Advanced Ruthenium-Catalyzed Synthesis of 3-Acylpyrrole Intermediates for Commercial Scale-Up
Introduction to Next-Generation Pyrrole Synthesis
The pyrrole scaffold represents one of the most ubiquitous and pharmacologically significant motifs in modern medicinal chemistry, serving as the core structure for a vast array of bioactive molecules ranging from histone deacetylase inhibitors to HMG-CoA reductase inhibitors. Within this family, 3-acylpyrrole derivatives stand out as star molecules due to their unique electronic properties and versatile utility as synthetic building blocks for complex functional materials and active pharmaceutical ingredients. However, the efficient construction of the 3-acylpyrrole core has historically presented significant challenges in organic synthesis, often requiring multi-step sequences or harsh reaction conditions that limit industrial applicability. The recent disclosure in patent CN113636968B introduces a groundbreaking synthetic methodology that addresses these longstanding limitations through an elegant transfer hydrogenation strategy. This innovative approach leverages a ruthenium-catalyzed tandem reaction between alpha-amino alcohols and alpha,beta-unsaturated alkynones, offering a streamlined pathway that combines high atom economy with operational simplicity. For pharmaceutical manufacturers and R&D teams, this technology represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-acylpyrrole compounds has relied heavily on methodologies that suffer from significant drawbacks regarding safety, cost, and environmental impact. Traditional routes often involve the use of stoichiometric amounts of toxic oxidants, such as IBX (2-iodoxybenzoic acid), which generate substantial quantities of hazardous waste and complicate the purification process. Other established methods utilize palladium catalysis in conjunction with acidic chlorides and terminal alkynes, which not only increases the raw material costs due to the high price of palladium but also necessitates rigorous removal of residual heavy metals to meet stringent pharmaceutical quality standards. Furthermore, many conventional protocols require multi-step sequences involving the isolation of unstable intermediates, leading to lower overall yields and increased production lead times. The reliance on harsh acidic conditions or sensitive reagents like aminoacetaldehyde diethyl acetals further restricts the substrate scope, making it difficult to introduce diverse functional groups without protecting group strategies. These cumulative inefficiencies create bottlenecks in the supply chain, driving up the cost of goods sold and limiting the ability to rapidly scale production for clinical or commercial demands.
The Novel Approach
In stark contrast to these legacy methods, the novel technique described in the patent data utilizes a sophisticated one-pot tandem reaction driven by a triruthenium dodecacarbonyl catalyst system. This approach fundamentally reimagines the bond construction process by employing a hydrogen transfer mechanism where ethanol serves as both the solvent and the hydrogen source, eliminating the need for external high-pressure hydrogen gas or dangerous chemical reducing agents. The reaction proceeds through a seamless sequence where the alpha-amino alcohol is dehydrogenated in situ to form a reactive amino carbonyl species, which subsequently condenses with the alkynone and undergoes cyclization to form the target pyrrole ring. 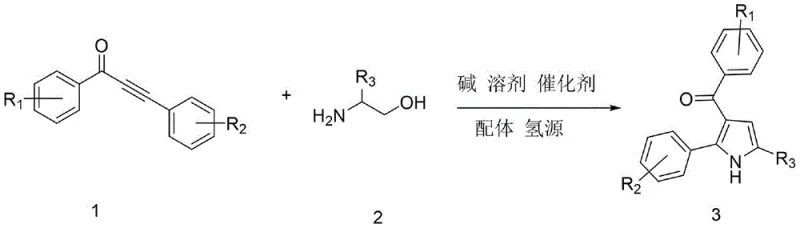 This transformation is notable for its exceptional step economy, converting simple, commercially available starting materials directly into complex heterocycles with high efficiency. The use of a robust ruthenium catalyst paired with a 4-methyl-1,10-phenanthroline ligand ensures high turnover numbers and excellent selectivity, minimizing the formation of byproducts. By operating under relatively mild thermal conditions initially followed by a higher temperature cyclization phase, this method achieves yields comparable to or exceeding traditional methods while drastically simplifying the workup procedure. This technological leap provides a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in terms of process robustness and environmental compliance.
This transformation is notable for its exceptional step economy, converting simple, commercially available starting materials directly into complex heterocycles with high efficiency. The use of a robust ruthenium catalyst paired with a 4-methyl-1,10-phenanthroline ligand ensures high turnover numbers and excellent selectivity, minimizing the formation of byproducts. By operating under relatively mild thermal conditions initially followed by a higher temperature cyclization phase, this method achieves yields comparable to or exceeding traditional methods while drastically simplifying the workup procedure. This technological leap provides a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in terms of process robustness and environmental compliance.
