Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
Introduction to Novel Quinazolinone Manufacturing Technologies
The pharmaceutical industry constantly seeks robust synthetic routes for privileged scaffolds, and the recent disclosure in patent CN113045503B presents a transformative approach to synthesizing 2-trifluoromethyl substituted quinazolinone compounds. These heterocyclic structures are critical motifs found in numerous bioactive molecules, including well-known agents such as Methaqualone and the natural product Rutaecarpine, which exhibit antifungal, antiviral, and anticancer properties. The introduction of a trifluoromethyl group into these frameworks significantly enhances physicochemical properties such as metabolic stability, lipophilicity, and bioavailability, making them highly desirable for modern drug discovery programs. This patent outlines a sophisticated yet operationally simple palladium-catalyzed carbonylation cascade reaction that overcomes the historical limitations of quinazolinone synthesis, offering a pathway that is both economically viable and chemically efficient for large-scale production.
Traditional methods for accessing these valuable intermediates have often been plagued by significant drawbacks, including the requirement for harsh reaction conditions, the use of unstable or expensive reagents like trifluoroacetic anhydride, and generally low substrate compatibility. In contrast, the methodology described in CN113045503B utilizes cheap and readily available starting materials, specifically trifluoroethylimidoyl chloride and various amines, to drive the reaction forward with high efficiency. The process is designed to be scalable, having been successfully demonstrated at the gram level, which provides a strong foundation for industrial application. For procurement and supply chain leaders, this represents a shift towards more reliable sourcing of high-purity pharmaceutical intermediates, as the simplified workflow reduces the risk of batch failures and supply disruptions common with more finicky legacy chemistries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 2-trifluoromethyl-substituted quinazolinones was largely constrained by a lack of versatile and efficient methodologies. Literature reports typically describe routes involving the cyclization of anthranilamide with ethyl trifluoroacetate or trifluoroacetic anhydride, which often necessitate rigorous exclusion of moisture and elevated temperatures that can degrade sensitive functional groups. Another common approach involves the reaction of anthranilates with unstable trifluoroacetamides, a strategy that suffers from poor atom economy and the generation of difficult-to-remove byproducts. Furthermore, methods utilizing isatoic anhydride or T3P-promoted cascade reactions are frequently limited by narrow substrate scopes, meaning that introducing diverse substituents on the aromatic ring often leads to drastic drops in yield or complete reaction failure. These inefficiencies translate directly into higher manufacturing costs and longer lead times for active pharmaceutical ingredient (API) production.
The Novel Approach
The breakthrough detailed in the patent data introduces a transition metal palladium-catalyzed carbonylation cascade that fundamentally changes the economic and technical landscape of quinazolinone synthesis. By employing trifluoroethylimidoyl chloride as a key building block, the reaction bypasses the need for pre-activated carboxylic acid derivatives, streamlining the synthetic sequence. The use of TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a safe and effective solid source of carbon monoxide, eliminating the safety hazards and specialized equipment required for handling gaseous CO. This novel approach not only tolerates a wide range of functional groups—including halogens, alkyls, and trifluoromethyl groups—but also delivers consistently high yields across diverse substrates. For a reliable pharmaceutical intermediate supplier, adopting this technology means offering clients a more cost-effective route to complex heterocycles without compromising on purity or structural integrity.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this reaction is crucial for R&D directors aiming to optimize the process for specific API candidates. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond coupling between the amine and the imidoyl chloride, generating a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst, specifically palladium trifluoroacetate coordinated with triphenylphosphine, undergoes oxidative addition into the carbon-iodine bond of the aromatic ring to form a divalent palladium intermediate. This step is critical for activating the aryl ring for subsequent cyclization. As the reaction proceeds at 110°C, the TFBen additive thermally decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form an acyl-palladium species. This insertion is the key carbonylation step that builds the quinazolinone core.
