Scalable Aryltriazolinone Manufacturing: A Cost-Effective Cyanate-Based Route for Pharmaceutical Intermediates
The pharmaceutical and agrochemical industries constantly seek robust synthetic pathways that balance high purity with economic viability, particularly for heterocyclic scaffolds like aryltriazolinones. Patent CN100418956C introduces a transformative methodology for the preparation of aryl-1,2,4-triazolin-5-ones, addressing critical bottlenecks in existing manufacturing technologies. This innovation shifts the paradigm from reliance on costly phosphoryl azides and corrosive halogens to a streamlined protocol utilizing inexpensive alkali metal cyanates and formaldehyde. For R&D directors and procurement strategists, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. The disclosed process not only enhances safety profiles by mitigating the use of hazardous oxidants but also facilitates a telescoped one-pot reaction sequence that drastically simplifies downstream processing. By leveraging this technology, manufacturers can achieve substantial cost reduction in pharmaceutical intermediate manufacturing while maintaining stringent quality standards required for active pharmaceutical ingredient (API) synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
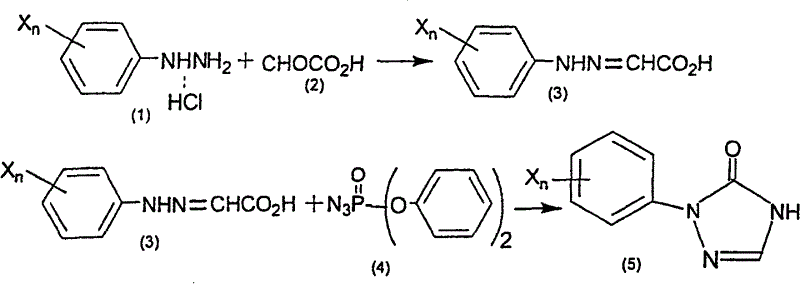
The Limitations of Conventional Methods
Historically, the synthesis of aryltriazolinones has been plagued by economic and safety inefficiencies inherent in traditional methodologies. As illustrated in prior art such as PCT International Publication WO 98/38176, conventional routes often necessitate the use of diphenylphosphoryl azide (DPPA), an exceptionally expensive reagent that severely inflates the cost of goods sold. Furthermore, these legacy processes typically require the isolation and purification of unstable arylhydrazone intermediates, adding multiple unit operations that increase solvent consumption and extend production lead times. Alternative methods described in documents like WO 91/3470 rely heavily on hypohalous acids or elemental halogens for the critical dehydrogenation step. These reagents are highly corrosive and generate significant volumes of halogen-containing wastewater, creating substantial environmental compliance burdens and disposal costs. The instability of intermediates under these harsh oxidative conditions often leads to variable yields and complex impurity profiles, making commercial scale-up of complex pharmaceutical intermediates challenging and risky for supply chain continuity.
The Novel Approach
The novel process detailed in CN100418956C fundamentally reengineers the synthetic pathway to overcome these historical limitations through the strategic use of cyanate chemistry. Instead of expensive azides, the method employs readily available alkali metal cyanates, such as sodium cyanate, coupled with simple organic acids to drive the cyclization. A key advantage is the ability to perform the reaction in a telescoped manner, where the arylhydrazone and subsequent aryltriazolidinone intermediates are generated in situ without isolation. This eliminates the need for intermediate workups, thereby reducing solvent usage and labor costs. Moreover, the invention offers a second embodiment utilizing molecular oxygen as the terminal oxidant in the presence of a catalyst, completely avoiding the use of corrosive halogens. This green chemistry approach not only lowers the environmental footprint but also enhances the safety profile of the manufacturing facility, making it an ideal solution for reliable aryltriazolinone supplier networks aiming for sustainable production practices.
