Advanced Chromone-Based Synthesis of Bioactive Pyrimidine Derivatives for Commercial Scale-Up
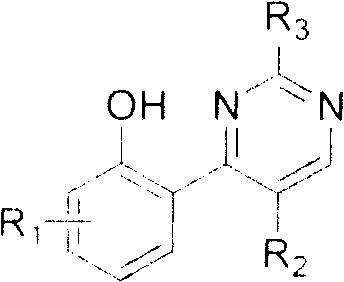
The pharmaceutical landscape is constantly evolving, demanding more efficient pathways to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN101033224B introduces a groundbreaking methodology for synthesizing a novel class of 2,4,5-substituted pyrimidine small molecule compounds, which have demonstrated significant potential as antitumor and antiviral agents. This technology leverages commercially available chromone derivatives as key starting materials, transforming them through a series of elegant multi-component reactions. By utilizing halogenated chromones, such as 3-iodochromone, the process enables precise functionalization at the C-3 position, facilitating the construction of diverse pyrimidine libraries. For R&D teams seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this chromone-based approach is critical for accelerating drug discovery pipelines.
The strategic value of this patent lies in its ability to generate high-value bioactive molecules with structural diversity. The described compounds exhibit potent activity against human liver cancer cells and Influenza A virus, making them prime candidates for further development. The synthesis avoids harsh conditions often associated with traditional pyrimidine formation, instead relying on manageable reaction parameters that are conducive to scale-up. This report delves into the technical specifics, comparing conventional limitations with this novel approach, and outlines the commercial advantages for procurement and supply chain stakeholders looking for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of substituted pyrimidines often involves multi-step sequences that suffer from low atom economy and poor regioselectivity. Classical methods typically require the condensation of 1,3-dicarbonyl compounds with amidines, which can lead to complex mixtures of isomers that are difficult to separate. Furthermore, introducing specific substituents at the 2, 4, and 5 positions simultaneously often necessitates protecting group strategies that add unnecessary steps and waste. The reliance on expensive catalysts or extreme temperatures in older protocols frequently results in decomposition of sensitive functional groups, limiting the scope of accessible chemical space. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In contrast, the methodology disclosed in CN101033224B offers a convergent strategy that significantly streamlines the synthetic route. By starting with 3-iodochromone, the process exploits the inherent reactivity of the halogenated position to introduce diverse aryl, heteroaryl, or thio-heterocyclic groups directly. This is followed by a condensation step with amidines to close the pyrimidine ring. This approach minimizes the number of isolation steps and reduces solvent consumption. The versatility of the chromone scaffold allows for the rapid generation of analogs by simply swapping the boronic acid or mercapto-heterocycle component. This modularity is essential for commercial scale-up of complex pharmaceutical intermediates, as it allows manufacturers to adapt quickly to changing project requirements without retooling entire production lines.
Mechanistic Insights into Chromone-Based Pyrimidine Construction
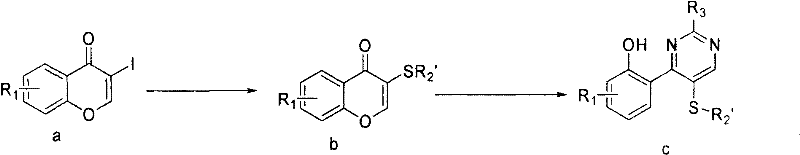
The core of this technology relies on a sophisticated interplay between nucleophilic substitution and cyclocondensation mechanisms. In the first pathway, the iodine atom at the 3-position of the chromone ring acts as a leaving group or a coupling handle. When reacted with mercapto-heterocycles like 2-mercapto-1-methylimidazole in the presence of a base such as potassium carbonate, a nucleophilic aromatic substitution or addition-elimination sequence occurs. This forms a stable thio-ether linkage, creating a crucial intermediate that positions the nitrogen atoms correctly for the subsequent ring closure. The use of polar aprotic solvents like DMF facilitates this transformation by stabilizing the charged intermediates, ensuring high conversion rates even at room temperature.
