Advanced Ruthenium-Catalyzed Synthesis of 3-Acylpyrrole Intermediates for Commercial Scale-Up
Advanced Ruthenium-Catalyzed Synthesis of 3-Acylpyrrole Intermediates for Commercial Scale-Up
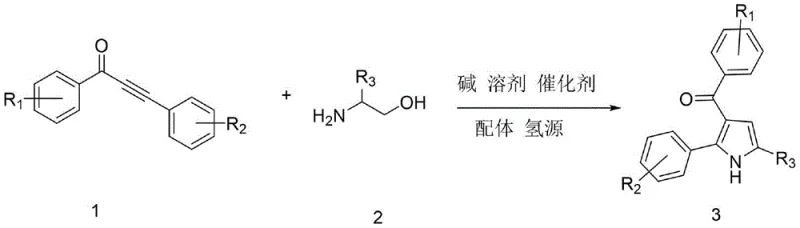
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex heterocyclic scaffolds, particularly the versatile 3-acylpyrrole motif found in numerous bioactive molecules. Patent CN113636968B introduces a groundbreaking synthetic methodology that leverages a ruthenium-catalyzed hydrogen transfer strategy to assemble these valuable structures from readily available precursors. This innovation represents a significant departure from traditional multi-step syntheses, offering a streamlined one-pot procedure that operates under relatively mild conditions while maintaining high functional group tolerance. By utilizing alpha-amino alcohols and alpha,beta-unsaturated alkynones as starting materials, this process not only simplifies the operational workflow but also aligns with modern green chemistry principles by minimizing waste generation. For R&D directors and process chemists, this patent provides a robust blueprint for accessing diverse pyrrole derivatives that serve as critical intermediates in the development of histone deacetylase inhibitors and other therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 3-acylpyrrole skeletons has relied on methodologies that suffer from significant drawbacks regarding scalability and environmental impact. Traditional approaches often involve the use of stoichiometric oxidants such as IBX (2-iodoxybenzoic acid) to mediate intramolecular oxidative cyclization, which generates substantial amounts of iodine-containing waste and requires expensive reagents that drive up production costs. Other established routes utilize palladium catalysis with toxic phosphine ligands or require harsh acidic conditions that limit substrate scope and complicate downstream purification. Furthermore, many classical syntheses are multi-step processes involving the isolation of unstable intermediates, which increases the risk of yield loss and extends the overall manufacturing lead time. These inefficiencies create bottlenecks for supply chain managers who require reliable, high-volume production of pharmaceutical intermediates without the burden of complex waste treatment protocols or the volatility of precious metal markets.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113636968B employs a sophisticated tandem reaction sequence driven by a triruthenium dodecacarbonyl catalyst system. This novel approach utilizes a hydrogen borrowing mechanism where ethanol serves as both a solvent component and a hydrogen source, effectively eliminating the need for external high-pressure hydrogen gas or hazardous reducing agents. The reaction proceeds through a seamless cascade where the initial condensation of the amino alcohol and alkynone is followed by cyclization and subsequent reduction, all occurring within a single reactor vessel. This one-pot strategy drastically reduces the number of unit operations required, thereby lowering energy consumption and labor costs associated with intermediate handling. The use of a robust ruthenium catalyst paired with a phenanthroline ligand ensures high turnover numbers and excellent selectivity, making this process ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Ru-Catalyzed Hydrogen Transfer Cyclization
The core of this synthetic breakthrough lies in the elegant mechanistic pathway facilitated by the ruthenium catalyst, which orchestrates a series of bond-forming and bond-breaking events with high precision. The cycle initiates with the dehydrogenation of the alpha-amino alcohol by the ruthenium species to generate a reactive amino aldehyde intermediate in situ, releasing hydrogen that is temporarily stored on the metal center. This transient aldehyde then undergoes a condensation reaction with the alpha,beta-unsaturated alkynone to form an enamine or imine species, which subsequently cyclizes to establish the pyrrole ring structure. The final step involves the transfer of the stored hydrogen back to the intermediate to saturate specific bonds or stabilize the final aromatic system, regenerating the active catalyst for the next cycle. This internal redox neutral process ensures that no external oxidants or reductants are consumed in a stoichiometric fashion, leading to an atom-economical transformation that maximizes the incorporation of starting materials into the final product.
