Unlocking Scalable Production of High-Purity 5-Trifluoromethyl Triazoles Through Innovative Base-Promoted Chemistry
Patent CN113121462B introduces a groundbreaking metal-free and azide-free synthesis route for 5-trifluoromethyl substituted 1,2,3-triazole compounds that serve as critical building blocks in pharmaceutical development due to their prevalence in bioactive molecules such as β3 adrenergic receptor agonists and various agrochemical applications. This innovative methodology addresses longstanding challenges in traditional triazole synthesis by eliminating hazardous reagents while maintaining high efficiency and scalability through a base-promoted mechanism that operates under mild conditions without transition metals or explosive azides. The process leverages readily available starting materials including diazo compounds and trifluoroethylimidoyl chloride under optimized reaction parameters that facilitate seamless scale-up from laboratory to commercial production volumes. With operational simplicity that reduces both environmental impact and production complexity while delivering consistently high-purity intermediates essential for pharmaceutical manufacturing processes worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
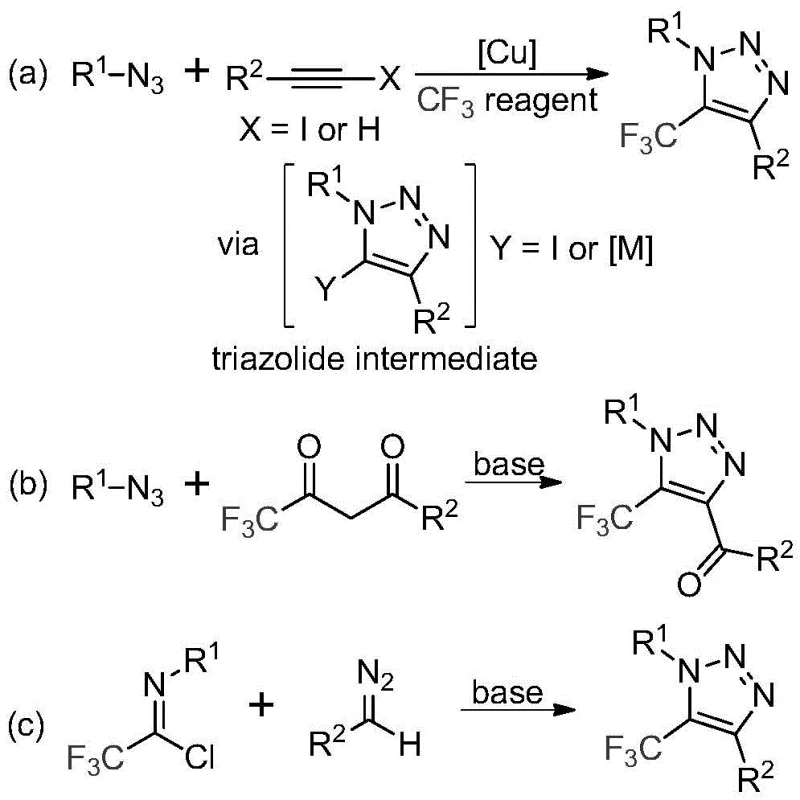
Traditional approaches for synthesizing trifluoromethyl-substituted triazoles predominantly rely on copper-catalyzed [3+2] cycloadditions between alkynes and organic azides followed by trifluoromethylation steps or organocatalytic reactions involving trifluoromethyl ketones which present significant operational hazards due to the inherent instability and toxicity of organic azides that are prone to explosive decomposition under routine handling conditions. Furthermore these methodologies require transition metal catalysts introducing complex purification challenges to remove trace metal residues that could compromise product purity in pharmaceutical applications while the multi-step nature contributes to reduced overall yields and increased production costs. The necessity for specialized trifluoromethylating reagents adds both financial burden and supply chain vulnerability creating substantial barriers to commercial implementation despite the valuable biological activity profiles of these heterocyclic compounds.
The Novel Approach
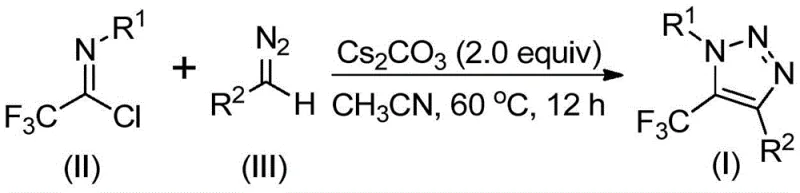
The patented methodology overcomes these constraints through an elegant base-promoted reaction between diazo compounds and trifluoroethylimidoyl chloride that operates without metals or azides entirely utilizing inexpensive cesium carbonate as a base promoter in acetonitrile solvent at moderate temperatures (50–70°C) to directly form the desired triazole ring structure with excellent functional group tolerance across diverse aromatic and aliphatic substrates. By eliminating hazardous reagents and transition metals this process achieves superior safety characteristics while simplifying purification protocols to basic filtration followed by column chromatography without requiring specialized equipment or hazardous material handling procedures. The reaction demonstrates remarkable versatility as evidenced by successful synthesis of fifteen distinct derivatives with yields ranging from forty percent to ninety-one percent making it exceptionally adaptable for producing customized intermediates required by pharmaceutical developers while significantly reducing both operational complexity and environmental impact compared to conventional methodologies.
