Advanced Synthetic Route for Gefitinib Intermediates Enhancing Commercial Scalability and Purity
Advanced Synthetic Route for Gefitinib Intermediates Enhancing Commercial Scalability and Purity
The pharmaceutical industry continuously seeks robust and economically viable synthetic pathways for high-value oncology drugs like Gefitinib, a potent EGFR tyrosine kinase inhibitor widely used in the treatment of non-small cell lung cancer. Patent CN102180844A introduces a transformative approach to the synthesis of Gefitinib and its key intermediates, addressing critical bottlenecks found in legacy manufacturing processes. This innovation pivots away from hazardous chlorinating agents and complex multi-step sequences, proposing a streamlined route that begins with the inexpensive and commercially abundant 3-hydroxy-4-methoxybenzonitrile. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the cost of goods sold (COGS) while enhancing the environmental sustainability of the production lifecycle. The technical depth of this patent lies in its strategic reordering of functional group transformations, specifically delaying the introduction of the morpholine side chain until after the formation of the critical formamidine intermediate, thereby improving overall yield and purity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Gefitinib has relied heavily on routes that involve the chlorination of a quinazolinone core to activate the C4 position for nucleophilic attack by the aniline moiety. As detailed in prior art such as WO9633980 and CN1300118, these conventional pathways typically necessitate the use of aggressive chlorinating reagents like phosphorus oxychloride (POCl3) or thionyl chloride (SOCl2). These reagents pose severe challenges for modern manufacturing facilities, including high corrosivity, significant generation of acidic waste streams, and stringent regulatory hurdles regarding worker safety and environmental discharge. Furthermore, the chlorination step often leads to the formation of polychlorinated by-products that are structurally similar to the desired intermediate, making purification arduous and resulting in substantial yield losses during recrystallization. The reliance on these harsh conditions also limits the choice of protecting groups and requires specialized corrosion-resistant reactor equipment, driving up capital expenditure and operational complexity for generic manufacturers seeking to enter the market.
The Novel Approach
In stark contrast, the methodology disclosed in CN102180844A circumvents these issues by employing a formamidine-mediated cyclization strategy that eliminates the need for direct ring chlorination. The process initiates with the alkylation of 3-hydroxy-4-methoxybenzonitrile, a low-cost commodity chemical, to install the propoxy side chain early in the sequence. This is followed by a regioselective nitration and subsequent reduction to generate the aniline precursor, which is then condensed with N,N-dimethylformamide dimethyl acetal (DMF-DMA) to form a reactive formamidine species. 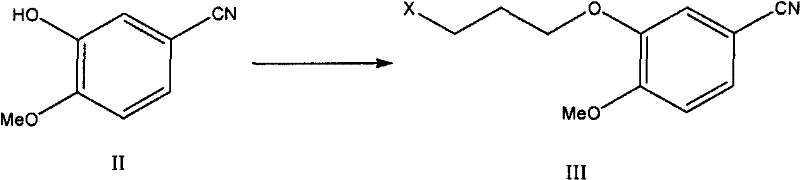 This strategic sequence allows for the construction of the quinazoline ring under much milder acidic conditions using glacial acetic acid, rather than harsh Lewis acids. By avoiding the chlorination step entirely, the process not only mitigates environmental risks but also simplifies the impurity profile, ensuring that the final API meets stringent pharmacopeial standards with less intensive downstream processing. This shift in synthetic logic demonstrates a clear evolution towards greener chemistry principles without compromising on the structural integrity or biological activity of the final therapeutic agent.
This strategic sequence allows for the construction of the quinazoline ring under much milder acidic conditions using glacial acetic acid, rather than harsh Lewis acids. By avoiding the chlorination step entirely, the process not only mitigates environmental risks but also simplifies the impurity profile, ensuring that the final API meets stringent pharmacopeial standards with less intensive downstream processing. This shift in synthetic logic demonstrates a clear evolution towards greener chemistry principles without compromising on the structural integrity or biological activity of the final therapeutic agent.
