Advanced Ruthenium-Catalyzed Synthesis of 3-Acylpyrrole Intermediates for Global Pharmaceutical Manufacturing
Introduction to Next-Generation Pyrrole Synthesis
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, particularly the pyrrole scaffold, which serves as a critical backbone for numerous bioactive molecules including histone deacetylase inhibitors and COX inhibitors. Patent CN113636968B introduces a transformative synthetic approach for 3-acylpyrrole compounds that addresses longstanding challenges in efficiency and environmental impact. This innovation leverages a sophisticated ruthenium-catalyzed transfer hydrogenation strategy to couple alpha-amino alcohols with alpha,beta-unsaturated alkynones in a streamlined one-pot operation. By integrating the oxidation of the amino alcohol and the subsequent cyclization into a single seamless sequence, the technology drastically reduces the step count compared to classical multi-step syntheses. Furthermore, the method operates under relatively mild thermal gradients and utilizes earth-abundant solvents, positioning it as a superior alternative for the sustainable manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to access 3-acylpyrrole derivatives often rely on harsh oxidative conditions or cumbersome multi-step sequences that hinder industrial scalability. For instance, established protocols frequently employ stoichiometric amounts of expensive and toxic oxidants such as IBX (2-iodoxybenzoic acid) to mediate intramolecular oxidative cyclization, which generates significant quantities of iodine-containing waste that requires complex disposal procedures. Other historical methods involve the use of precious metal catalysts like palladium in conjunction with sensitive phosphine ligands that are prone to oxidation and degradation, leading to inconsistent batch-to-batch reproducibility. Additionally, many prior art processes necessitate the isolation of unstable intermediates, such as beta-amino enones, which increases the overall processing time and exposes the material to potential decomposition before the final ring closure can occur. These factors collectively contribute to elevated production costs and supply chain vulnerabilities for manufacturers relying on legacy technologies.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a catalytic borrowing hydrogen mechanism that elegantly bypasses the need for external oxidants while maintaining high atom economy. The process initiates with the dehydrogenation of the alpha-amino alcohol to generate a reactive amino aldehyde intermediate in situ, which immediately condenses with the alkynone substrate to form the requisite enamine species. This tandem sequence proceeds efficiently in a single reactor vessel using tert-amyl alcohol as a green solvent and potassium carbonate as a mild, inexpensive base. The use of a triruthenium dodecacarbonyl catalyst system ensures high turnover numbers and excellent functional group tolerance, allowing for the incorporation of diverse substituents on the aromatic rings without compromising yield. Consequently, this approach not only simplifies the operational workflow but also significantly enhances the safety profile of the manufacturing process by eliminating hazardous reagents.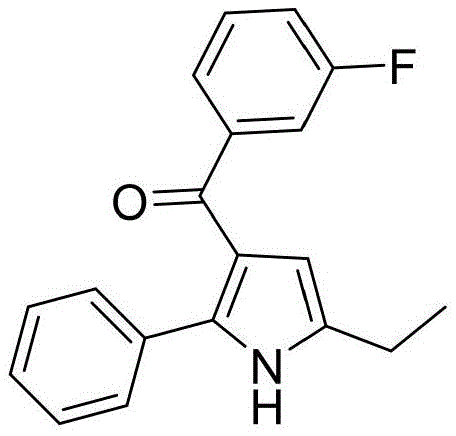
Mechanistic Insights into Ru-Catalyzed Cyclization
The core of this technological breakthrough lies in the intricate catalytic cycle mediated by the triruthenium dodecacarbonyl complex in the presence of the 4-methyl-1,10-phenanthroline ligand. The mechanism begins with the activation of the alpha-amino alcohol by the ruthenium center, which abstracts hydrogen to form a transient ruthenium-hydride species and the corresponding amino aldehyde. This aldehyde then undergoes a condensation reaction with the alpha,beta-unsaturated alkynone to generate an enamine intermediate, setting the stage for the crucial cyclization event. The ruthenium-hydride species subsequently delivers the borrowed hydrogen back to the system, facilitating the aromatization of the pyrrole ring and regenerating the active catalyst for the next cycle. This redox-neutral pathway is thermodynamically favorable and avoids the accumulation of high-energy intermediates that often lead to polymerization or decomposition in oxidative methods.