Mechanistic Insights into Base-Promoted Triazole Formation
The reaction mechanism proceeds through a base-mediated nucleophilic addition where the diazo compound attacks the electrophilic carbon of trifluoroethylimidoyl chloride forming a key intermediate that subsequently undergoes intramolecular cyclization through a concerted addition-cyclization sequence that directly constructs the triazole ring system without requiring external oxidants or catalysts. This mechanistic pathway avoids the formation of potentially unstable triazolide intermediates common in traditional copper-catalyzed routes by leveraging the inherent reactivity of the diazo functionality under basic conditions ensuring high regioselectivity for the desired five-trifluoromethyl substitution pattern while maintaining compatibility with sensitive functional groups across various substrate combinations as confirmed by extensive experimental validation.
Impurity control is achieved through precise stoichiometric balance between reactants typically maintained at one-to-one-point-five-to-two molar ratio of imidoyl chloride to diazo compound to cesium carbonate along with molecular sieves to maintain anhydrous conditions during reaction preventing hydrolysis side reactions that could generate impurities. The absence of transition metals eliminates concerns about metal contamination that could affect product quality in pharmaceutical applications while mild reaction temperature optimization prevents thermal decomposition pathways that might produce unwanted byproducts. Post-reaction purification via silica gel chromatography effectively removes minor impurities resulting from incomplete conversion ensuring consistent delivery of products meeting stringent pharmaceutical purity requirements as verified through comprehensive HRMS and NMR characterization across multiple examples.
How to Synthesize Five-Trifluoromethyl Triazole Efficiently
This innovative synthesis route represents a significant advancement in heterocyclic chemistry offering pharmaceutical manufacturers a streamlined pathway to produce high-value triazole intermediates with superior safety profiles compared to conventional methods. The process eliminates multiple pain points associated with traditional approaches including hazardous reagent handling complex purification requirements and scalability limitations while maintaining excellent functional group tolerance across diverse substrate combinations as demonstrated through extensive experimental validation. Detailed standardized operating procedures have been developed based on this patent's technical disclosures which provide precise guidance for implementing this methodology at commercial scale; comprehensive step-by-step instructions are provided below to ensure successful technology transfer from laboratory development to industrial production environments.
- Combine trifluoroethylimidoyl chloride (0.2 mmol), diazo compound (0.3 mmol), cesium carbonate (0.4 mmol), and molecular sieves (40 mg) in acetonitrile (5 mL) under inert atmosphere
- Heat reaction mixture at controlled temperature (60°C optimal) with continuous stirring for specified duration (8–16 hours) while monitoring conversion
- Execute post-reaction processing through filtration followed by silica gel column chromatography using optimized eluent systems
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial commercial benefits specifically addressing critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing organizations seeking reliable sources for complex heterocyclic intermediates. The elimination of hazardous materials reduces regulatory compliance burdens while simplified processing requirements lower operational costs throughout the production lifecycle creating significant competitive advantages for manufacturers adopting this technology. Enhanced process robustness combined with readily available starting materials ensures greater supply chain resilience compared to conventional routes dependent on specialized reagents with limited suppliers.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes both procurement costs and complex purification steps required to remove trace metal residues thereby substantially reducing overall production expenses without compromising product quality or yield consistency across multiple substrate combinations as demonstrated in experimental validation.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials including diazo compounds and trifluoroethylimidoyl chloride which can be readily synthesized from common precursors ensures greater supply chain flexibility compared to routes requiring specialized or hazardous reagents with limited global suppliers thus significantly improving material availability security.
- Scalability and Environmental Compliance: The straightforward single-step process operating under mild conditions enables seamless scale-up from laboratory development to commercial production volumes while generating minimal waste streams compared to multi-step conventional methods thereby enhancing environmental sustainability credentials without requiring additional capital investment.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by industry professionals regarding implementation of this patented methodology based on specific advantages highlighted in the patent documentation including safety improvements process efficiency and quality control aspects relevant to pharmaceutical manufacturing environments where stringent regulatory requirements apply.
Q: How does this method eliminate safety hazards compared to conventional triazole synthesis?
A: The patented process replaces explosive organic azides with stable diazo compounds while eliminating transition metal catalysts entirely through cesium carbonate promotion, significantly reducing explosion risks and eliminating metal contamination concerns without compromising reaction efficiency.
Q: What scalability advantages does this synthesis route offer for commercial production?
A: The methodology demonstrates seamless scalability from laboratory gram-scale reactions to industrial production volumes as evidenced by consistent yields across diverse substrates without requiring specialized equipment or hazardous reagent handling procedures.
Q: How does the absence of metal catalysts impact pharmaceutical purity requirements?
A: By avoiding transition metals entirely, the process eliminates costly metal removal steps required in API manufacturing while ensuring compliance with stringent purity specifications through simplified purification protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Five-Trifluoromethyl Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation capable of meeting global regulatory standards including ICH guidelines. As a trusted CDMO partner specializing in complex heterocyclic systems like triazoles we combine deep synthetic expertise with robust manufacturing capabilities to deliver consistent high-quality intermediates meeting both technical requirements and commercial timelines across multiple therapeutic areas where these critical building blocks are utilized.
Request a Customized Cost-Saving Analysis from our technical procurement team today to evaluate how this efficient synthesis route can optimize your supply chain; we will provide specific COA data and route feasibility assessments tailored to your production needs along with comprehensive technical support throughout implementation phases.
