Scalable Electroreduction Technology for High-Purity Gefitinib Intermediates and Commercial量产
Scalable Electroreduction Technology for High-Purity Gefitinib Intermediates and Commercial量产
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing processes, particularly in the synthesis of complex oncology intermediates. A pivotal development in this arena is detailed in patent CN110747489B, which discloses a novel electroreduction preparation method for 2-amino-4-methoxybenzoic acid derivatives. These compounds serve as critical building blocks for the synthesis of Gefitinib, a potent Epidermal Growth Factor Receptor (EGFR) tyrosine kinase inhibitor used in the treatment of non-small cell lung cancer and breast cancer. The patented technology replaces traditional, pollution-heavy chemical reduction methods with a clean electrochemical approach, utilizing electrons as the primary reducing agent. This innovation not only addresses stringent environmental regulations but also offers a pathway to higher purity profiles essential for modern API manufacturing. By leveraging a diaphragm electrolytic cell system, the process achieves exceptional conversion rates while minimizing the formation of hazardous byproducts, positioning it as a superior alternative for reliable pharmaceutical intermediate suppliers seeking to optimize their production lines.
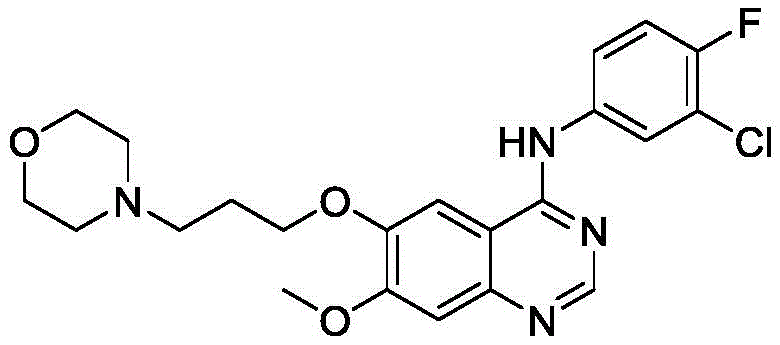
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of nitro groups in benzoic acid derivatives has relied heavily on stoichiometric chemical reducing agents such as sodium hydrosulfite, iron powder with acetic acid, or catalytic hydrogenation using precious metals like palladium. While effective to a degree, these methods suffer from significant drawbacks that impact both cost and quality. For instance, the use of iron powder generates massive amounts of iron sludge, creating a severe environmental burden and complicating waste disposal protocols. Furthermore, metal residues from iron or tin dichloride reductions can persist in the product, necessitating rigorous and costly purification steps to meet the strict heavy metal limits required for anticancer drugs. Catalytic hydrogenation, although cleaner, involves the use of expensive palladium catalysts which can form difficult-to-separate complexes with amino intermediates, potentially compromising the purity of the final product and posing safety risks due to high-pressure operations.
The Novel Approach
The electroreduction method described in the patent fundamentally alters the reduction landscape by employing electricity to drive the conversion of 4-methoxy-2-nitrobenzoic acid derivatives into their corresponding amino forms. This approach eliminates the need for external chemical reducing agents, thereby removing the source of inorganic waste and metal contamination. The process operates under mild conditions, typically between 20°C and 80°C, and utilizes a divided cell setup to prevent re-oxidation of the product at the anode. Experimental data from the patent highlights the superiority of this method; for example, the electroreduction of methyl 4-methoxy-5-(3-chloropropoxy)-2-nitrobenzoate yielded 93.8% of the desired amine, a marked improvement over the 77% yield obtained via traditional iron reduction. This increase in efficiency, coupled with the inherent safety of avoiding high-pressure hydrogenation, makes the electroreduction route a compelling choice for cost reduction in API manufacturing.
Mechanistic Insights into Cathodic Nitro Reduction
Understanding the mechanistic pathway of this electroreduction is crucial for R&D directors aiming to replicate and scale the process. The reaction proceeds through a multi-step electron transfer mechanism at the cathode surface. Initially, the nitro group accepts electrons and protons to form a nitroso intermediate, which is further reduced to a hydroxylamine species before finally arriving at the stable amine product. The patent specifies that controlling the cathode working electrode voltage between 1.00V and 2.50V relative to a reference electrode is critical. This potential window ensures that the reduction proceeds selectively to the amine without over-reduction or the formation of condensation byproducts such as azo or hydrazo compounds, which are common pitfalls in uncontrolled chemical reductions.
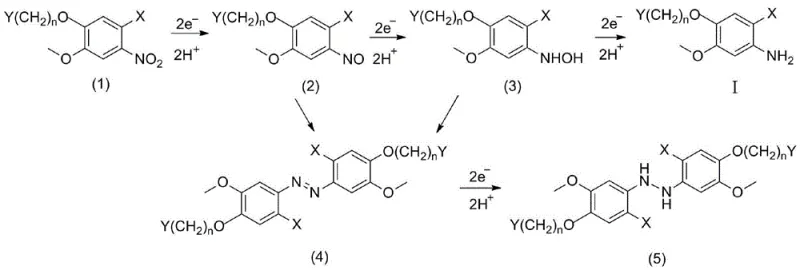
Impurity control is another significant advantage of this electrochemical method. In traditional chemical reductions, side reactions often lead to complex impurity profiles that are difficult to characterize and remove. The electroreduction process, however, allows for precise tuning of the reaction parameters, such as current density (optimized between 20.0 mA/cm² and 250.0 mA/cm²) and electrolyte composition. By maintaining these parameters within the specified ranges, the selectivity for the target 2-amino-4-methoxybenzoic acid derivative is maximized. The use of a diaphragm cell further protects the product by physically separating the anodic and cathodic compartments, preventing the oxidation of the newly formed amine at the anode. This results in a crude product of exceptionally high purity, significantly reducing the burden on downstream purification processes and ensuring a robust impurity profile for regulatory submissions.
