Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for High-Value Pharmaceutical Intermediates
Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for High-Value Pharmaceutical Intermediates
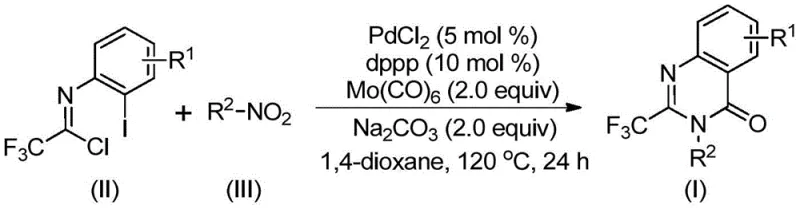
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, safer, and cost-effective synthetic routes. A groundbreaking development in this sector is detailed in patent CN112480015A, which discloses a highly efficient method for synthesizing 2-trifluoromethyl substituted quinazolinone compounds via a multicomponent one-pot method. Quinazolinones are a privileged scaffold in medicinal chemistry, found in numerous bioactive molecules exhibiting antifungal, antibacterial, antiviral, and anticancer properties. The introduction of a trifluoromethyl group further enhances these molecules by improving metabolic stability, lipophilicity, and bioavailability. This patent presents a transformative approach that leverages transition metal palladium catalysis to construct these complex heterocycles directly from inexpensive nitro compounds and trifluoroethylimidoyl chloride, bypassing the limitations of traditional multi-step syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has been fraught with synthetic challenges that hinder large-scale production. Conventional methods often rely on the condensation of anthranilic acid derivatives with various reagents, or the cyclization of nitro-substituted benzamides. A significant drawback of many prior art methods is the reliance on harsh reaction conditions, such as high-pressure carbon monoxide atmospheres, which necessitate specialized and expensive reactor equipment, posing substantial safety risks in an industrial setting. Furthermore, traditional routes frequently require pre-activated substrates, such as 2-bromoformanilides or specific anhydrides, which are not only costly to procure but also generate significant stoichiometric waste. Other methods utilizing ruthenium or platinum catalysts often suffer from narrow substrate scopes and moderate yields, making them economically unviable for the mass production of high-purity pharmaceutical intermediates required by global supply chains.
The Novel Approach
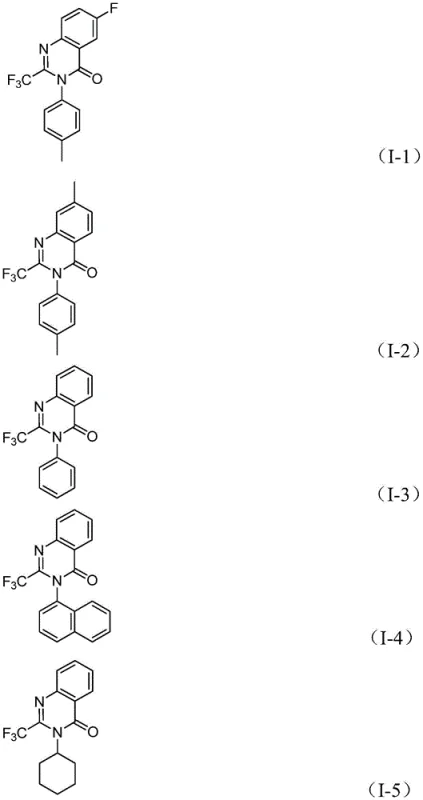
In stark contrast to these cumbersome legacy processes, the method described in CN112480015A offers a streamlined, atom-economical solution. By employing a palladium-catalyzed carbonylation serial reaction, this novel approach utilizes cheap and readily available nitro compounds as the nitrogen source, effectively combining reduction and cyclization into a single operational step. The use of molybdenum hexacarbonyl (Mo(CO)6) as a solid carbon monoxide surrogate is a critical innovation, eliminating the need for handling toxic CO gas while ensuring a steady supply of the carbonyl source within the reaction matrix. This one-pot strategy not only simplifies the workflow by removing intermediate isolation steps but also demonstrates exceptional substrate compatibility, tolerating a wide range of functional groups including halogens, alkyls, and trifluoromethyl groups. The result is a robust protocol capable of delivering high yields, often exceeding 90%, which is a substantial improvement over the variable efficiencies of older techniques.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
For R&D directors focused on process optimization, understanding the mechanistic underpinnings of this transformation is vital for troubleshooting and scaling. The reaction initiates with the reduction of the nitro group on the aromatic ring to an amine species, facilitated by the molybdenum hexacarbonyl under thermal conditions. This generated amine then undergoes a base-promoted coupling with the trifluoroethylimidoyl chloride to form a trifluoroacetamidine derivative intermediate. Subsequently, the palladium catalyst, likely generated in situ from PdCl2 and the dppp ligand, inserts into the carbon-iodine bond of the imidoyl chloride moiety (or a related intermediate), forming a reactive divalent palladium species. As the temperature is maintained at 120°C, the Mo(CO)6 decomposes to release carbon monoxide, which inserts into the carbon-palladium bond to create an acyl palladium intermediate. This key step constructs the carbonyl functionality essential for the quinazolinone ring. Finally, intramolecular nucleophilic attack by the nitrogen atom, promoted by the base, closes the ring to form a seven-membered palladium intermediate, which undergoes reductive elimination to release the final 2-trifluoromethyl-substituted quinazolinone product and regenerate the active catalyst.
