Optimizing Sertindole Production: A Technical Analysis of Aqueous Copper-Catalyzed Coupling and Scalable Intermediates
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex antipsychotic agents, and the synthesis of Sertindole represents a critical area of process innovation. Patent CN1243001C details a transformative approach to manufacturing this potent 5-HT2 receptor antagonist, specifically addressing the bottlenecks associated with traditional organic solvent-based methodologies. By shifting the paradigm from hazardous, high-boiling solvents like N-methylpyrrolidone (NMP) to environmentally benign aqueous media, this technology offers a compelling value proposition for reliable sertindole supplier networks aiming to enhance sustainability. The core breakthrough lies in the copper-catalyzed coupling of 2,5-dichlorobenzoic acid derivatives with N-(4-fluorophenyl)glycine directly in water, a feat previously considered challenging due to solubility and reactivity constraints. This report analyzes the technical depth of this patent to provide R&D and procurement leaders with actionable insights into cost reduction in API manufacturing and supply chain resilience.
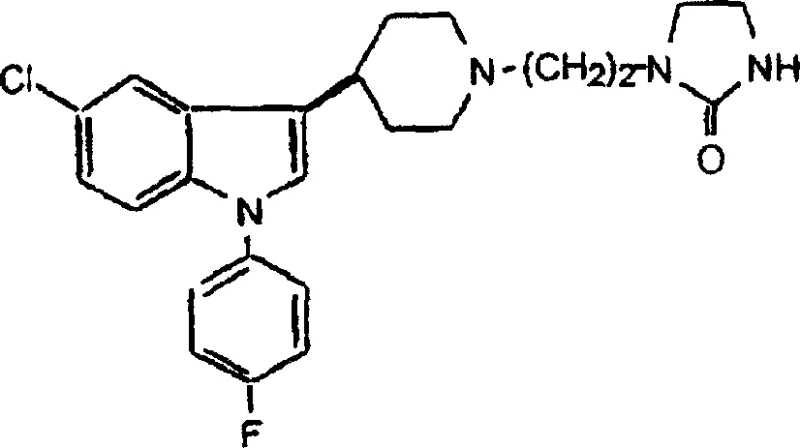
The structural complexity of Sertindole, as depicted above, necessitates precise control over regioselectivity and impurity profiles, particularly during the formation of the indole nucleus and the subsequent attachment of the piperidine moiety. The patent elucidates a pathway that not only improves yield but also simplifies the purification train, which is essential for meeting stringent regulatory standards for high-purity pharmaceutical intermediates. Understanding these mechanistic nuances is vital for partners evaluating the commercial viability of scaling this synthesis from kilogram to multi-ton production capacities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described by Perregaard et al., relied heavily on N-methylpyrrolidone (NMP) as the reaction medium for the critical Ullmann-type coupling step. While chemically feasible on a small laboratory scale, the use of NMP presents severe drawbacks for industrial application, primarily due to its high boiling point and poor water solubility which complicate downstream processing. The removal of NMP requires energy-intensive distillation or extensive extraction protocols, significantly increasing the operational expenditure and carbon footprint of the manufacturing process. Furthermore, reactions conducted in NMP at elevated temperatures ranging from 120°C to 130°C tend to generate substantial amounts of tarry by-products, which degrade the overall yield and complicate the isolation of the desired glycine intermediate. These tars often require complex chromatographic or multiple recrystallization steps to remove, rendering the process economically unviable for large-scale commercial production of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN1243001C leverages an aqueous alkaline environment to facilitate the copper-catalyzed coupling, effectively eliminating the need for problematic dipolar aprotic solvents. By conducting the reaction in water, potentially with co-solvents like ethylene glycol, the process achieves higher yields with negligible formation of tarry impurities, thereby streamlining the purification workflow. The reaction temperature is lowered to the reflux temperature of the aqueous medium, typically around 100°C or lower, which reduces thermal stress on the reagents and minimizes decomposition pathways. This shift not only enhances the safety profile of the operation by removing high-boiling, difficult-to-handle solvents but also aligns with green chemistry principles by utilizing water as the primary reaction medium. The result is a more robust and scalable process that significantly reduces the environmental burden associated with solvent waste disposal and recovery.
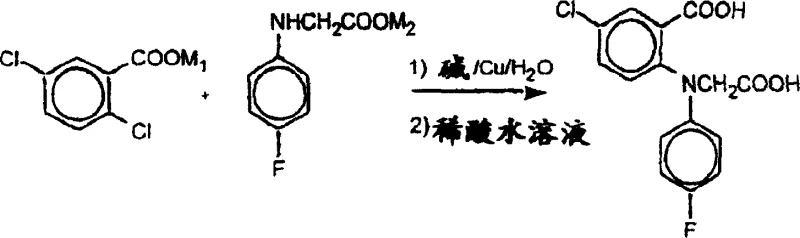
Mechanistic Insights into Aqueous Copper-Catalyzed Coupling
The success of this novel synthesis hinges on the precise manipulation of reaction conditions to facilitate the nucleophilic aromatic substitution in a polar protic solvent. The mechanism involves the generation of alkali metal salts of both 2,5-dichlorobenzoic acid and N-(4-fluorophenyl)glycine, which react in the presence of a copper-bronze catalyst. The choice of base is critical; alkali metal carbonates, particularly potassium carbonate, are preferred to neutralize the hydrochloric acid generated during the coupling without inducing excessive hydrolysis of the chlorobenzoic acid substrate. The patent specifies a molar ratio of base to acid that is slightly greater than stoichiometric to maintain alkalinity, yet controlled enough to prevent the degradation of the starting materials. This delicate balance ensures that the copper catalyst remains active and accessible to the substrates, promoting the formation of the C-N bond efficiently while suppressing side reactions that lead to impurity formation.