How to Synthesize 2-Amino-4-methoxybenzoic Acid Derivatives Efficiently
The synthesis of these valuable intermediates involves setting up a diaphragm electrolytic cell with specific electrode materials, such as a copper cathode and a Dimensionally Stable Anode (DSA). The catholyte is prepared by dissolving the nitro precursor in an acidic medium or a mixture containing inorganic ammonium salts and organic solvents to ensure adequate conductivity and solubility. The reaction is driven by a constant current or voltage source, with temperature control maintained via a water bath. Detailed operational parameters, including electrolyte concentrations and specific workup procedures involving alkalization and solvent extraction, are critical for achieving the reported high yields. For a comprehensive, step-by-step technical guide on executing this synthesis in a pilot or production environment, please refer to the standardized protocol below.
- Prepare the catholyte by dissolving the 4-methoxy-2-nitrobenzoic acid derivative in an acidic solution or a mixture of inorganic ammonium salt, organic solvent, and water within a diaphragm electrolytic cell.
- Utilize an acidic aqueous solution as the anolyte and apply a constant current density between 20.0 mA/cm² and 250.0 mA/cm², maintaining the cathode working electrode voltage between 1.00V and 2.50V relative to the reference electrode.
- Maintain the electrolysis temperature between 20°C and 80°C until completion, then isolate the 2-amino-4-methoxybenzoic acid derivative through alkalization, extraction, and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electroreduction technology translates into tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for stoichiometric amounts of reducing agents like sodium hydrosulfite or iron powder, manufacturers reduce their dependency on volatile commodity chemical markets and mitigate the risks associated with the storage and handling of hazardous substances. This streamlining of inputs leads to a more resilient supply chain, less susceptible to disruptions caused by the availability of specific reagents. Furthermore, the reduction in waste generation directly correlates to lower disposal costs and reduced environmental compliance burdens, which are increasingly significant factors in the total cost of ownership for chemical manufacturing.
- Cost Reduction in Manufacturing: The transition to electroreduction offers significant economic advantages by removing the cost of purchasing and disposing of chemical reducing agents. Traditional methods often require large excesses of reagents to drive reactions to completion, generating substantial waste that incurs disposal fees. In contrast, the electrochemical method uses electricity, which is generally cheaper and more consistent in price than specialty chemicals. Additionally, the higher yields observed (e.g., increasing from 77% to over 93% in specific examples) mean that less starting material is wasted, directly improving the atom economy and lowering the cost per kilogram of the final intermediate. The elimination of expensive palladium catalysts further contributes to substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: Relying on electricity as a reagent decouples the production process from the supply chains of specific chemical reducers, which can be subject to geopolitical or logistical fluctuations. The equipment required for electroreduction, such as diaphragm cells and power supplies, is durable and widely available, ensuring long-term operational continuity. Moreover, the simplified workup procedure—often requiring only neutralization and extraction rather than complex filtration of metal sludges—reduces the turnaround time for each batch. This efficiency allows for faster response to market demand and shorter lead times for high-purity pharmaceutical intermediates, providing a competitive edge in a fast-paced industry.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on industrial emissions and waste, the green credentials of this process become a major asset. The absence of heavy metal waste and toxic byproducts facilitates easier permitting and compliance with environmental standards. The process is inherently scalable; increasing production capacity primarily involves scaling the electrode surface area and cell volume, a well-understood engineering challenge. This scalability ensures that the method can grow from laboratory benchtop to multi-ton commercial production without fundamental changes to the chemistry, supporting the commercial scale-up of complex pharmaceutical intermediates with minimal risk.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electroreduction technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this method into existing production workflows. The answers highlight the balance between technical performance and operational practicality.
Q: How does electroreduction improve the purity of Gefitinib intermediates compared to traditional iron powder reduction?
A: Traditional iron powder reduction often generates significant sludge and metal impurities that are difficult to remove, potentially affecting the heavy metal specifications of the final API. Electroreduction uses electrons as the reducing agent, eliminating metal contamination entirely and allowing for precise control over reaction selectivity via electrode potential, resulting in significantly higher purity intermediates suitable for oncology drugs.
Q: What are the specific yield improvements observed in the electroreduction of nitrobenzoates?
A: According to patent data, electroreduction of methyl 4-methoxy-5-(3-chloropropoxy)-2-nitrobenzoate achieved a yield of 93.8%, whereas the conventional iron/acetic acid method yielded only 77%. Similarly, morpholine-substituted intermediates showed yields up to 98.3% via electroreduction, demonstrating a substantial efficiency gain.
Q: Is this electroreduction process scalable for industrial API production?
A: Yes, the process is designed for scalability. It operates at moderate temperatures (20°C-80°C) and utilizes standard diaphragm electrolytic cells with durable electrodes like DSA anodes and copper cathodes. The elimination of hazardous chemical reducing agents simplifies waste treatment and enhances safety, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-methoxybenzoic Acid Derivative Supplier
The electroreduction technology outlined in patent CN110747489B represents a significant leap forward in the synthesis of key oncology intermediates, offering a blend of high efficiency, environmental sustainability, and cost-effectiveness. At NINGBO INNO PHARMCHEM, we recognize the transformative potential of such advanced manufacturing techniques. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required for global pharmaceutical markets. We are committed to delivering high-purity pharmaceutical intermediates that adhere to the highest quality standards, ensuring that your supply chain remains robust and compliant.
We invite you to explore how our expertise in electroorganic synthesis can optimize your production of Gefitinib intermediates. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to bring this innovative, green chemistry solution to your commercial manufacturing line, ensuring a reliable supply of critical materials for life-saving therapies.