From an impurity control perspective, the choice of ligand and base plays a pivotal role in suppressing side reactions such as over-reduction of the carbonyl group or oligomerization of the alkynone. The 4-methyl-1,10-phenanthroline ligand provides a specific steric and electronic environment around the ruthenium center that favors the desired 5-exo-dig cyclization mode over competing pathways. Furthermore, the use of potassium carbonate as a base ensures that the reaction medium remains sufficiently basic to promote enamine formation without being so strong as to trigger hydrolysis of the sensitive acyl group or the ester functionalities that might be present in complex substrates. This precise tuning of reaction parameters results in a clean crude product profile, which significantly reduces the burden on downstream purification units and improves the overall yield of the isolated API intermediate.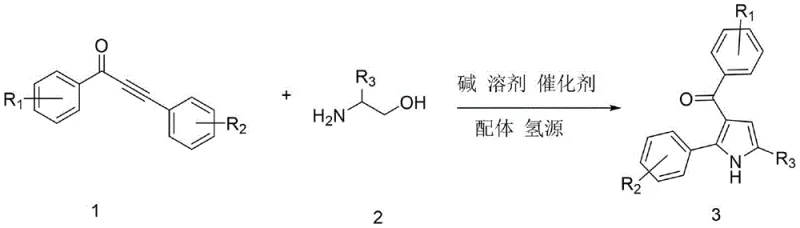
How to Synthesize 3-Acylpyrrole Efficiently
Implementing this synthesis requires strict adherence to the optimized thermal profile and reagent ratios defined in the patent to ensure maximum conversion and selectivity. The process is designed to be operationally simple, requiring standard stainless steel or glass-lined reactors capable of handling temperatures up to 150°C under inert atmosphere. Operators must carefully monitor the two-stage heating protocol, as the initial lower temperature phase is critical for the formation of the imine/enamine intermediates before the higher temperature drives the final cyclization and aromatization. Detailed standard operating procedures regarding the addition order of the catalyst and ligand are essential to prevent premature catalyst deactivation. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and workup procedures, please refer to the standardized synthesis guide below.
- Combine alpha-amino alcohol, alpha,beta-unsaturated alkynone, triruthenium dodecacarbonyl catalyst, ligand, ethanol, base, and solvent in a reactor under nitrogen.
- Stir the mixture at 80°C for 1.5 hours to initiate the catalytic cycle and intermediate formation.
- Increase temperature to 150°C and maintain for 18 hours to complete the cyclization, followed by cooling, filtration, and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ruthenium-catalyzed route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift away from stoichiometric oxidants and precious palladium catalysts towards a more robust ruthenium system directly translates to a reduction in raw material volatility and cost exposure. By consolidating multiple synthetic steps into a single pot, the process inherently lowers the consumption of solvents and energy, while simultaneously reducing the labor hours required for intermediate isolations and transfers. This streamlining of the manufacturing workflow enhances the overall throughput of the facility, allowing for faster response times to market demand fluctuations without the need for significant capital investment in new equipment. Ultimately, this technology provides a more resilient and cost-efficient supply chain for critical pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants like IBX and the replacement of palladium catalysts with a more economical ruthenium system fundamentally alters the cost structure of production. Since the catalyst loading is low and the ligand is stable, the recurring cost of goods sold is significantly decreased compared to traditional oxidative methods. Furthermore, the simplified workup procedure, which involves basic filtration and distillation rather than complex chromatographic separations at early stages, reduces the consumption of silica gel and eluents. These cumulative savings allow for a more competitive pricing strategy for the final 3-acylpyrrole intermediates, providing a distinct margin advantage in contract manufacturing negotiations.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically alpha-amino alcohols and alpha,beta-unsaturated alkynones, are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. Unlike specialized reagents required for older methods which may have long lead times or restricted availability, these commoditized feedstocks ensure a steady and predictable flow of materials into the production line. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, further stabilizing the supply chain against upstream disruptions. This reliability is crucial for maintaining continuous production schedules for long-term pharmaceutical contracts.
- Scalability and Environmental Compliance: The process is inherently scalable due to its one-pot nature and the use of high-boiling, stable solvents like tert-amyl alcohol which are well-suited for large-volume reactors. From an environmental standpoint, the atom-economical nature of the transfer hydrogenation reaction minimizes the generation of chemical waste, aligning with increasingly stringent global regulations on industrial emissions and effluent discharge. The absence of heavy metal waste streams associated with stoichiometric oxidants simplifies the wastewater treatment process and reduces the environmental compliance burden. This green chemistry profile not only future-proofs the manufacturing site against regulatory changes but also enhances the corporate sustainability metrics valued by modern pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and expected outcomes. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the value proposition of this new manufacturing route.
Q: What are the primary advantages of this ruthenium-catalyzed method over traditional oxidative cyclization?
A: This method utilizes a transfer hydrogenation strategy that eliminates the need for stoichiometric toxic oxidants like IBX, resulting in a greener process with simpler waste management and reduced raw material costs.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the protocol employs readily available starting materials and robust reaction conditions (80°C to 150°C) in standard solvents like tert-amyl alcohol, making it highly amenable to scale-up in commercial reactors.
Q: How does the catalyst system ensure high selectivity for the 3-acylpyrrole scaffold?
A: The combination of triruthenium dodecacarbonyl with the 4-methyl-1,10-phenanthroline ligand facilitates a precise borrowing hydrogen mechanism that directs regioselective cyclization, minimizing side reactions and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acylpyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the ruthenium-catalyzed route described in CN113636968B and is fully prepared to implement this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-acylpyrrole intermediate delivered meets the highest quality standards required for drug substance manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient route. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the profitability and sustainability of your supply chain.
