Revolutionizing Triazole Synthesis: Scalable Production of High-Purity 5-Trifluoromethyl Intermediates for Pharmaceutical Applications
The patent CN113121462B discloses an innovative metal-free synthesis method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, representing a significant advancement in the production of high-value pharmaceutical intermediates. This novel approach eliminates the need for hazardous azide compounds and transition metal catalysts that characterize conventional methodologies, offering a safer and more sustainable pathway for manufacturing these critical building blocks used in drug development. The process utilizes readily available starting materials under mild reaction conditions (50–70°C), enabling efficient production of high-purity intermediates essential for pharmaceutical applications where stringent quality requirements must be met.
Advanced Reaction Mechanism and Purity Control
The patented process employs a base-promoted reaction between trifluoroethylimidoyl chloride and diazo compounds under mild conditions using cesium carbonate as the base in acetonitrile solvent. Unlike traditional copper-catalyzed azide-alkyne cycloaddition methods that require toxic azides followed by additional trifluoromethylation steps, this methodology proceeds through a unique mechanism involving intermolecular nucleophilic addition followed by intramolecular 5-endo-dig cyclization as illustrated in 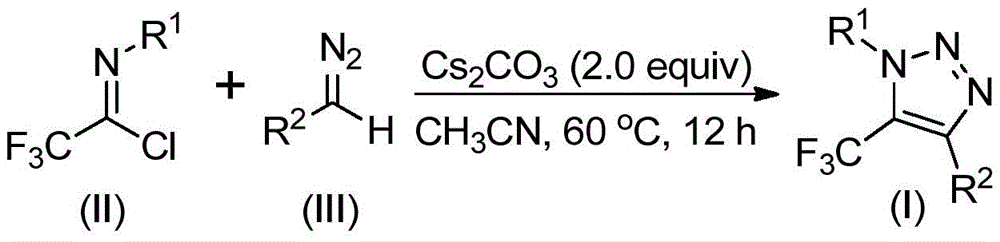 . The absence of transition metals completely eliminates potential metal contamination in the final product, which is particularly critical for pharmaceutical applications where strict metal limits are enforced by regulatory agencies such as the FDA and EMA.
. The absence of transition metals completely eliminates potential metal contamination in the final product, which is particularly critical for pharmaceutical applications where strict metal limits are enforced by regulatory agencies such as the FDA and EMA.
Impurity profile management is significantly enhanced through this novel synthetic route as demonstrated by comprehensive NMR and HRMS data provided for compounds (I-1) through (I-5) shown in 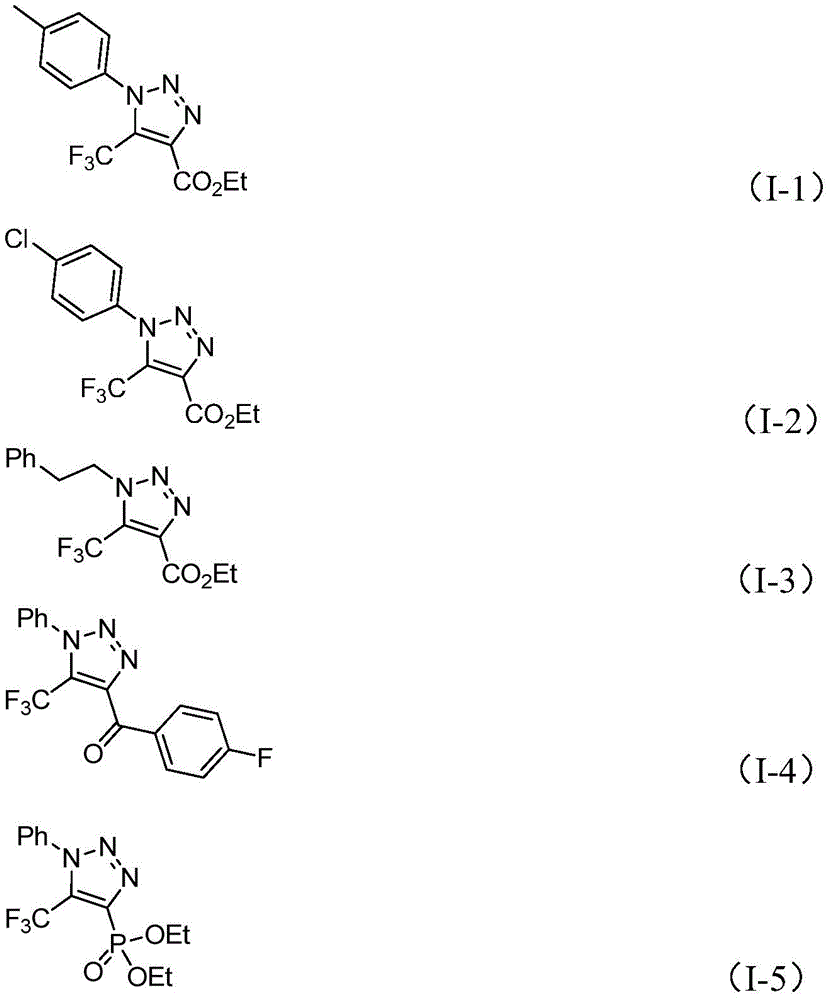 . Traditional methods often generate multiple impurities due to side reactions involving azide decomposition or incomplete metal removal processes requiring multiple purification steps. In contrast, this patent describes a cleaner