Advanced Manufacturing of Sitagliptin Intermediates: A Technical Analysis of Scalable Grignard Chemistry
The global pharmaceutical landscape continues to demand more efficient and cost-effective pathways for the production of high-volume anti-diabetic agents, specifically focusing on the critical building blocks required for Dipeptidyl Peptidase-4 (DPP-4) inhibitors. Patent CN102199102B introduces a transformative synthetic methodology for generating key Sitagliptin intermediates, addressing long-standing bottlenecks associated with traditional chiral synthesis routes. This technical disclosure outlines a novel approach utilizing a Grignard coupling strategy that bypasses the need for complex enzymatic resolutions or expensive transition metal hydrogenation steps often seen in legacy processes. By leveraging a mixed anhydride activation technique followed by nucleophilic addition, the described method achieves superior control over stereochemistry and functional group tolerance. For procurement leaders and technical directors evaluating supply chain resilience, this patent represents a pivotal shift towards more robust and economically viable manufacturing protocols. The ability to synthesize the core trifluorophenyl substituted scaffold using readily available organometallic reagents significantly de-risks the production timeline. Furthermore, the operational simplicity of the reaction conditions suggests a lower barrier to entry for commercial scale-up, making it an attractive option for generic manufacturers seeking to optimize their cost of goods sold without compromising on quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Sitagliptin and its precursors has been plagued by reliance on sophisticated and costly catalytic systems that introduce significant supply chain vulnerabilities. Traditional routes often employ asymmetric hydrogenation using precious metal complexes such as Ruthenium-BINAP catalysts, which not only carry a high price tag but also necessitate rigorous downstream purification to meet strict residual metal specifications mandated by regulatory bodies. Additionally, alternative pathways involving diazo transfer reactions pose substantial safety hazards due to the explosive nature of diazo intermediates, requiring specialized equipment and stringent safety protocols that drive up capital expenditure. These conventional methods frequently suffer from lengthy synthetic sequences involving multiple protection and deprotection steps, which cumulatively reduce the overall mass balance and increase waste generation. The cumulative effect of these inefficiencies is a manufacturing process that is highly sensitive to raw material price fluctuations and equipment availability. Moreover, the use of cryogenic conditions in some organolithium-based approaches increases energy consumption and limits the flexibility of production scheduling. For a reliable sitagliptin intermediate supplier, overcoming these inherent limitations is essential to maintaining competitive pricing and consistent delivery performance in a volatile market.
The Novel Approach
In stark contrast to the complexities of prior art, the methodology disclosed in CN102199102B utilizes a streamlined Grignard addition to a pre-activated mixed anhydride, offering a direct and scalable solution to the synthesis of the key keto-ester intermediate. This approach capitalizes on the high nucleophilicity of the 2,4,5-trifluorophenylmagnesium bromide reagent to efficiently construct the carbon-carbon bond required for the API backbone. The reaction proceeds under mild thermal conditions, typically between 0°C and 25°C, which eliminates the need for energy-intensive cryogenic cooling and allows for safer operation in standard glass-lined or stainless steel reactors. By avoiding the use of hazardous diazo compounds and expensive chiral catalysts, this novel route significantly simplifies the safety profile and reduces the environmental footprint of the manufacturing process. The strategic use of benzyl and tert-butoxycarbonyl protecting groups ensures high chemoselectivity, preventing unwanted side reactions at the amine functionality during the carbonyl addition step. This results in a cleaner reaction profile with fewer by-products, thereby reducing the burden on downstream purification units such as chromatography or crystallization. Ultimately, this process facilitates cost reduction in pharmaceutical intermediates manufacturing by shortening the cycle time and improving the overall yield of the critical intermediate.
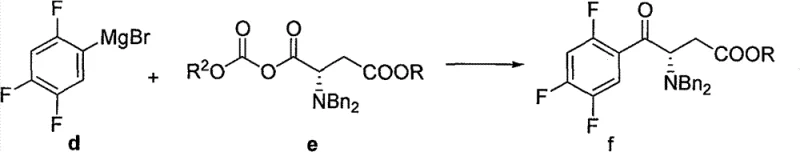
Mechanistic Insights into Grignard-Mediated Carbonyl Addition
The core of this synthetic innovation lies in the precise mechanistic execution of the nucleophilic attack of the Grignard reagent onto the mixed anhydride species. The process begins with the in situ generation of the 2,4,5-trifluorophenylmagnesium bromide, where the electron-withdrawing fluorine atoms on the aromatic ring modulate the reactivity of the organometallic species to prevent excessive basicity that could lead to enolization. Upon addition to the mixed anhydride derived from the protected amino acid, the Grignard reagent selectively attacks the more electrophilic carbonyl carbon adjacent to the leaving group. This selectivity is crucial for maintaining the integrity of the ester functionality elsewhere in the molecule, ensuring that the desired ketone is formed without over-addition to form a tertiary alcohol. The reaction mechanism is further stabilized by the coordination of the magnesium cation with the carbonyl oxygens, which lowers the activation energy for the transition state. Following the addition, the tetrahedral intermediate collapses to expel the carbonate leaving group, yielding the target beta-keto ester structure with high fidelity. Understanding this mechanistic nuance is vital for high-purity sitagliptin precursor production, as it allows process chemists to fine-tune stoichiometry and addition rates to minimize impurity formation.