Mechanistic Insights into Ru-Catalyzed Transfer Hydrogenation Cyclization
The success of this synthetic route lies in the intricate mechanistic interplay between the ruthenium catalyst and the substrates, facilitating a borrowing hydrogen pathway that drives the thermodynamic equilibrium towards product formation. The catalytic cycle initiates with the dehydrogenation of the alpha-amino alcohol substrate by the ruthenium center, generating a reactive alpha-amino ketone or aldehyde intermediate along with a ruthenium-hydride species. This dehydrogenation step is critical as it activates the amine component for subsequent nucleophilic attack without the need for pre-oxidation reagents. The generated amino carbonyl species then undergoes a condensation reaction with the electron-deficient alpha,beta-unsaturated alkynone, forming an enamine intermediate that is primed for cyclization. The presence of the base, typically potassium carbonate, facilitates the deprotonation steps necessary for the intramolecular nucleophilic attack of the nitrogen onto the alkyne moiety.  Following the cyclization event, the ruthenium-hydride species formed in the initial step transfers hydrogen back to the system, aromatizing the pyrrole ring and regenerating the active catalyst. This closed-loop hydrogen transfer mechanism ensures that no external oxidants or reductants are consumed, resulting in water and acetaldehyde (from ethanol) as the primary byproducts. The ligand environment provided by 4-methyl-1,10-phenanthroline stabilizes the ruthenium center against decomposition at the elevated temperatures required for the final cyclization step, ensuring consistent catalytic performance throughout the 18-hour reaction window. Understanding this mechanism is vital for R&D directors aiming to optimize the process further, as it highlights the importance of maintaining anhydrous conditions and precise temperature control to maximize the efficiency of the hydrogen borrowing cycle.
Following the cyclization event, the ruthenium-hydride species formed in the initial step transfers hydrogen back to the system, aromatizing the pyrrole ring and regenerating the active catalyst. This closed-loop hydrogen transfer mechanism ensures that no external oxidants or reductants are consumed, resulting in water and acetaldehyde (from ethanol) as the primary byproducts. The ligand environment provided by 4-methyl-1,10-phenanthroline stabilizes the ruthenium center against decomposition at the elevated temperatures required for the final cyclization step, ensuring consistent catalytic performance throughout the 18-hour reaction window. Understanding this mechanism is vital for R&D directors aiming to optimize the process further, as it highlights the importance of maintaining anhydrous conditions and precise temperature control to maximize the efficiency of the hydrogen borrowing cycle.
From an impurity control perspective, this mechanism offers significant advantages over oxidative cyclization methods. Since the reaction does not involve strong oxidants, there is a marked reduction in the formation of over-oxidized byproducts or polymeric tars that often plague pyrrole syntheses. The specificity of the ruthenium catalyst for the transfer hydrogenation pathway ensures that side reactions such as homocoupling of the alkyne or polymerization of the enone are minimized. Furthermore, the use of tert-amyl alcohol as a co-solvent provides a high-boiling medium that supports the high-temperature cyclization step while maintaining good solubility for both polar and non-polar intermediates. This solubility profile prevents the precipitation of intermediates which could otherwise lead to incomplete reactions or difficult-to-remove solid impurities. The result is a crude product profile that is significantly cleaner than those obtained from palladium-catalyzed or IBX-mediated routes, thereby reducing the burden on downstream purification units. For quality control teams, this translates to a more predictable impurity profile and easier validation of the cleaning procedures for manufacturing equipment, ultimately supporting the production of high-purity OLED material or pharmaceutical intermediates with minimal batch-to-batch variation.
How to Synthesize 3-Acylpyrrole Efficiently
The practical implementation of this synthesis involves a carefully orchestrated sequence of mixing, heating, and purification steps designed to maximize yield and safety in a standard laboratory or pilot plant setting. The process begins with the charging of a Schlenk tube or reactor with the requisite molar equivalents of the alpha-amino alcohol and the alpha,beta-unsaturated alkynone, ensuring a precise 1:1 stoichiometry to prevent the accumulation of unreacted starting materials. To this mixture, the catalytic system comprising triruthenium dodecacarbonyl and the phenanthroline ligand is added, followed by the introduction of the hydrogen source, ethanol, and the base, potassium carbonate. The choice of tert-amyl alcohol as the primary solvent is critical, as it provides the necessary thermal stability for the subsequent high-temperature phase while dissolving the inorganic base effectively. Detailed standardized synthesis steps see the guide below.
- Charge the reactor with alpha-amino alcohol, alpha,beta-unsaturated alkynone, triruthenium dodecacarbonyl catalyst, 4-methyl-1,10-phenanthroline ligand, ethanol, potassium carbonate base, and tert-amyl alcohol solvent.
- Stir the mixture at 80°C under nitrogen protection for 1.5 hours to initiate the dehydrogenation and condensation phases.
