Scalable Synthesis of Potent Pyrimidine Antitumor Intermediates for Global Pharmaceutical Supply Chains
Scalable Synthesis of Potent Pyrimidine Antitumor Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry is constantly seeking novel scaffolds that offer superior efficacy against resistant tumor types, and the technology disclosed in patent CN102115470A represents a significant advancement in this domain. This patent details a class of 2,4,5-substituted pyrimidine small molecule compounds that exhibit remarkable antitumor activity by targeting microtubule polymerization. Unlike traditional cytotoxic agents, these novel derivatives demonstrate potent inhibition of cell proliferation with IC50 values reaching the nanomolar level across diverse cancer cell lines, including drug-resistant strains. For R&D directors and procurement specialists, understanding the synthetic accessibility and structural versatility of these molecules is crucial for integrating them into modern oncology pipelines. The core structure, defined by General Formula A, combines an indole or azaindole moiety with a pyrimidine ring, creating a unique pharmacophore that warrants close attention for future drug development programs.
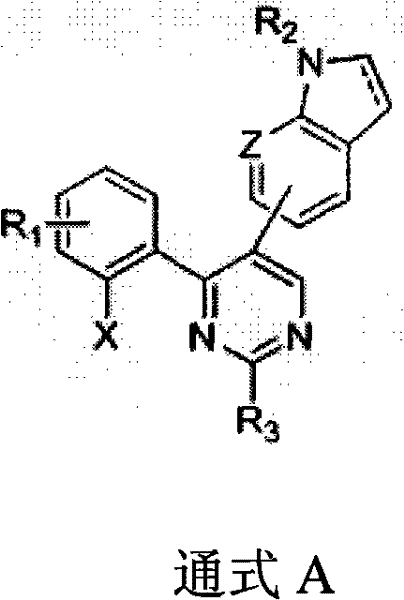
The strategic value of this technology lies not only in its biological potency but also in the robustness of its manufacturing process. By leveraging well-established cross-coupling reactions, the synthesis avoids the extreme conditions often associated with heterocyclic chemistry, thereby reducing operational risks and energy consumption. This makes the technology particularly attractive for reliable pharmaceutical intermediate supplier networks aiming to secure long-term supply contracts. The ability to modulate substituents at multiple positions allows for extensive structure-activity relationship (SAR) studies, enabling medicinal chemists to optimize pharmacokinetic properties without compromising the core synthetic strategy. As we delve deeper into the technical specifics, it becomes evident that this platform offers a balanced solution for both innovation and manufacturability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex pyrimidine-fused heterocycles has been plagued by inefficient multi-step sequences that rely on harsh reagents and difficult purification protocols. Conventional routes often involve the use of homogeneous palladium catalysts that are expensive and challenging to remove from the final API, leading to potential heavy metal contamination issues that require additional downstream processing. Furthermore, traditional methods frequently suffer from low regioselectivity, resulting in complex mixtures of isomers that drastically reduce overall yield and increase waste generation. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for supply chain heads who need to ensure consistent material availability for clinical trials. The reliance on sensitive reagents also limits the scalability of these processes, making it difficult to transition from gram-scale laboratory synthesis to kilogram or ton-scale commercial production without significant process re-engineering.
The Novel Approach
In contrast, the methodology presented in this patent utilizes a streamlined two-step sequence that significantly enhances process efficiency and environmental compatibility. The core strategy employs a Suzuki-Miyaura coupling between an iodochromone derivative and various indole boronic acids, catalyzed by heterogeneous 10% Pd/C. This choice of catalyst is pivotal, as it allows for easy removal via simple filtration, eliminating the need for complex scavenging resins or extensive washing procedures. Following the coupling, the intermediate ketone undergoes a direct condensation with amidines or guanidines under mild basic conditions to form the target pyrimidine ring. This approach not only simplifies the workflow but also demonstrates excellent functional group tolerance, allowing for the introduction of diverse substituents without protecting group manipulations. The versatility of this route is further exemplified by the ability to perform subsequent modifications, such as Buchwald-Hartwig amination, to generate advanced derivatives.