reaction pathway with fewer side products due to the elimination of hazardous intermediates that typically cause side reactions in conventional processes. The simplified workup procedure—consisting of filtration, silica gel mixing, and column chromatography—further contributes to superior product purity without requiring specialized equipment for metal removal or hazardous material handling protocols.
. Traditional methods often generate multiple impurities due to side reactions involving azide decomposition or incomplete metal removal processes requiring multiple purification steps. In contrast, this patent describes a cleaner reaction pathway with fewer side products due to the elimination of hazardous intermediates that typically cause side reactions in conventional processes. The simplified workup procedure—consisting of filtration, silica gel mixing, and column chromatography—further contributes to superior product purity without requiring specialized equipment for metal removal or hazardous material handling protocols.
Commercial Advantages for Procurement and Supply Chain Management
This innovative manufacturing process addresses critical pain points in the production of complex triazole intermediates, offering substantial benefits for procurement and supply chain operations in pharmaceutical manufacturing. By eliminating hazardous materials and simplifying the synthetic pathway, the method provides a more reliable and cost-effective solution for producing these high-value intermediates at commercial scale while meeting stringent quality requirements.
- Reduced raw material costs and supply chain complexity: The process utilizes readily available starting materials—trifluoroethylimidoyl chloride can be rapidly synthesized from common aromatic amines, triphenylphosphine, carbon tetrachloride, and trifluoroacetic acid, while aryl diazo compounds are conveniently prepared from corresponding acid chlorides and trimethylsilyl reagents as shown in
 . This eliminates dependency on specialized or hazardous azide compounds that require special handling protocols, reducing both material costs and supply chain vulnerabilities associated with restricted chemical handling requirements. The elimination of transition metal catalysts also removes the need for expensive catalyst procurement and associated supply chain management complexities while maintaining high reaction efficiency with inexpensive cesium carbonate as the base promoter.