Mechanistic Insights into Cyanate-Mediated Cyclization and Oxidation
The core of this technological advancement lies in a sophisticated three-stage mechanistic sequence that ensures high conversion and selectivity. The first stage involves the condensation of an arylhydrazine with formaldehyde to form an arylhydrazone intermediate. This step is kinetically favorable and proceeds rapidly under mild acidic or basic catalysis in solvents like tert-butanol. In the second stage, the introduction of an alkali metal cyanate and a proton source triggers a cyclization reaction. The cyanate ion acts as a carbon and nitrogen source, attacking the hydrazone to close the five-membered triazolidinone ring. This cyclization is highly efficient and avoids the formation of side products common in azide-based routes. The final stage is the oxidative dehydrogenation of the saturated triazolidinone ring to the aromatic triazolinone system. This can be achieved using mild oxidants like sodium hypochlorite or, more elegantly, via aerobic oxidation using oxygen gas and a transition metal catalyst. This mechanistic flexibility allows manufacturers to tailor the oxidation step based on available infrastructure and safety protocols.
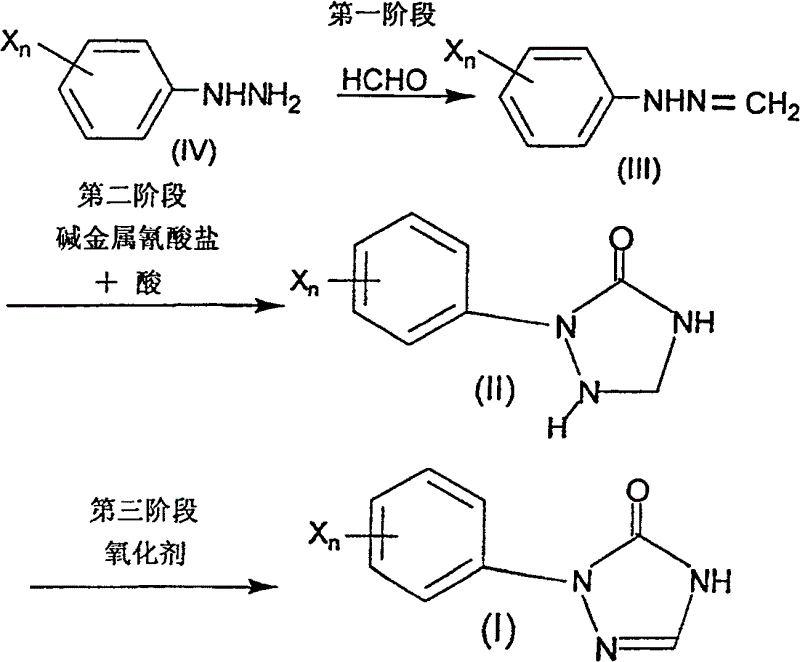
Impurity control is intrinsically built into this mechanism through the stability of the intermediates and the specificity of the reagents. Unlike halogen-based oxidations which can lead to ring chlorination or over-oxidation, the cyanate-mediated cyclization is chemoselective for the hydrazine moiety. The use of oxygen in the second method further minimizes inorganic salt waste, resulting in a cleaner crude product that requires less rigorous purification. The reaction conditions, typically maintained between 0°C and 60°C, prevent thermal degradation of the sensitive triazoline ring. By avoiding the harsh conditions associated with DPPA or elemental bromine, the process preserves the integrity of sensitive substituents on the aryl ring, such as halogens or alkyl groups. This results in a superior impurity profile, which is critical for meeting the stringent purity specifications demanded by regulatory bodies for drug substances. The ability to recycle solvents like tert-butanol further enhances the economic and environmental efficiency of the overall process.
How to Synthesize Aryltriazolinones Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the preparation of the arylhydrazone by reacting the corresponding arylhydrazine salt with formaldehyde in a mixed solvent system. Following this, sodium cyanate and an organic acid are added directly to the reaction mixture to induce cyclization without isolating the hydrazone. The final transformation involves the addition of an oxidant or the introduction of oxygen gas to aromatize the ring. This telescoped approach minimizes handling of potentially unstable intermediates and streamlines the workflow for operators. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up.