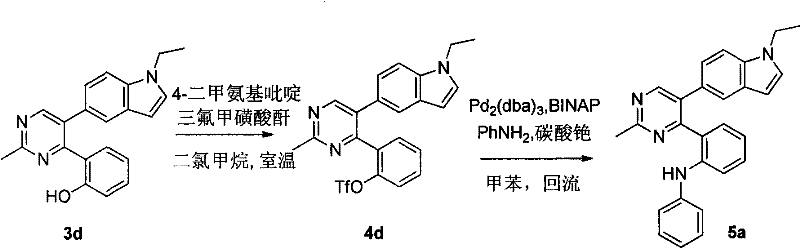
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling and Cyclization
The success of this synthetic route hinges on the precise orchestration of the palladium-catalyzed cross-coupling mechanism. In the first step, the oxidative addition of the aryl iodide (iodochromone) to the palladium surface initiates the catalytic cycle. The presence of a base, typically potassium carbonate, facilitates the transmetallation step by activating the boronic acid species, allowing for the transfer of the indole moiety to the palladium center. Subsequent reductive elimination releases the coupled product and regenerates the active catalyst. The use of a biphasic solvent system comprising acetonitrile and water is critical here, as it solubilizes both the organic substrates and the inorganic base while maintaining the stability of the catalyst. This mechanistic pathway ensures high conversion rates even at moderate temperatures of 50°C, minimizing thermal degradation of sensitive intermediates. The robustness of this coupling is evidenced by the successful synthesis of various analogues with different substitution patterns on the indole ring.
Following the coupling, the formation of the pyrimidine ring proceeds through a nucleophilic attack of the amidine or guanidine on the carbonyl group of the chromone intermediate. Under basic conditions at 70-80°C, this condensation is followed by cyclization and aromatization to yield the final pyrimidine scaffold. From an impurity control perspective, this step is remarkably clean, primarily generating water as a byproduct. However, careful monitoring of reaction time is essential to prevent over-reaction or decomposition of the product. The final purification via silica gel column chromatography using petroleum ether and ethyl acetate effectively removes any unreacted starting materials or minor side products, ensuring high chemical purity. This level of control is vital for meeting the stringent quality standards required for pharmaceutical intermediates intended for preclinical and clinical evaluation.
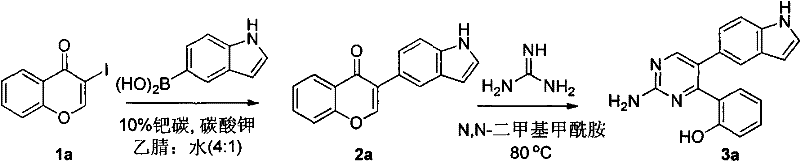
How to Synthesize Pyrimidine Antitumor Intermediates Efficiently
The practical implementation of this synthesis requires adherence to specific operational parameters to maximize yield and safety. The process begins with the preparation of the reaction mixture under inert atmosphere to prevent catalyst deactivation. Precise stoichiometry is maintained to ensure complete consumption of the expensive iodochromone starting material. The reaction progress is monitored using standard analytical techniques to determine the optimal endpoint before proceeding to the workup phase. The subsequent condensation step demands careful temperature control to facilitate ring closure without inducing side reactions. For a detailed breakdown of the standardized operating procedures, including exact reagent quantities and safety precautions, please refer to the technical guide below.
- Perform Suzuki coupling of iodochromone with indole boronic acid using 10% Pd/C catalyst in acetonitrile/water at 50°C.
- Isolate the intermediate ketone and react with guanidine or amidine in DMF with potassium carbonate at 70-80°C.
