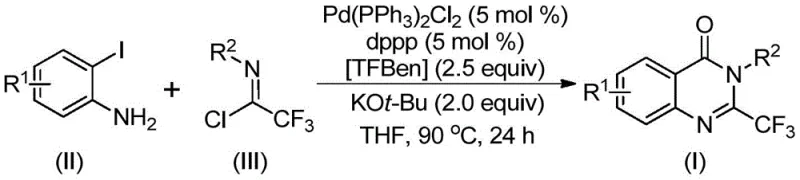
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access privileged scaffolds like quinazolinones, which are ubiquitous in bioactive molecules ranging from anticonvulsants to antitumor agents. Patent CN112125856A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone derivatives, addressing critical bottlenecks in traditional synthetic routes. This innovation leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing readily available o-iodoaniline and trifluoroacetimidoyl chloride as starting materials. A pivotal advancement in this protocol is the substitution of hazardous carbon monoxide gas with 1,3,5-tricarboxylate phenol ester (TFBen) as a solid CO surrogate. This strategic shift not only mitigates severe safety risks associated with toxic gas handling but also streamlines the operational complexity, making the process highly attractive for industrial adoption. For R&D directors and procurement managers alike, this technology represents a significant leap forward in accessing high-purity pharmaceutical intermediates with improved metabolic stability and lipophilicity profiles inherent to trifluoromethyl groups.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinones has been plagued by substantial technical and economic hurdles that hinder efficient commercial scale-up. Conventional strategies often rely on the cyclization of anthranilamide with ethyl trifluoroacetate or trifluoroacetic anhydride, which frequently necessitates harsh reaction conditions and expensive, pre-activated substrates. Alternative methods involving T3P-promoted tandem reactions or the use of unstable trifluoroacetamides suffer from narrow substrate scopes and inconsistent yields, leading to significant batch-to-batch variability. Furthermore, traditional carbonylation approaches typically require the direct use of carbon monoxide gas, a colorless and odorless toxin that demands rigorous safety protocols, specialized high-pressure reactors, and extensive engineering controls. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, creating friction in the supply chain for critical active pharmaceutical ingredient (API) intermediates. The reliance on such cumbersome methodologies limits the ability of manufacturers to rapidly iterate on molecular designs or respond to market demand fluctuations.
The Novel Approach
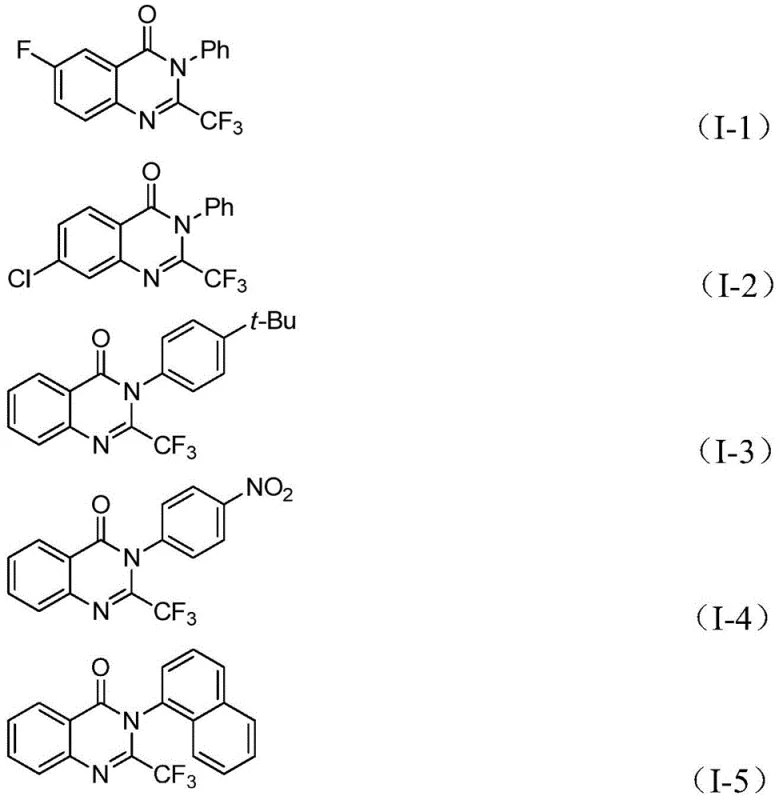
In stark contrast, the novel methodology described in the patent offers a streamlined, safe, and highly efficient pathway to these valuable heterocycles. By employing a palladium-catalyzed system with a solid carbon monoxide surrogate, the process eliminates the need for gaseous CO infrastructure entirely. The reaction operates under relatively mild thermal conditions at 90°C in common organic solvents like tetrahydrofuran (THF), utilizing inexpensive and commercially abundant starting materials. This approach demonstrates exceptional functional group tolerance, accommodating a wide array of substituents including halogens, alkyls, and nitro groups without compromising yield. The operational simplicity allows for a straightforward workup procedure involving filtration and standard column chromatography, significantly reducing downstream processing time. For a reliable pharmaceutical intermediate supplier, this translates to a more agile manufacturing capability that can support diverse medicinal chemistry campaigns while maintaining stringent quality standards and cost-effectiveness.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The mechanistic elegance of this transformation lies in its multi-step tandem sequence orchestrated by the palladium catalyst. The reaction is hypothesized to initiate with a base-promoted intermolecular carbon-nitrogen bond coupling between the o-iodoaniline and the trifluoroacetimidoyl chloride, generating a trifluoroacetamidine intermediate in situ. Subsequently, the palladium catalyst undergoes oxidative addition into the carbon-iodine bond of the aromatic ring, forming a reactive divalent palladium species. Under the applied thermal conditions, the solid surrogate TFBen decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to create an acyl-palladium intermediate. This key step effectively builds the carbonyl functionality of the quinazolinone core without external gas feed. The presence of the base further facilitates the formation of a palladium-nitrogen bond, closing the seven-membered ring palladium intermediate before final reductive elimination releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active catalyst. This intricate cycle ensures high atom economy and minimizes the formation of unwanted byproducts.