Following the initial substitution, the introduction of the amidine component triggers the formation of the pyrimidine core. The amidine nitrogen attacks the carbonyl carbon of the chromone system, initiating a cascade that results in the opening of the pyrone ring and subsequent recyclization into the pyrimidine structure. This mechanism is highly sensitive to steric and electronic effects, which is why the patent details specific conditions for different substituents. For instance, electron-withdrawing groups on the chromone may accelerate the nucleophilic attack, while bulky groups might require extended reaction times. Understanding these mechanistic subtleties allows process chemists to optimize reaction parameters, ensuring consistent quality and reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize 2,4,5-Substituted Pyrimidines Efficiently
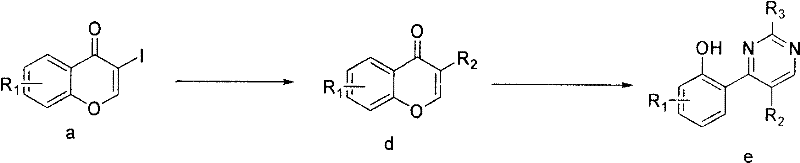
The practical execution of this synthesis involves dissolving the 3-iodochromone precursor in a suitable solvent system, typically DMF or acetonitrile, and adding the requisite coupling partner. For the thio-substitution route, mercapto-heterocycles and potassium carbonate are added, and the mixture is stirred at room temperature for several hours to ensure complete consumption of the starting material. Subsequently, the amidine is introduced, and the reaction is allowed to proceed overnight. Workup involves solvent evaporation followed by purification via silica gel column chromatography, using gradients of dichloromethane and methanol. Detailed standardized synthesis steps see the guide below.
- React 3-iodochromone with potassium carbonate and mercapto-heterocycles in DMF at room temperature to form the thio-substituted intermediate.
- Condense the intermediate with various amidine compounds under stirring overnight to construct the pyrimidine ring system.
- Purify the final crude solid via silica gel column chromatography using dichloromethane and methanol eluents to obtain high-purity targets.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this chromone-based synthesis offers tangible benefits that extend beyond mere chemical novelty. The reliance on commercially available starting materials, such as substituted chromones and common boronic acids, mitigates the risk of raw material shortages. Unlike proprietary catalysts that may be subject to export controls or limited supply, the reagents used here—potassium carbonate, palladium catalysts, and standard organic solvents—are commodity chemicals available from multiple global vendors. This diversification of the supply base enhances resilience against market fluctuations and ensures continuity of supply for critical drug development programs.
- Cost Reduction in Manufacturing: The streamlined nature of the reaction sequence directly impacts the cost of goods sold. By reducing the number of synthetic steps and avoiding expensive protecting group manipulations, the overall material throughput is improved. The ability to perform key transformations at room temperature or mild heating (50-70°C) significantly lowers energy consumption compared to high-temperature reflux processes. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, reducing the burden on downstream purification and lowering solvent waste disposal costs.
- Enhanced Supply Chain Reliability: The robustness of the chemistry contributes to more predictable manufacturing timelines. The tolerance of the reaction to various functional groups means that a single set of conditions can often be applied to a wide range of analogs, simplifying process validation. This flexibility allows suppliers to respond rapidly to requests for new derivatives without extensive re-optimization. For procurement managers, this translates to shorter quotation cycles and more reliable delivery schedules for custom synthesis projects, ensuring that clinical trial material is available when needed.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram pilot production. The use of standard workup procedures like extraction and crystallization, rather than specialized techniques like preparative HPLC, facilitates this transition. Additionally, the avoidance of heavy metal contaminants in the final product (through efficient filtration of Pd/C catalysts) aligns with stringent regulatory guidelines for pharmaceutical ingredients. This environmental compatibility reduces the complexity of waste treatment and supports sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for technical decision-makers.
Q: What are the primary starting materials for this pyrimidine synthesis?
A: The process utilizes commercially available chromone compounds, specifically halogenated derivatives like 3-iodochromone, which serve as versatile scaffolds for subsequent coupling reactions.
Q: How does this method improve impurity control compared to traditional routes?
A: By employing specific multi-component reactions with defined stoichiometry and mild conditions, side reactions are minimized, allowing for easier purification via standard column chromatography.
Q: Is this synthesis suitable for large-scale pharmaceutical manufacturing?
A: Yes, the use of common solvents like DMF and acetonitrile, along with ambient to moderate temperature requirements, supports scalability from gram to multi-kilogram production batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidine Derivatives Supplier
As the demand for advanced antiviral and antitumor agents grows, having a manufacturing partner with deep expertise in heterocyclic chemistry is paramount. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of pyrimidine intermediates meets the highest international standards, ready for immediate integration into your drug discovery workflows.
We invite you to leverage our technical capabilities to accelerate your development timeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific compound requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized chromone-based synthesis can enhance your supply chain efficiency and reduce overall project costs.