- React arylhydrazine with formaldehyde in a solvent like tert-butanol to form the arylhydrazone intermediate.
- Add alkali metal cyanate and an organic acid to the reaction mixture to cyclize the hydrazone into aryltriazolidinone.
- Introduce an oxidizing agent such as sodium hypochlorite or oxygen to dehydrogenate the triazolidinone into the final aryltriazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cyanate-based process offers compelling strategic advantages that extend beyond simple raw material substitution. The elimination of diphenylphosphoryl azide removes a major cost driver and supply bottleneck, as DPPA is often subject to price volatility and limited availability from specialized vendors. By switching to commodity chemicals like formaldehyde and sodium cyanate, manufacturers can secure a more stable and predictable supply chain, reducing the risk of production stoppages. The telescoped nature of the reaction reduces the number of processing steps, which directly translates to lower utility consumption, reduced labor hours, and decreased equipment occupancy time. These operational efficiencies contribute to substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The primary economic benefit stems from the replacement of high-cost reagents with inexpensive bulk chemicals. Diphenylphosphoryl azide is a premium reagent, whereas sodium cyanate and formaldehyde are produced on a massive industrial scale at a fraction of the cost. Additionally, the ability to perform the reaction in a single vessel without intermediate isolation significantly reduces solvent purchase and disposal costs. The process also minimizes the generation of hazardous waste, particularly when using the oxygen oxidation method, which lowers the expenses associated with environmental compliance and waste treatment facilities. These factors combine to create a leaner, more cost-effective production model that improves profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks enhances the resilience of the supply chain against market fluctuations. Formaldehyde and alkali metal cyanates are widely available from multiple global suppliers, reducing dependency on single-source vendors for critical reagents. The simplified process flow also means shorter cycle times, enabling faster turnaround from order to delivery. This agility is crucial for responding to sudden increases in demand from downstream API manufacturers. Furthermore, the reduced hazard profile of the reagents simplifies logistics and storage requirements, lowering insurance costs and facilitating easier transportation of materials across international borders, ensuring consistent supply continuity for global partners.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scalability, with the one-pot methodology easily transferable from laboratory glassware to large-scale stainless steel reactors. The avoidance of corrosive halogens and the potential to use oxygen as a green oxidant aligns perfectly with modern environmental, health, and safety (EHS) standards. This reduces the burden on wastewater treatment plants and minimizes the risk of accidental exposure to toxic substances. The ability to recycle solvents like tert-butanol further demonstrates a commitment to sustainable manufacturing practices. These attributes make the technology highly attractive for companies aiming to expand their production capacity while adhering to increasingly strict global regulations on chemical manufacturing and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aryltriazolinone synthesis technology. These insights are derived directly from the patent specifications and experimental data, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios. The answers cover aspects ranging from raw material compatibility to purification strategies, ensuring a comprehensive overview for decision-makers.
Q: How does this process reduce manufacturing costs compared to traditional methods?
A: This method replaces expensive reagents like diphenylphosphoryl azide (DPPA) with inexpensive commodity chemicals such as formaldehyde and sodium cyanate, significantly lowering raw material costs.
Q: Is the process safe for large-scale industrial production?
A: Yes, the second method utilizes oxygen as the oxidant instead of corrosive halogens or hypohalous acids, reducing hazardous waste and improving operational safety for commercial scale-up.
Q: Can the intermediates be isolated during the synthesis?
A: No, the process is designed as a telescoped one-pot reaction where intermediates like arylhydrazone and aryltriazolidinone are not isolated, streamlining the workflow and improving overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryltriazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the potential of the cyanate-mediated process described in CN100418956C and is fully prepared to implement this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and robust. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this synthesis, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. We are committed to delivering high-purity aryltriazolinones that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through this innovative technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with us, you gain access to our deep expertise in process development and our commitment to reliability. Please contact us today to request specific COA data and route feasibility assessments for your target aryltriazolinone derivatives. Let us help you engineer a more efficient and profitable future for your chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
