital for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the rhodium-catalyzed aminocarbonylation described in patent CN112812032A to deliver superior products. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of acetamide compound meets the highest international standards. Our commitment to quality assurance means that you can rely on us for consistent supply and technical support throughout your product lifecycle.
We invite you to explore how our capabilities can enhance your supply chain and reduce your overall manufacturing costs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, helping you identify opportunities for efficiency and budget optimization. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. Let us help you navigate the complexities of chemical synthesis with a partner dedicated to innovation, reliability, and mutual growth.