- Raise the temperature to 150°C and maintain for 18 hours to complete the cyclization, then cool, filter, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed transfer hydrogenation technology offers profound strategic benefits that extend beyond mere chemical efficiency. The primary driver for cost optimization lies in the substitution of expensive and hazardous reagents with commodity chemicals; ethanol serves as a cheap, renewable hydrogen donor, replacing the need for high-pressure hydrogen infrastructure or costly silane reducing agents. Additionally, the elimination of stoichiometric oxidants like IBX removes a significant line item from the raw material budget and drastically reduces the cost associated with hazardous waste disposal. The one-pot nature of the reaction consolidates multiple synthetic transformations into a single vessel operation, which significantly reduces labor costs, energy consumption, and equipment occupancy time. These factors combine to deliver substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing in tender processes without compromising on margin. The robustness of the catalyst system also implies longer campaign runs with less frequent catalyst replenishment, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of earth-abundant iron-group metals like ruthenium in low catalytic loadings, avoiding the extreme price volatility associated with palladium or platinum group metals. By utilizing ethanol as the hydrogen source, the process bypasses the capital expenditure required for specialized high-pressure hydrogenation reactors, allowing production to occur in standard glass-lined or stainless steel vessels. The simplified workup procedure, which involves basic filtration and distillation rather than complex extractions or chromatographic separations on a large scale, reduces solvent consumption and processing time. Consequently, the overall cost of goods is significantly lowered, providing a buffer against raw material price fluctuations and enabling more aggressive pricing strategies for bulk orders.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the reliance on readily available starting materials such as substituted acetophenones and amino alcohols, which are produced globally at massive scales. Unlike specialized reagents that may have single-source suppliers or long lead times, the key inputs for this synthesis are commodity chemicals with stable supply networks. The operational simplicity of the reaction, which tolerates a wide range of functional groups without the need for sensitive protecting groups, reduces the risk of batch failures due to raw material variability. This reliability ensures consistent delivery schedules for clients, reducing the lead time for high-purity pharmaceutical intermediates and mitigating the risk of production stoppages. Furthermore, the absence of highly toxic reagents simplifies logistics and storage requirements, lowering the regulatory burden on the supply chain.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns perfectly with modern green chemistry principles and increasingly stringent regulatory frameworks. The atom economy of the transfer hydrogenation is superior to oxidative methods, generating minimal waste and reducing the E-factor of the process. The use of tert-amyl alcohol and ethanol, which are less toxic and more biodegradable than chlorinated solvents often used in heterocycle synthesis, facilitates easier wastewater treatment and solvent recovery. The process is inherently safer as it avoids the handling of pyrophoric reagents or high-pressure gas cylinders, reducing the risk of industrial accidents. This safety profile, combined with the reduced environmental footprint, makes the commercial scale-up of complex heterocycles much smoother, as it faces fewer hurdles during environmental impact assessments and regulatory audits.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the technical specifications and operational parameters of this synthesis. The following responses are derived directly from the experimental data and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance for process development teams. These insights address critical concerns regarding catalyst performance, substrate flexibility, and purification requirements that are paramount for successful technology transfer.
Q: What are the primary advantages of this Ru-catalyzed method over traditional Pd-catalyzed routes?
A: Unlike traditional methods that often require expensive palladium catalysts, toxic oxidants like IBX, or harsh acidic conditions, this novel approach utilizes a ruthenium-catalyzed transfer hydrogenation strategy. It employs ethanol as a green hydrogen source and operates under neutral to basic conditions, significantly reducing toxic waste generation and simplifying the downstream purification process for high-purity intermediates.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It utilizes readily available raw materials such as alpha-amino alcohols and alkynones, and avoids the use of high-pressure hydrogen gas cylinders by using ethanol as the hydrogen donor. The one-pot tandem nature of the reaction minimizes unit operations, making it highly attractive for commercial scale-up of complex heterocycles in a GMP environment.
Q: What is the functional group tolerance of this catalytic system?
A: The catalytic system demonstrates excellent functional group compatibility. It successfully tolerates various substituents on the aromatic rings, including electron-withdrawing groups like trifluoromethyl and halogens, as well as electron-donating groups like methyl and methoxy. This broad substrate scope allows for the efficient synthesis of diverse 3-acylpyrrole derivatives required for medicinal chemistry optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylpyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team of process chemists has extensively analyzed the potential of this ruthenium-catalyzed route, confirming its viability for the production of high-purity 3-acylpyrrole compounds required in next-generation drug discovery programs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of this transfer hydrogenation chemistry are perfectly translated to the plant floor. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, delivering intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your upcoming projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and target cost structures. We encourage you to reach out today to request specific COA data for our existing library of pyrrole derivatives or to discuss route feasibility assessments for your proprietary targets. Let us help you accelerate your development timeline and secure a sustainable supply chain for your critical intermediates.