This intricate cascade is meticulously controlled to minimize impurity formation, a critical factor for meeting stringent pharmaceutical purity specifications. The choice of 1,3-bis(diphenylphosphino)propane (dppp) as the ligand is particularly strategic, as it stabilizes the palladium center and facilitates the specific geometry required for the cyclization step, thereby suppressing side reactions such as homocoupling or incomplete carbonylation. The use of sodium carbonate as a mild base ensures that the reaction environment remains conducive to the cyclization without degrading sensitive functional groups on the substrate. This high level of chemoselectivity means that the crude reaction mixture contains fewer byproducts, significantly reducing the burden on downstream purification processes like column chromatography or recrystallization, which translates directly to higher overall process efficiency and lower production costs.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it an ideal candidate for technology transfer from lab to pilot plant. The procedure involves charging a reaction vessel with the palladium catalyst system, the solid CO source, and the organic substrates in a suitable solvent such as 1,4-dioxane. The mixture is then heated to reflux conditions for a defined period, typically between 16 to 30 hours, allowing the multicomponent reaction to reach completion. Following the reaction, a simple workup involving filtration to remove metal residues and inorganic salts, followed by standard purification techniques, yields the target compound in high purity. For a detailed, step-by-step breakdown of the exact molar ratios, specific temperature profiles, and purification protocols validated in the patent examples, please refer to the standardized guide below.
- Mix palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and the nitro compound in an organic solvent like dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow for nitro reduction, coupling, and cyclization.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final 2-trifluoromethyl substituted quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers compelling economic and logistical benefits that address common pain points in the sourcing of complex heterocyclic intermediates. The shift towards using nitro compounds as starting materials is particularly advantageous, as these commodities are produced on a massive industrial scale for other sectors, ensuring a stable and abundant supply chain that is less susceptible to the volatility often seen with specialized fine chemical building blocks. Moreover, the elimination of high-pressure gas handling equipment reduces the capital expenditure (CAPEX) required for manufacturing facilities, allowing for production in standard glass-lined or stainless steel reactors that are already prevalent in most contract manufacturing organizations. This flexibility enhances supply chain resilience by enabling production across a wider network of qualified vendors without the need for specialized infrastructure upgrades.
- Cost Reduction in Manufacturing: The economic impact of this method is driven primarily by the drastic simplification of the synthetic sequence. By consolidating multiple chemical transformations into a single one-pot operation, manufacturers can significantly reduce labor costs, energy consumption, and solvent usage associated with intermediate isolation and purification steps. The use of inexpensive nitroarenes and the avoidance of precious metal catalysts like ruthenium or platinum further drive down the raw material bill of materials (BOM). Additionally, the high yields reported across various substrates mean that less starting material is wasted, maximizing the output per batch and effectively lowering the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on exotic or hard-to-source reagents. This protocol mitigates such risks by utilizing commercially available, off-the-shelf reagents such as palladium chloride, dppp, and molybdenum hexacarbonyl, all of which are standard inventory items for major chemical suppliers. The robustness of the reaction conditions, which tolerate a broad spectrum of functional groups, means that the same process can be applied to synthesize a library of analogues without needing to re-optimize conditions for each new derivative. This versatility allows suppliers to respond rapidly to changing demand patterns for different drug candidates, ensuring that lead times for high-purity pharmaceutical intermediates remain short and predictable.
- Scalability and Environmental Compliance: As regulatory pressures regarding waste disposal and environmental impact intensify, the green chemistry attributes of this method become increasingly valuable. The one-pot nature of the reaction inherently reduces the volume of waste solvents and aqueous streams generated compared to multi-step linear syntheses. Furthermore, the use of a solid CO surrogate eliminates the risk of carbon monoxide leaks, a critical safety and environmental hazard. The process is demonstrated to be scalable to the gram level and beyond, suggesting that it can be smoothly transitioned to multi-kilogram or ton-scale production with minimal engineering challenges, facilitating the commercial scale-up of complex pharmaceutical intermediates while maintaining strict adherence to environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope, safety profile, and scalability. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable foundation for process assessment. Understanding these nuances is essential for making informed decisions about integrating this methodology into existing production pipelines or new drug development programs.
Q: What are the primary advantages of using nitro compounds over pre-activated amines in this synthesis?
A: Nitro compounds are significantly cheaper and more readily available than their amine counterparts. Furthermore, this method eliminates the need for separate reduction steps or harsh reducing agents, streamlining the process into a single pot.
Q: How does the use of Mo(CO)6 improve safety compared to traditional carbonylation methods?
A: Traditional methods often require high-pressure carbon monoxide gas, which poses severe safety risks. Mo(CO)6 acts as a solid CO surrogate, releasing carbon monoxide in situ under heating, thereby avoiding the need for specialized high-pressure equipment and enhancing operational safety.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly states that the method can be expanded to the gram level and is suitable for large-scale application. The use of common solvents like dioxane and commercially available catalysts supports easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
The synthesis of 2-trifluoromethyl substituted quinazolinones represents a critical capability for the development of next-generation therapeutics, and mastering this chemistry requires a partner with deep technical expertise and proven manufacturing capacity. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced catalytic methodologies to deliver high-quality intermediates that meet the rigorous demands of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage clinical trials through to full-scale commercial launch. We operate state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone we supply is free from critical impurities and fully compliant with international regulatory standards.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. By leveraging our expertise in palladium-catalyzed carbonylation and multicomponent reactions, we can help you achieve significant process efficiencies and cost savings. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data, route feasibility assessments, and competitive pricing models to demonstrate how our advanced manufacturing capabilities can become a strategic asset to your organization.
