Iodine-Catalyzed Innovation: Commercial-Scale Production of High-Purity Trifluoromethyl Triazoles for Pharmaceutical Applications
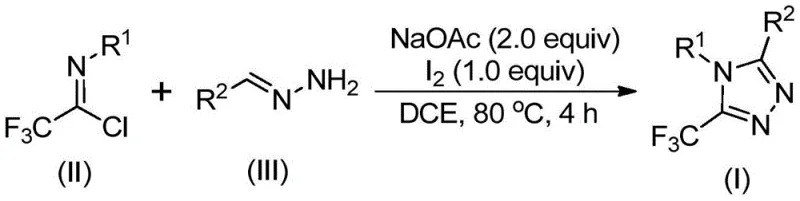
Patent CN110467579B represents a significant advancement in heterocyclic chemistry by introducing an iodine-promoted methodology for synthesizing 5-trifluoromethyl substituted 1,2,4-triazole compounds that addresses critical limitations in existing synthetic routes through elimination of expensive heavy metal catalysts while maintaining high reaction efficiency under standard laboratory conditions. This innovative approach leverages readily available starting materials including sodium acetate, trifluoroethylimidoyl chloride, and hydrazone derivatives which can be sourced from multiple commercial suppliers without complex purification steps or specialized handling requirements. Crucially, the reaction proceeds effectively without stringent anhydrous or anaerobic environments thereby reducing operational complexity and associated costs in manufacturing settings while demonstrating exceptional versatility through substrate design flexibility that enables synthesis of diverse triazole derivatives with varying substitution patterns at key positions. The methodology establishes a robust foundation for scalable production of these valuable compounds that serve as essential intermediates in pharmaceutical development pipelines where trifluoromethyl groups significantly enhance bioavailability and metabolic stability as documented in recent medicinal chemistry literature. Furthermore, this patent provides comprehensive experimental validation across fifteen distinct substrate combinations demonstrating consistent performance metrics that directly translate to commercial manufacturing viability without requiring specialized infrastructure or hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for trifluoromethylated triazoles typically rely on expensive transition metal catalysts such as palladium or copper complexes which introduce significant cost burdens through both catalyst procurement and subsequent heavy metal removal processes that require specialized equipment and generate hazardous waste streams. These methods often demand strictly controlled anhydrous and anaerobic conditions that necessitate expensive glovebox systems or Schlenk techniques thereby increasing operational complexity and limiting scalability in industrial settings where moisture sensitivity creates substantial batch-to-batch variability concerns. Furthermore, conventional routes frequently employ hazardous trifluoromethylation reagents that pose safety risks during handling and storage while exhibiting narrow substrate tolerance that restricts structural diversity in final products thus limiting their applicability across different pharmaceutical development programs. The requirement for multi-step purification protocols to remove metal residues adds considerable time and cost to production cycles while potentially compromising yield through intermediate degradation during lengthy processing sequences that are incompatible with modern continuous manufacturing paradigms.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iodine-promoted cyclization strategy that eliminates transition metals entirely while operating effectively under standard laboratory conditions without requiring specialized moisture or oxygen exclusion equipment. By utilizing elemental iodine as an inexpensive promoter alongside sodium acetate as a mild base, this approach achieves high-yielding triazole formation through a well-defined mechanistic pathway involving base-mediated carbon-nitrogen bond formation followed by oxidative iodination and intramolecular cyclization steps that proceed efficiently at moderate temperatures of approximately 80°C in common solvents like dichloroethane. The process demonstrates remarkable substrate flexibility allowing diverse substitution patterns at both the R¹ and R² positions through simple precursor modifications while maintaining consistent performance across various functional groups including halogens, alkyl chains, and heterocyclic moieties as evidenced by comprehensive experimental data covering fifteen distinct substrate combinations. Critically, the absence of air-sensitive components enables straightforward scale-up from laboratory to plant scale without process re-engineering while the simplified workup procedure involving basic filtration followed by single-column chromatography significantly reduces production timelines compared to conventional multi-step purification protocols.
Mechanistic Insights into Iodine-Promoted Triazole Cyclization
The reaction mechanism begins with base-mediated deprotonation of the hydrazone component by sodium acetate which facilitates nucleophilic attack on the electrophilic carbon of trifluoroethylimidoyl chloride forming an initial carbon-nitrogen bond that generates a key trifluoroacetamidine intermediate through intermolecular coupling. This intermediate subsequently undergoes spontaneous isomerization followed by base-promoted oxidative iodination where elemental iodine serves as both oxidant and iodine source to form a critical iodinated species that enables intramolecular electrophilic substitution at the ortho position relative to the nitrogen atom. The resulting cyclic intermediate then undergoes aromatization through dehydroiodination to yield the final five-membered triazole ring structure with precise regiocontrol ensuring consistent placement of the trifluoromethyl group at the desired position without requiring additional directing groups or protecting strategies. This mechanistic pathway operates efficiently under mild thermal conditions due to the synergistic effect between sodium acetate and iodine which lowers activation barriers while maintaining excellent functional group tolerance across diverse aromatic and heteroaromatic substrates as demonstrated by successful reactions with electron-donating methyl groups as well as electron-withdrawing nitro substituents.
The process inherently minimizes impurity formation through its stepwise mechanism where each transformation occurs under controlled conditions that prevent common side reactions such as over-halogenation or hydrolysis that plague conventional methods requiring harsher reagents or higher temperatures. The mild reaction environment preserves sensitive functional groups while ensuring complete conversion through optimized stoichiometry where sodium acetate serves dual roles as base and phase-transfer catalyst that enhances solubility of intermediates without promoting decomposition pathways. Post-reaction workup involves simple filtration to remove inorganic salts followed by standard column chromatography using silica gel which effectively separates any minor byproducts formed during the cyclization step while maintaining high product purity suitable for pharmaceutical applications where stringent quality specifications are required. This inherent selectivity reduces the need for additional purification steps that would otherwise increase production costs and decrease overall process efficiency while ensuring consistent impurity profiles across different batch sizes.