. This eliminates dependency on specialized or hazardous azide compounds that require special handling protocols, reducing both material costs and supply chain vulnerabilities associated with restricted chemical handling requirements. The elimination of transition metal catalysts also removes the need for expensive catalyst procurement and associated supply chain management complexities while maintaining high reaction efficiency with inexpensive cesium carbonate as the base promoter. - Shorter production timelines and improved supply reliability: The simplified reaction sequence with fewer processing steps directly translates to reduced manufacturing cycle times compared to conventional methods requiring multiple synthetic steps and extensive purification to remove metal residues as depicted in
 . The patent demonstrates that the reaction can be easily scaled to gram quantities without significant process re-engineering challenges, indicating strong potential for commercial scale-up to multi-kilogram production volumes required for clinical trials or commercial manufacturing. This scalability combined with the absence of hazardous intermediates enables faster batch turnaround times with more predictable production schedules while maintaining consistent product quality across different substituent variations.
. The patent demonstrates that the reaction can be easily scaled to gram quantities without significant process re-engineering challenges, indicating strong potential for commercial scale-up to multi-kilogram production volumes required for clinical trials or commercial manufacturing. This scalability combined with the absence of hazardous intermediates enables faster batch turnaround times with more predictable production schedules while maintaining consistent product quality across different substituent variations. - Reduced environmental impact and waste treatment costs: By eliminating transition metals and hazardous azide compounds from the synthetic pathway, this method significantly reduces generation of toxic waste streams requiring specialized treatment procedures typically needed for metal-contaminated waste streams or explosive materials handling protocols. The simplified workup procedure minimizes solvent usage by eliminating multiple purification steps required to remove metal contaminants while using common solvents like acetonitrile that can be readily recovered through standard distillation techniques. This environmental benefit translates directly to cost savings through reduced waste treatment expenses while decreasing regulatory compliance burdens associated with hazardous waste management protocols required by environmental protection agencies worldwide.
Comparative Analysis: Traditional vs. Novel Synthetic Approaches
The Limitations of Conventional Methods
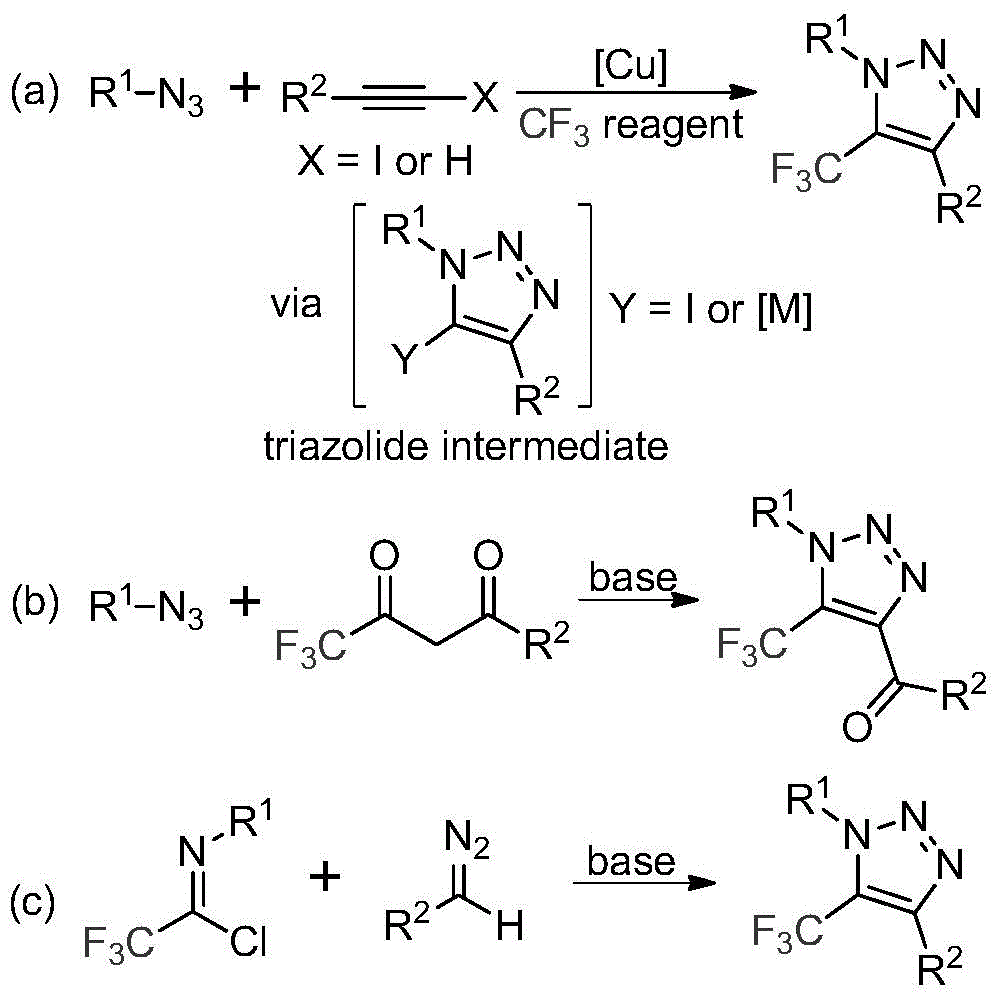