- Purify the final pyrimidine product via silica gel column chromatography using petroleum ether and ethyl acetate eluents.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing route offers substantial benefits that align with the strategic goals of cost reduction in API manufacturing and supply chain resilience. The use of commercially available starting materials, such as iodochromone and substituted indole boronic acids, ensures a stable supply base that is not subject to the volatility of exotic reagent markets. The heterogeneous nature of the palladium catalyst not only simplifies the purification process but also allows for potential catalyst recovery and recycling, contributing to significant cost savings over large production batches. Furthermore, the avoidance of cryogenic conditions or high-pressure equipment reduces the capital expenditure required for facility setup, making this technology accessible for contract manufacturing organizations looking to expand their oncology portfolio. These factors collectively enhance the economic viability of producing these high-value intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive homogeneous catalysts and complex purification steps drastically lowers the cost of goods sold. By utilizing 10% Pd/C, which is filterable and reusable, the process minimizes metal waste and reduces the burden on wastewater treatment systems. The mild reaction conditions also translate to lower energy consumption, as there is no need for prolonged heating or cooling cycles. Additionally, the high atom economy of the Suzuki coupling and condensation reactions ensures that a maximum proportion of raw materials ends up in the final product, reducing raw material costs per kilogram of output. This efficiency is critical for maintaining competitive pricing in the global pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on robust, well-understood chemical transformations mitigates the risk of batch failures that can disrupt supply timelines. The scalability of the Suzuki coupling reaction is well-documented in industrial settings, allowing for seamless transition from pilot plant to full-scale production. The use of common solvents like acetonitrile, DMF, and ethyl acetate further simplifies logistics, as these are readily available from multiple suppliers worldwide. This redundancy in the supply chain ensures that production schedules can be maintained even if a specific vendor faces shortages. Moreover, the stability of the intermediates allows for flexible inventory management, enabling manufacturers to stock key precursors without significant degradation concerns.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing hazardous waste generation. The aqueous workup and filtration steps reduce the volume of organic solvents required for purification, lowering the environmental footprint of the manufacturing process. The absence of toxic heavy metal residues in the final product simplifies regulatory compliance, as strict limits on residual palladium are easier to meet with heterogeneous catalysis. This environmental stewardship is increasingly important for pharmaceutical companies aiming to meet sustainability targets. The straightforward scale-up path ensures that increasing production volumes to meet market demand does not compromise safety or quality, providing a reliable foundation for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its potential for integration into their operations. The following questions address common inquiries regarding the reaction mechanism, scalability, and quality control measures associated with this technology. These insights are derived directly from the experimental data and procedural descriptions found in the patent documentation, providing a factual basis for decision-making. By clarifying these aspects, we aim to facilitate a smoother transition from laboratory curiosity to commercial reality for these promising antitumor agents.
Q: What is the primary mechanism of action for these pyrimidine compounds?
A: These compounds function as microtubule polymerization inhibitors, demonstrating potent antitumor activity comparable to vinorelbine in various cell lines.
Q: Are the synthesis conditions suitable for large-scale production?
A: Yes, the process utilizes heterogeneous Pd/C catalysts which are easily filtered, and moderate temperatures (50-80°C), facilitating safe commercial scale-up.
Q: How is product purity ensured in this manufacturing route?
A: Purity is maintained through rigorous workup procedures including filtration of catalysts, aqueous extractions, and final purification via column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of these pyrimidine-based antitumor intermediates and are committed to supporting their development from bench to bedside. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest industry standards. Our expertise in palladium-catalyzed couplings and heterocyclic chemistry allows us to optimize these routes for maximum efficiency, delivering high-quality materials that accelerate your drug discovery timelines.
We invite you to collaborate with us to explore the full capabilities of this technology for your specific oncology programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, highlighting opportunities for process optimization and budget efficiency. Please contact us to request specific COA data and route feasibility assessments for the compounds discussed in patent CN102115470A. Together, we can drive the next generation of cancer therapies forward with reliable, scalable, and cost-effective chemical solutions.