How to Synthesize Trifluoromethyl Triazoles Efficiently
This patented methodology provides a robust framework for manufacturing high-purity trifluoromethyl triazole intermediates through a carefully optimized sequence that balances reactivity with operational simplicity while maintaining excellent yield consistency across diverse substrate combinations as validated by comprehensive experimental data covering fifteen distinct examples with varying substitution patterns. The process eliminates traditional barriers to commercial implementation by operating under standard laboratory conditions without requiring specialized equipment or hazardous reagents while delivering products that meet pharmaceutical quality standards through straightforward purification protocols. Detailed standardized synthesis procedures have been developed based on extensive process optimization studies that ensure reproducibility across different manufacturing scales from laboratory to plant level while maintaining strict adherence to safety protocols throughout all operational phases. The following step-by-step guide outlines the precise implementation protocol derived from patent CN110467579B that enables reliable production of these valuable intermediates with minimal technical barriers to adoption.
- Combine sodium acetate (2.0 equivalents), trifluoroethylimidoyl chloride (I equivalent), and hydrazone derivative (2 equivalents) in dichloroethane solvent under ambient conditions without anhydrous requirements.
- Heat the reaction mixture to precisely controlled temperature of 80°C with continuous stirring for four hours to facilitate carbon-nitrogen bond formation and intermediate isomerization.
- Add elemental iodine (0.5 equivalents) and maintain heating at constant temperature for one additional hour before standard workup procedures including filtration and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach directly addresses critical pain points faced by procurement and supply chain professionals through its elimination of expensive catalyst systems while simultaneously improving process reliability and reducing environmental impact across the entire production lifecycle from raw material sourcing to final product delivery. The methodology's compatibility with standard manufacturing infrastructure significantly lowers capital investment requirements compared to conventional approaches requiring specialized equipment for handling air-sensitive reagents or removing heavy metal contaminants from final products. By leveraging readily available starting materials with established global supply chains, this process ensures consistent raw material availability while minimizing vulnerability to single-source dependencies that could disrupt production schedules during periods of market volatility or geopolitical instability.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes significant expenses associated with catalyst procurement and subsequent heavy metal removal processes from final products while reducing waste treatment costs through simplified reaction workup procedures that generate fewer hazardous byproducts compared to conventional methods requiring multiple purification steps. This streamlined approach reduces both raw material costs and downstream processing requirements while maintaining high product quality standards through inherent reaction selectivity that minimizes impurity formation during synthesis.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials with established global supply chains ensures consistent raw material availability while eliminating dependencies on specialized reagents that could create bottlenecks during periods of market volatility or geopolitical instability. The process operates effectively under standard laboratory conditions without requiring specialized handling equipment or controlled environments which significantly reduces production cycle variability while enabling rapid response to changing demand patterns through flexible manufacturing scheduling.
- Scalability and Environmental Compliance: The absence of air-sensitive components allows straightforward scale-up from laboratory to plant scale without process re-engineering while maintaining consistent yield profiles across different batch sizes as demonstrated by successful gram-scale experiments documented in the patent literature. The simplified workup procedure generates minimal hazardous waste streams compared to conventional methods requiring multiple purification steps thus reducing environmental impact while meeting increasingly stringent regulatory requirements for sustainable chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns raised by procurement teams regarding implementation feasibility and quality assurance when adopting this patented manufacturing approach for pharmaceutical intermediate production based on detailed analysis of patent CN110467579B's experimental data and process specifications.
Q: Why does this method eliminate heavy metal catalysts compared to conventional triazole syntheses?
A: The iodine-promoted mechanism enables efficient cyclization through oxidative iodination pathways that bypass traditional transition metal requirements. This innovation removes costly catalyst procurement and complex metal removal steps while maintaining high regioselectivity across diverse substrate combinations as demonstrated in patent examples.
Q: How does this process ensure pharmaceutical-grade purity without extensive purification?
A: The reaction's inherent selectivity minimizes side product formation through controlled base-promoted steps and mild iodine-mediated cyclization. Standard filtration followed by single-column chromatography suffices for achieving stringent purity specifications required in drug intermediate manufacturing.
Q: What scalability advantages does this methodology offer for commercial production?
A: The absence of air-sensitive reagents allows straightforward scale-up from laboratory to plant scale without specialized equipment. The process maintains consistent yields across various substrate combinations as demonstrated in multiple gram-scale experiments with readily available starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
This patented technology represents a significant advancement in triazole synthesis methodology that aligns perfectly with NINGBO INNO PHARMCHEM's expertise as a leading CDMO provider specializing in complex heterocyclic compounds where we bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation capable of detecting trace impurities at parts-per-million levels required for pharmaceutical applications. Our integrated manufacturing platform combines deep technical knowledge with flexible production capabilities that enable rapid implementation of this innovative process while ensuring seamless technology transfer from laboratory to commercial scale through our dedicated process development teams who specialize in optimizing patented methodologies for industrial implementation.
We invite you to initiate a technical discussion with our procurement team to explore how this methodology can enhance your supply chain resilience while delivering substantial value through our Customized Cost-Saving Analysis service which provides detailed route feasibility assessments tailored to your specific production requirements along with comprehensive COA data packages demonstrating our capability to consistently meet your quality specifications for these critical pharmaceutical intermediates.