From an impurity control perspective, the use of a solid CO source provides a controlled, steady release of carbon monoxide, preventing local concentration spikes that could lead to over-carbonylation or polymerization side reactions. The mild reaction temperature of 90°C further suppresses thermal degradation of sensitive functional groups, ensuring a cleaner crude reaction profile. This is particularly crucial for complex substrates containing electron-withdrawing groups like nitro or halogen substituents, which might be susceptible to reduction or dehalogenation under more vigorous conditions. The robustness of the catalytic system, utilizing Pd(PPh3)2Cl2 and dppp ligand, ensures consistent turnover numbers across different substrate classes. For quality assurance teams, this mechanistic stability implies a predictable impurity spectrum that is easier to characterize and purge during purification, ultimately delivering high-purity intermediates suitable for subsequent GMP manufacturing steps.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The implementation of this synthesis route is designed for practicality and scalability in a standard laboratory or pilot plant setting. The protocol requires precise stoichiometric control of the palladium catalyst, ligand, and base to maximize conversion while minimizing metal residue in the final product. The use of THF as the preferred solvent ensures optimal solubility for all reactants and intermediates, facilitating homogeneous reaction kinetics. Operators should monitor the reaction progress carefully within the 16 to 30-hour window to ensure complete consumption of the starting o-iodoaniline, as indicated by the patent data showing high yields under these specific temporal conditions. The detailed standardized synthesis steps below outline the exact procedural requirements for replicating this high-efficiency transformation.
- Combine palladium catalyst (Pd(PPh3)2Cl2), ligand (dppp), base (KOt-Bu), solid CO source (TFBen), trifluoroacetimidoyl chloride, and o-iodoaniline in an organic solvent like THF.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to allow the carbonylation and cyclization to proceed to completion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the pure 2-trifluoromethyl-substituted quinazolinone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of toxic carbon monoxide gas removes a major regulatory and safety burden, drastically simplifying facility requirements and insurance costs associated with hazardous material handling. This shift allows for production in a wider range of manufacturing sites without the need for specialized high-pressure gas infrastructure, thereby enhancing supply chain flexibility and geographic diversification. Furthermore, the reliance on cheap, commodity-grade starting materials like o-iodoaniline derivatives and trifluoroacetimidoyl chlorides ensures a stable and cost-effective raw material supply base that is less susceptible to market volatility compared to exotic reagents. The simplified post-treatment process reduces solvent consumption and waste generation, aligning with modern green chemistry principles and lowering environmental compliance costs.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of expensive and hazardous reagents with affordable alternatives. By avoiding the use of high-pressure carbon monoxide equipment and specialized safety containment systems, capital expenditure (CAPEX) for new production lines is significantly reduced. Additionally, the low catalyst loading of 5 mol% combined with the use of inexpensive ligands minimizes the cost contribution of precious metals to the overall bill of materials. The high conversion rates and clean reaction profiles reduce the burden on purification steps, leading to lower solvent usage and higher overall throughput. These factors collectively contribute to a substantial reduction in the cost of goods sold, making the final API intermediates more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available commercial starting materials that do not rely on single-source suppliers or complex logistics chains. The solid nature of the carbon monoxide surrogate (TFBen) simplifies storage and transportation, eliminating the risks and delays associated with the delivery of compressed gases. This stability ensures that production schedules can be maintained consistently without interruptions due to raw material shortages or safety inspections related to hazardous gas storage. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility supplies, further securing the reliability of delivery timelines for downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or tonne scale is straightforward due to the absence of gas-liquid mass transfer limitations typically seen in carbonylation reactions. The homogeneous nature of the reaction in THF allows for efficient heat and mass transfer in large reactors, ensuring consistent product quality across batches. From an environmental standpoint, the avoidance of toxic gas emissions and the use of a solid surrogate significantly reduce the facility's environmental footprint. Waste streams are easier to treat as they do not contain residual dissolved toxic gases, simplifying effluent management and ensuring compliance with increasingly stringent environmental regulations. This makes the process not only scalable but also sustainable for long-term commercial operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing manufacturing workflows. The answers provide clarity on safety, scope, and operational parameters to assist decision-makers in assessing the technology's fit for their specific applications.
Q: Why is TFBen used instead of carbon monoxide gas in this synthesis?
A: TFBen (1,3,5-tricarboxylate phenol ester) serves as a solid, safe surrogate for toxic carbon monoxide gas. This eliminates the need for high-pressure gas cylinders and specialized containment equipment, significantly enhancing operational safety and simplifying the regulatory compliance for manufacturing facilities.
Q: What is the substrate scope for this quinazolinone synthesis method?
A: The method demonstrates excellent compatibility with various substituents. It tolerates electron-withdrawing and electron-donating groups on both the o-iodoaniline and the imidoyl chloride components, including halogens (F, Cl, Br), alkyl groups, nitro groups, and naphthyl systems, allowing for diverse library synthesis.
Q: What are the typical reaction conditions and catalyst loading?
A: The reaction typically proceeds at 90°C in THF solvent for 16 to 30 hours. The catalyst system uses Pd(PPh3)2Cl2 and dppp ligand at a low loading of approximately 5 mol% relative to the substrate, making it economically viable for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to drive drug discovery and development forward. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into reliable commercial supply. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality and safety makes us an ideal partner for navigating the complexities of modern API manufacturing.
We invite you to collaborate with us to leverage this innovative palladium-catalyzed carbonylation technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and accelerate your time to market.