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the formation of over-oxidized byproducts or halogenated impurities often associated with oxidative cyclization methods. The mild basic conditions provided by potassium carbonate prevent the degradation of sensitive functional groups such as esters or nitriles that might be present on the aromatic rings of the substrates. Additionally, the high temperature stability of the ruthenium-phenanthroline complex allows the reaction to proceed at 150°C, providing the necessary activation energy to drive the cyclization to completion without decomposing the catalyst. This robustness translates to a cleaner crude reaction profile, which simplifies the purification process and ensures that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications for residual metals and organic impurities.
How to Synthesize 3-Acylpyrrole Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and precise temperature control to maximize the efficiency of the catalytic cycle. The process begins with the charging of the reactor with the specific molar ratios of substrates, catalyst, and ligand under an inert atmosphere to prevent catalyst deactivation by oxygen. Following an initial induction period at moderate temperatures to allow for catalyst activation and intermediate formation, the system is heated to reflux conditions to drive the cyclization and hydrogen transfer steps to completion. Detailed standard operating procedures regarding the specific workup and purification steps, including solvent removal and column chromatography parameters, are outlined in the technical guide below to ensure reproducible results across different batch sizes.
- Combine alpha-amino alcohol, alpha,beta-unsaturated alkynone, triruthenium dodecacarbonyl catalyst, 4-methyl-1,10-phenanthroline ligand, ethanol, base, and tert-amyl alcohol solvent in a reactor.
- Stir the mixture at 80°C under nitrogen protection for 1.5 hours to initiate the catalytic cycle and intermediate formation.
- Raise the temperature to 150°C and maintain reaction for 18 hours, followed by cooling, filtration, distillation, and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed methodology presents a compelling value proposition centered on cost optimization and supply reliability. By shifting away from stoichiometric oxidants and multi-step sequences, manufacturers can achieve a significant reduction in raw material expenditures and waste disposal fees, directly impacting the bottom line of production budgets. The reliance on commercially available and inexpensive starting materials such as alpha-amino alcohols and simple alkynones mitigates the risk of supply chain disruptions often caused by the scarcity of specialized reagents. Furthermore, the simplified one-pot nature of the reaction reduces the requirement for extensive equipment usage and labor hours, allowing facilities to increase throughput and respond more agilely to market demand fluctuations without compromising product quality.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants like IBX and the use of low-loading ruthenium catalysts result in substantial cost savings per kilogram of produced intermediate. The process avoids the need for costly purification steps to remove heavy metal residues or iodine byproducts, further streamlining the manufacturing economics. Additionally, the use of ethanol as a hydrogen source is far more economical than purchasing high-pressure hydrogen gas or specialized hydride reagents, contributing to a leaner cost structure for large-scale operations.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis are commodity chemicals with stable global supply chains, reducing the vulnerability to geopolitical or logistical bottlenecks. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without the need for highly specialized or custom-built reactor systems. This reliability ensures that downstream customers receive their orders on time, supporting continuous drug development pipelines and preventing costly delays in clinical trial material production.
- Scalability and Environmental Compliance: The one-pot design inherently minimizes solvent usage and waste generation, aligning with increasingly strict environmental regulations and sustainability goals. The process generates fewer hazardous byproducts, simplifying the permitting process for new manufacturing lines and reducing the long-term liability associated with chemical waste management. This environmental compatibility makes the technology future-proof, ensuring that production capacity can be expanded to meet growing market needs without encountering regulatory hurdles related to emissions or effluent treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing manufacturing portfolios and for assessing the potential return on investment for process development initiatives.
Q: What are the primary advantages of this ruthenium-catalyzed method over traditional oxidative cyclization?
A: This method utilizes a hydrogen transfer strategy which eliminates the need for stoichiometric oxidants like IBX, significantly reducing chemical waste and raw material costs while improving atom economy.
Q: What is the optimal catalyst loading for industrial application?
A: The patent specifies a highly efficient catalyst loading where the molar ratio of triruthenium dodecacarbonyl to the substrate is approximately 0.02:1, ensuring cost-effectiveness for large-scale production.
Q: Which solvents and bases are compatible with this synthesis route?
A: The process utilizes tert-amyl alcohol as the preferred solvent due to its high boiling point and stability, paired with potassium carbonate as a mild and inexpensive base to facilitate the cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylpyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN113636968B for the rapid advancement of pharmaceutical programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and risk-free. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of 3-acylpyrrole intermediate meets the highest quality standards required by global regulatory agencies. Our commitment to technical excellence allows us to navigate the complexities of ruthenium catalysis and deliver products with consistent performance profiles.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener synthesis route for your supply chain. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in catalytic transformations can accelerate your development timelines while optimizing your overall production costs.