Impurity control is further enhanced by the physical properties of the aqueous medium, which favors the precipitation of the product upon acidification, allowing for easy separation from soluble inorganic salts and catalyst residues. The protocol describes a workup involving the addition of the filtered reaction mixture to dilute hydrochloric acid, causing the product to crystallize as a fine yellow-brown solid. This crystallization behavior is distinct from the oily or tarry residues often obtained in organic solvents, indicating a cleaner reaction profile. Subsequent purification via recrystallization from hot toluene or aqueous ethanol yields the intermediate with purity exceeding 98%, demonstrating the efficacy of the aqueous route in producing high-purity pharmaceutical intermediates suitable for downstream processing into the final active ingredient.
How to Synthesize Sertindole Intermediates Efficiently
The synthesis of the key glycine intermediate and the subsequent indole derivative requires strict adherence to the optimized parameters regarding temperature, stoichiometry, and acid concentration to ensure reproducibility and high yield. The process begins with the preparation of the alkali metal salts, followed by the copper-catalyzed coupling in water, and concludes with the cyclization and functionalization steps to build the indole core. Detailed standard operating procedures for each stage, including specific heating rates and filtration techniques, are essential for transferring this technology from the laboratory to the pilot plant. For a comprehensive guide on the exact experimental conditions and step-by-step execution, please refer to the standardized synthesis protocol below.
- React alkali metal salts of 2,5-dichlorobenzoic acid and N-(4-fluorophenyl)glycine in an aqueous alkaline environment with a copper-bronze catalyst at reflux temperatures.
- Isolate the glycine intermediate via acid precipitation and purify using hot toluene recrystallization to achieve high purity specifications.
- Condense the resulting 5-chloro-1-(4-fluorophenyl)indole with 4-piperidone hydrochloride in a mixture of acetic acid and concentrated hydrochloric acid to form the tetrahydropyridyl indole core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this aqueous-based synthesis route offers profound strategic benefits that extend beyond simple yield improvements. The elimination of N-methylpyrrolidone (NMP) and trifluoroacetic acid (TFA) from the process map removes two significant cost drivers and supply chain risks associated with hazardous material handling and disposal. By substituting these expensive and regulated solvents with water, acetic acid, and hydrochloric acid, the manufacturing process becomes inherently more cost-effective and less susceptible to regulatory fluctuations regarding solvent emissions. This shift facilitates cost reduction in API manufacturing by simplifying the infrastructure requirements, as there is no longer a need for specialized high-vacuum distillation units or aggressive corrosion-resistant reactors typically mandated for TFA usage.
- Cost Reduction in Manufacturing: The replacement of high-boiling organic solvents with water drastically simplifies the solvent recovery and recycling infrastructure, leading to substantial operational savings. Since water does not require energy-intensive distillation for removal and can be easily treated or reused, the utility costs associated with heating and cooling are significantly lowered. Furthermore, the avoidance of tarry by-products reduces the loss of raw materials and minimizes the need for costly chromatographic purification steps, directly improving the gross margin of the production run. The use of commodity chemicals like hydrochloric acid and acetic acid instead of specialized fluorinated acids further drives down the raw material procurement costs, making the overall process economically superior for large volume production.
- Enhanced Supply Chain Reliability: Relying on water as the primary reaction medium decouples the production schedule from the volatility of the organic solvent market, ensuring a more stable and predictable supply of critical intermediates. The raw materials required for this process, such as 2,5-dichlorobenzoic acid and 4-piperidone, are widely available commodity chemicals, reducing the risk of supply disruptions caused by niche reagent shortages. Additionally, the simplified logistics of handling non-hazardous aqueous waste streams streamline the environmental compliance workflow, preventing potential production stoppages due to waste disposal capacity limits. This robustness ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: The aqueous nature of the reaction mixture inherently supports safer scale-up operations, as the risks associated with flammable organic vapors and exothermic runaway reactions are markedly diminished. The process generates significantly less hazardous waste, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. By avoiding the use of persistent organic pollutants and volatile fluorinated compounds, the facility reduces its environmental liability and simplifies the permitting process for capacity expansion. This environmental compatibility makes the technology ideal for the commercial scale-up of complex pharmaceutical intermediates in regions with strict ecological oversight, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications of adopting this technology. Understanding these specifics is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines.
Q: What are the primary advantages of using an aqueous medium over NMP for Sertindole intermediate synthesis?
A: Replacing N-methylpyrrolidone (NMP) with water eliminates the need for complex solvent recovery systems, drastically reduces the formation of tarry by-products, and lowers the reaction temperature to the reflux point of water, enhancing both safety and operational efficiency.
Q: How does the new process control impurities during the copper-catalyzed coupling step?
A: The process utilizes a controlled addition of alkali metal carbonates to neutralize generated HCl without causing excessive hydrolysis of the dichlorobenzoic acid, while the use of water as a solvent inherently suppresses the polymerization side reactions common in high-boiling organic solvents.
Q: Why is the substitution of trifluoroacetic acid (TFA) with hydrochloric acid significant for scale-up?
A: Trifluoroacetic acid is highly volatile and corrosive, posing significant handling risks and equipment corrosion issues at large scales; replacing it with a mixture of acetic acid and hydrochloric acid mitigates these hazards while preventing the formation of undesired disubstituted piperidine impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sertindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the aqueous copper-catalyzed pathway described in CN1243001C and possesses the technical capability to implement these optimizations immediately. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Sertindole intermediate delivered meets the highest quality standards required by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage these process improvements for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and reliability for your Sertindole sourcing needs.