Mechanistic Insights into Formamidine-Mediated Cyclization
The core chemical innovation of this patent resides in the efficient conversion of the substituted benzonitrile derivative into the quinazoline core via a formamidine intermediate. Mechanistically, the reaction of the 2-aminobenzonitrile derivative with DMF-DMA generates an N'-aryl-N,N-dimethylformamidine, which serves as a masked electrophile at the formyl carbon. 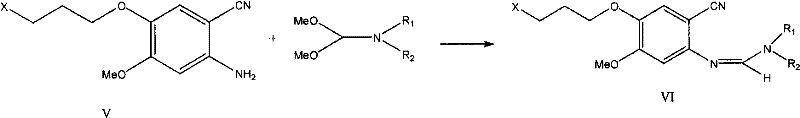 Upon heating in the presence of 3-chloro-4-fluoroaniline and a solvent like glacial acetic acid, the exocyclic nitrogen of the formamidine undergoes a condensation reaction with the aniline, followed by an intramolecular cyclization where the nitrile nitrogen attacks the imine carbon. This cascade effectively closes the pyrimidine ring of the quinazoline system. The beauty of this mechanism lies in its atom economy and the volatility of the dimethylamine by-product, which drives the equilibrium forward and simplifies workup. Unlike the traditional Vilsmeier-Haack type activations that require stoichiometric amounts of POCl3, this cyclization proceeds cleanly, minimizing the risk of over-reaction or degradation of the sensitive fluorochloro-aniline moiety. The result is a highly selective transformation that preserves the stereochemical and functional integrity of the molecule throughout the ring-closing event.
Upon heating in the presence of 3-chloro-4-fluoroaniline and a solvent like glacial acetic acid, the exocyclic nitrogen of the formamidine undergoes a condensation reaction with the aniline, followed by an intramolecular cyclization where the nitrile nitrogen attacks the imine carbon. This cascade effectively closes the pyrimidine ring of the quinazoline system. The beauty of this mechanism lies in its atom economy and the volatility of the dimethylamine by-product, which drives the equilibrium forward and simplifies workup. Unlike the traditional Vilsmeier-Haack type activations that require stoichiometric amounts of POCl3, this cyclization proceeds cleanly, minimizing the risk of over-reaction or degradation of the sensitive fluorochloro-aniline moiety. The result is a highly selective transformation that preserves the stereochemical and functional integrity of the molecule throughout the ring-closing event.
From an impurity control perspective, this mechanism offers distinct advantages by reducing the potential for halogen scrambling or oxidative degradation often seen in harsher chlorination protocols. The use of mild acid catalysis ensures that the methoxy and morpholine groups remain stable, preventing demethylation or ring-opening side reactions that could compromise the quality of the active pharmaceutical ingredient. Furthermore, the intermediate formamidine species can be isolated or carried forward as an oil, providing flexibility in process design. The patent data indicates that the reaction can be monitored effectively via TLC, allowing for precise endpoint determination to prevent the accumulation of unreacted starting materials or hydrolysis products. This level of control is paramount for maintaining a consistent impurity profile across different production batches, a key requirement for regulatory approval in global markets.
How to Synthesize Gefitinib Efficiently
The practical execution of this synthetic route involves a series of well-defined unit operations that are compatible with standard multipurpose chemical reactors. The process begins with the nucleophilic substitution of the phenolic hydroxyl group, followed by electrophilic aromatic substitution for nitration, and concludes with the reductive and cyclization steps. Each stage has been optimized for temperature and stoichiometry to maximize throughput. For a detailed breakdown of the specific reaction conditions, reagent ratios, and workup procedures required to implement this technology in a GMP environment, please refer to the standardized protocol below.
- Perform nucleophilic substitution on 3-hydroxy-4-methoxybenzonitrile with 1-chloro-3-bromopropane using potassium carbonate to form the chloropropoxy intermediate.