Traditional synthesis of trifluoromethyl-substituted 1,2,3-triazoles primarily relies on two approaches: copper-catalyzed [3+2] cycloaddition of alkynes with organic azides followed by trifluoromethylation (pathway a), or organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones (pathway b), both requiring hazardous azide compounds as shown in  . These methods suffer from significant drawbacks including safety concerns due to explosive hazards associated with azide handling that necessitate specialized facilities with explosion-proof equipment and trained personnel certified in hazardous material handling procedures. Additionally, copper-catalyzed methods generate metal-contaminated products requiring extensive purification steps to meet pharmaceutical quality standards which increases processing time while reducing overall yield through multiple isolation procedures.
. These methods suffer from significant drawbacks including safety concerns due to explosive hazards associated with azide handling that necessitate specialized facilities with explosion-proof equipment and trained personnel certified in hazardous material handling procedures. Additionally, copper-catalyzed methods generate metal-contaminated products requiring extensive purification steps to meet pharmaceutical quality standards which increases processing time while reducing overall yield through multiple isolation procedures.
The Novel Approach
The patented methodology overcomes these limitations through an elegant metal-free process utilizing stable starting materials without hazardous intermediates as demonstrated by the reaction scheme in 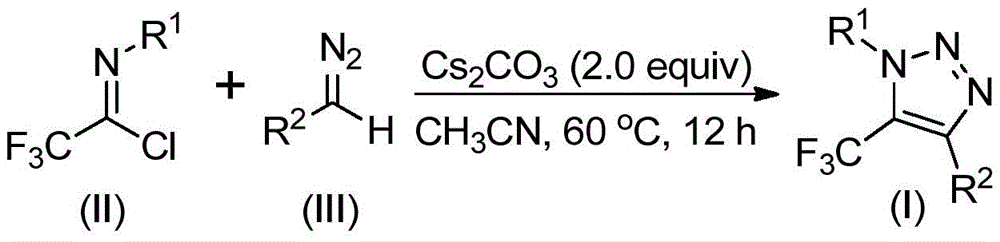 . The process demonstrates excellent functional group tolerance across various substituents (R¹ including phenyl groups with methyl or methoxy substitutions; R² including benzoyl or phospholipid groups), as evidenced by successful synthesis of diverse compounds (I-1 through I-5) shown in structural examples from
. The process demonstrates excellent functional group tolerance across various substituents (R¹ including phenyl groups with methyl or methoxy substitutions; R² including benzoyl or phospholipid groups), as evidenced by successful synthesis of diverse compounds (I-1 through I-5) shown in structural examples from  . This versatility allows rapid adaptation to different pharmaceutical development needs without requiring significant process modifications while maintaining consistent product quality across different structural variants required by various drug development programs.
. This versatility allows rapid adaptation to different pharmaceutical development needs without requiring significant process modifications while maintaining consistent product quality across different structural variants required by various drug development programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN113121462B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