Impurity control in this pathway is achieved through the careful management of reaction parameters and the inherent stability of the intermediates involved. The use of mild bases such as triethylamine during the anhydride formation step prevents racemization of the chiral center, preserving the optical purity required for the final biological activity. Furthermore, the subsequent hydrogenolysis step to remove the benzyl protecting group is conducted using palladium on carbon under controlled hydrogen pressure, which is a well-understood and easily scalable heterogeneous catalytic process. This step effectively cleaves the benzyl group while leaving the tert-butoxycarbonyl moiety intact, demonstrating excellent orthogonality in protecting group strategy. The workup procedure involves standard aqueous quenching and organic extraction, which are highly efficient at removing inorganic salts and magnesium residues. By avoiding complex chromatographic separations in the early stages, the process ensures that the commercial scale-up of complex pharmaceutical intermediates remains economically feasible. The robustness of this chemical sequence provides a significant advantage in terms of batch-to-batch consistency, a critical metric for regulatory compliance and supply chain reliability.
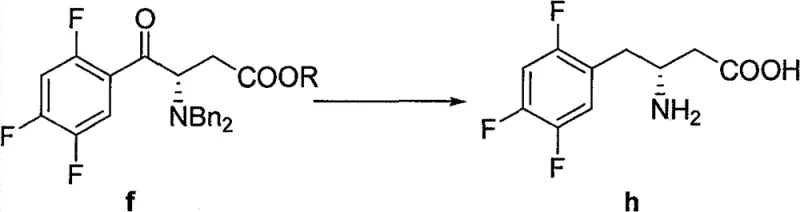
How to Synthesize Sitagliptin Intermediate Efficiently
The implementation of this synthetic route requires a systematic approach to unit operations, beginning with the preparation of the activated anhydride species in an anhydrous environment to prevent hydrolysis. Operators must ensure strict moisture control during the Grignard reagent formation, as water can deactivate the organometallic species and lead to the formation of unreactive fluorobenzene by-products. The addition of the Grignard reagent to the anhydride should be performed slowly with efficient agitation to manage the exotherm and maintain the reaction temperature within the optimal range specified in the patent examples. Detailed standardized synthesis steps are provided below to guide process engineers in replicating these results at pilot and production scales.
- Preparation of Mixed Anhydride: React protected amino acid derivatives with ethyl chloroformate in the presence of a tertiary amine base to form the activated mixed anhydride intermediate.
- Grignard Reagent Formation: Generate 2,4,5-trifluorophenylmagnesium bromide by reacting the corresponding bromobenzene with magnesium turnings in an ether solvent under inert atmosphere.
- Coupling and Workup: Add the Grignard reagent to the mixed anhydride at controlled temperatures, followed by aqueous quenching and extraction to isolate the keto-ester intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this Grignard-based methodology offers profound benefits that extend beyond mere technical feasibility, directly impacting the bottom line and supply security. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the risk associated with the volatility of platinum group metal prices. Furthermore, the reliance on commodity chemicals such as magnesium turnings, chloroformates, and common organic solvents ensures that the raw material supply chain is diversified and resilient against geopolitical disruptions. The simplified process flow reduces the number of isolation steps, which in turn lowers labor costs and decreases the consumption of utilities such as steam and cooling water. For procurement managers, this translates into a more predictable cost structure and the ability to negotiate better long-term contracts with suppliers. The enhanced safety profile also reduces insurance premiums and regulatory compliance costs associated with handling hazardous materials. Overall, this technology enables reducing lead time for high-purity API intermediates by streamlining the production schedule and minimizing downtime for equipment cleaning and changeover.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this route is the substitution of expensive chiral catalysts with stoichiometric Grignard reagents derived from inexpensive starting materials. By removing the requirement for high-pressure hydrogenation equipment specialized for asymmetric synthesis, capital investment is significantly lowered. The high yields reported in the patent examples indicate minimal material loss, which directly improves the mass efficiency of the process. Additionally, the use of recyclable solvents and the avoidance of complex purification techniques further contribute to substantial cost savings. These factors combine to create a manufacturing economics model that is highly competitive in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production is not held hostage by the supply constraints of niche reagents. Magnesium and trifluorobromobenzene are produced on a massive industrial scale, guaranteeing consistent availability and stable pricing. The robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-validation effort, providing flexibility in case of regional supply disruptions. This geographical diversification capability is crucial for maintaining continuous supply to global markets. The simplified logistics of handling non-hazardous intermediates also streamline transportation and storage requirements.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing reaction types that are standard in the fine chemical industry. The absence of toxic heavy metals simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. The reduction in solvent usage and energy consumption contributes to a lower carbon footprint, supporting corporate sustainability goals. The ability to run the reaction at near-ambient temperatures reduces the load on HVAC and refrigeration systems. These environmental advantages not only ensure compliance but also enhance the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or licensing agreements.
Q: What are the primary advantages of this Grignard route over transition metal catalysis?
A: This route eliminates the need for expensive precious metal catalysts such as Ruthenium or Rhodium, significantly reducing raw material costs and removing the regulatory burden of heavy metal residue testing in the final API.
Q: How does this process address impurity control concerns?
A: By utilizing mild reaction temperatures ranging from 0°C to 25°C and avoiding hazardous diazo compounds, the process minimizes side reactions and thermal degradation, resulting in a cleaner crude profile and higher overall purity.
Q: Is this synthetic route suitable for multi-ton commercial production?
A: Yes, the methodology relies on standard unit operations such as Grignard formation and liquid-liquid extraction, which are well-established in large-scale chemical manufacturing, ensuring robust scalability and supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN102199102B to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust manufacturing realities. We are committed to delivering stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to monitor every stage of the synthesis. Our facility is designed to handle reactive chemistries safely, including Grignard reactions and hydrogenations, with comprehensive safety systems in place. By partnering with us, clients gain access to a supply chain that is both agile and resilient, capable of adapting to changing market demands while maintaining the highest quality standards.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. Request a Customized Cost-Saving Analysis today to understand the potential economic impact of switching to this more efficient manufacturing process. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your volume needs. Let us help you secure a sustainable and cost-effective supply of high-quality Sitagliptin intermediates for your downstream operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