- Conduct nitration of the intermediate using nitric acid and sulfuric acid, followed by reduction of the nitro group to an amino group using sodium hydrosulfite.
- React the amino intermediate with DMF-DMA to form a formamidine derivative, substitute the chloride with morpholine, and finally cyclize with 3-chloro-4-fluoroaniline to yield Gefitinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates directly into enhanced operational resilience and cost efficiency. The primary driver of value is the substitution of expensive and hazard-prone reagents with commoditized chemicals that are available from multiple global suppliers. By eliminating the dependency on phosphorus oxychloride, manufacturers can significantly reduce their expenditure on hazardous waste disposal and corrosion-resistant infrastructure maintenance. This shift not only lowers the variable cost per kilogram of production but also de-risks the supply chain against regulatory crackdowns on toxic effluent discharge. The ability to source starting materials like 3-hydroxy-4-methoxybenzonitrile from a broad vendor base ensures continuity of supply, preventing production stoppages due to single-source bottlenecks.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental simplification of the synthetic tree. By removing the chlorination step, the process avoids the need for specialized scrubbing systems and neutralization tanks required to handle hydrogen chloride and phosphoric acid waste. This reduction in auxiliary processing requirements leads to substantial cost savings in both utility consumption and waste management fees. Additionally, the higher selectivity of the formamidine cyclization reduces the loss of valuable intermediates during purification, effectively increasing the overall mass yield of the process. These efficiencies compound to lower the total cost of ownership for the manufacturing asset, allowing for more competitive pricing strategies in the generic drug market.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials such as 1-chloro-3-bromopropane and morpholine ensures that the supply chain remains robust against market fluctuations. Unlike specialized reagents that may have long lead times or limited production capacity, the inputs for this route are produced at scale by the basic chemical industry. This abundance allows procurement teams to negotiate favorable long-term contracts and maintain strategic inventory buffers without tying up excessive working capital. Furthermore, the mild reaction conditions reduce the risk of unplanned downtime caused by equipment failure or safety incidents, ensuring a steady flow of intermediates to the final API production line.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is straightforward due to the absence of exothermic hazards associated with chlorination reactions. The thermal profile of the nitration and cyclization steps is manageable with standard jacketed reactors, facilitating a smooth technology transfer. From an environmental standpoint, the process aligns with green chemistry initiatives by minimizing the use of heavy metals and corrosive halogens. This compliance posture simplifies the permitting process for new manufacturing sites and enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies who prioritize responsible sourcing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Gefitinib synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of adopting this route for your specific production needs.
Q: What are the primary advantages of this new Gefitinib synthesis route over conventional methods?
A: The primary advantages include the use of cheap and readily available starting materials like 3-hydroxy-4-methoxybenzonitrile, mild reaction conditions that do not require special equipment, and the avoidance of highly toxic chlorinating agents such as phosphorus oxychloride or thionyl chloride, which significantly reduces environmental pollution and safety hazards.
Q: How does this process address impurity control in Gefitinib manufacturing?
A: By bypassing the traditional chlorination step of the quinazoline ring which often generates difficult-to-remove chlorinated by-products, this route utilizes a formamidine cyclization strategy. This approach inherently limits the formation of halogenated impurities associated with POCl3 usage, leading to a cleaner crude product profile and simplifying the downstream purification process.
Q: Is this synthetic method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrialization. It operates under mild temperatures (typically between 80°C and 120°C for key steps) and uses common solvents like toluene, acetic acid, and DMF. The absence of extreme conditions or hazardous reagents makes it highly scalable and safer for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefitinib Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a new synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical setting. We operate under stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Gefitinib intermediate meets the highest international standards. Our commitment to quality assurance means that we can navigate the complexities of impurity profiling and process validation with speed and precision, accelerating your time to market.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this greener, more efficient pathway. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of high-quality intermediates into your supply chain.