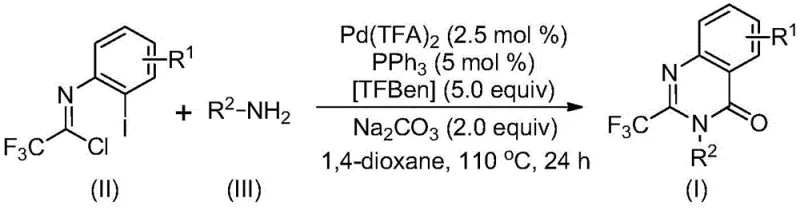
Following the CO insertion, the presence of a base, such as sodium carbonate, facilitates the formation of a palladium-nitrogen bond, closing the ring to generate a seven-membered palladacycle intermediate. The final step involves reductive elimination, which releases the desired 2-trifluoromethyl-substituted quinazolinone product and regenerates the active palladium catalyst for the next cycle. This elegant catalytic cycle ensures high turnover numbers and minimizes the accumulation of palladium residues in the final product, a critical factor for meeting stringent regulatory limits on heavy metals in pharmaceuticals. The robustness of this mechanism allows for the synthesis of various derivatives, as evidenced by the successful preparation of compounds I-1 through I-5 with different R1 and R2 substituents, demonstrating the versatility required for modern medicinal chemistry campaigns.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to specific parameters regarding reagent stoichiometry and reaction conditions to maximize yield and purity. The protocol requires the precise combination of palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, the specific trifluoroethylimidoyl chloride, and the chosen amine in an aprotic organic solvent such as 1,4-dioxane. Maintaining the reaction temperature at 110°C for a duration of 16 to 30 hours is essential to drive the carbonylation cascade to completion while ensuring full conversion of the starting materials. Detailed standardized operating procedures for this synthesis, including workup and purification steps, are provided below to ensure reproducibility and safety.
- Combine palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, trifluoroethylimidoyl chloride, and amine in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 110°C and stir continuously for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl substituted quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediate manufacturing achieved through the use of commodity-grade starting materials. Unlike legacy routes that depend on specialized, moisture-sensitive reagents, this process utilizes amines and imidoyl chlorides that are widely available in the global chemical market, ensuring a stable and continuous supply chain even during periods of raw material volatility. The elimination of hazardous gaseous carbon monoxide in favor of a solid CO surrogate further reduces the capital expenditure required for specialized reactor infrastructure, making the technology accessible for both small-scale development and large-scale commercial production.
- Cost Reduction in Manufacturing: The economic efficiency of this process is driven by the high atom economy and the avoidance of expensive coupling reagents or protecting group strategies often seen in alternative syntheses. By utilizing a catalytic amount of palladium and inexpensive ligands, the overall cost of goods sold (COGS) for the final quinazolinone intermediate is significantly lowered. Additionally, the simplified post-treatment process, which involves basic filtration and standard column chromatography, reduces labor hours and solvent consumption, contributing to a leaner manufacturing footprint and improved profit margins for downstream API producers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions translates directly into improved supply chain reliability. Because the method tolerates a broad spectrum of functional groups and does not require ultra-dry or cryogenic conditions, the risk of batch rejection due to minor environmental fluctuations is minimized. This reliability allows suppliers to offer shorter lead times for high-purity pharmaceutical intermediates, as the production schedule is less susceptible to the delays often caused by the need for re-running failed batches. The ability to source diverse amines easily further diversifies the supply base, mitigating the risk of single-source dependency.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with green chemistry principles. The use of 1,4-dioxane as a solvent is well-established in industrial settings, and the reaction generates minimal hazardous waste compared to stoichiometric oxidation or chlorination methods. The process has already been validated at the gram scale with excellent results, indicating a clear path for commercial scale-up of complex pharmaceutical intermediates to the multi-kilogram or tonne level. This scalability ensures that as drug candidates move from clinical trials to commercial launch, the manufacturing process can expand seamlessly without requiring a complete process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on reaction scope, substrate compatibility, and practical application in drug synthesis. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing pipelines.
Q: What are the primary advantages of this palladium-catalyzed method over traditional quinazolinone synthesis?
A: Unlike conventional methods that rely on harsh conditions, expensive pre-activated substrates like isatoic anhydride, or low-yielding cascade reactions, this novel approach utilizes cheap and readily available trifluoroethylimidoyl chlorides. It offers superior functional group tolerance, higher reaction efficiency, and eliminates the need for difficult post-treatment steps associated with older protocols.
Q: Can this synthetic route be applied to complex drug molecules like Rutaecarpine?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the high-yield synthesis of Rutaecarpine. The process involves a three-step sequence achieving a total yield of 77%, proving its viability for constructing complex fused-ring nitrogen-containing heterocycles found in bioactive natural products.
Q: What represents the key cost-saving factor in this manufacturing process?
A: The significant cost reduction stems from the use of commercially available and inexpensive starting materials, specifically various types of amines and trifluoroethylimidoyl chlorides. Furthermore, the use of TFBen as a solid carbon monoxide substitute simplifies the operational setup compared to handling gaseous CO, thereby reducing equipment and safety costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 2-trifluoromethyl quinazolinones and related intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the palladium-catalyzed carbonylation technology described in CN113045503B allows us to offer competitive pricing and consistent quality for this valuable scaffold.
We invite pharmaceutical partners to collaborate with us to leverage these technological advancements for their specific projects. Whether you require custom synthesis of novel analogs or bulk supply of established intermediates, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for efficiency, cost, and reliability in the competitive global marketplace.
