Advanced Manufacturing of Sulfurized Imidazole Derivatives for Cardiovascular APIs
Advanced Manufacturing of Sulfurized Imidazole Derivatives for Cardiovascular APIs
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical cardiovascular intermediates, particularly those targeting the Angiotensin II receptor. Patent CN1097419A presents a groundbreaking methodology for the preparation of sulfurized derivatives of imidazoles, specifically designed to serve as potent intermediates for antihypertensive agents. This technology addresses the growing demand for high-purity pharmaceutical intermediates by offering a versatile synthetic pathway that accommodates various substituents at the 2, 4, and 5 positions of the imidazole ring. For global procurement teams and R&D directors, understanding the nuances of this patent is essential for securing a reliable supply chain for next-generation cardiovascular therapeutics. The disclosed method not only enhances structural diversity but also improves the overall efficiency of producing complex biphenyl-imidazole scaffolds.
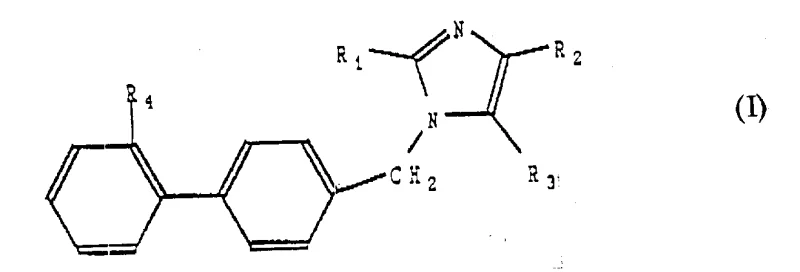
The core innovation lies in the flexibility of the substituent groups, allowing for the precise tuning of pharmacological properties. By manipulating the R groups, manufacturers can access a wide library of analogues, including those with specific sulfur-containing functionalities like alkylthio, alkylsulfinyl, and alkylsulfonyl groups. This structural versatility is paramount for optimizing bioavailability and metabolic stability in final drug products. Furthermore, the patent ensures that all possible racemic, enantiomeric, and diastereomeric forms are covered, providing comprehensive intellectual property protection for derivative works. As a leading entity in fine chemical manufacturing, leveraging this proprietary knowledge allows for the development of superior generic alternatives and novel drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for imidazole-based Angiotensin II antagonists often suffer from significant drawbacks regarding regioselectivity and functional group tolerance. Conventional methods frequently rely on harsh cyclization conditions that can degrade sensitive biphenyl moieties or require expensive transition metal catalysts that introduce difficult-to-remove impurities. Additionally, the introduction of sulfur functionalities at specific positions on the imidazole ring has historically been challenging, often resulting in low yields and complex purification protocols. These inefficiencies translate directly into higher production costs and extended lead times, creating bottlenecks for supply chain managers aiming to meet market demand. The reliance on multi-step protection and deprotection strategies in older methodologies further exacerbates waste generation and reduces the overall atom economy of the process.
The Novel Approach
In contrast, the novel approach detailed in CN1097419A utilizes a streamlined sequence that builds the imidazole ring through a highly efficient thioimidate intermediate. This strategy bypasses many of the regiochemical issues associated with direct alkylation of pre-formed imidazoles. By constructing the heterocycle from a linear precursor containing a nitrile and an amide functionality, the process ensures precise placement of the sulfur substituent. The use of mild oxidants for the initial transformation of biphenyl halides to aldehydes preserves the integrity of the sensitive sulfonamide linkage. This method significantly simplifies the downstream processing, as the cyclization step proceeds cleanly under acid catalysis or with dehydrating agents like phosphorus pentachloride. Consequently, this represents a substantial advancement in cost reduction in pharmaceutical intermediate manufacturing by minimizing unit operations and maximizing yield.
Mechanistic Insights into Thioimidate-Mediated Cyclization
The mechanistic elegance of this process centers on the formation and subsequent cyclization of a thioimidate species. Initially, a biphenyl sulfonamide derivative bearing a halomethyl group is oxidized to the corresponding aldehyde. This aldehyde then undergoes reductive amination with an alpha-amino nitrile ester, establishing the carbon-nitrogen backbone required for the imidazole ring. Following N-acylation to protect the secondary amine and introduce the side chain, the critical thioimidate formation occurs. This involves the nucleophilic attack of a thiol or thiolate species onto the nitrile carbon, generating an imidoyl sulfide intermediate. This step is crucial as it installs the sulfur functionality at the 4-position of the future imidazole ring with high fidelity.
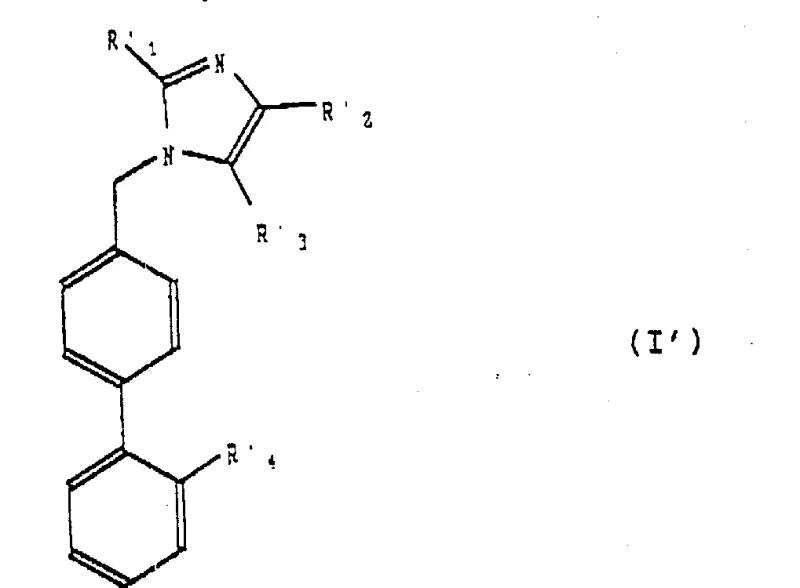
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement strategies and supply chain resilience. The reliance on commercially available starting materials, such as simple biphenyl derivatives and standard acyl chlorides, mitigates the risk of raw material shortages. Unlike processes dependent on scarce noble metal catalysts, this method utilizes abundant reagents, ensuring long-term supply continuity. The simplified purification profile, resulting from fewer side reactions and cleaner cyclization, reduces the consumption of chromatography media and solvents. This directly correlates to significant cost reduction in API manufacturing, as downstream processing often accounts for a major portion of total production expenses. By minimizing waste generation and solvent usage, the process also aligns with increasingly strict environmental regulations, avoiding potential compliance penalties.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of standard organic reagents drastically lower the bill of materials. Furthermore, the high yield of the cyclization step reduces the amount of starting material required per kilogram of final product. The ability to crystallize intermediates rather than relying solely on chromatographic purification further drives down operational expenditures. These factors combine to create a highly competitive cost structure, allowing for better margin management in volatile markets.
- Enhanced Supply Chain Reliability: The synthetic route is robust against minor fluctuations in reaction conditions, ensuring consistent output quality. Since the reagents are commodity chemicals, sourcing is not limited to single-supplier bottlenecks. This diversification of the supply base enhances reliability and reduces lead time for high-purity pharmaceutical intermediates. Manufacturers can maintain leaner inventory levels without risking production stoppages, improving overall working capital efficiency.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to kilogram scales in the patent examples. The reaction conditions do not require extreme pressures or cryogenic temperatures, making them suitable for standard glass-lined steel reactors found in most multipurpose plants. Additionally, the reduced solvent intensity and avoidance of heavy metals simplify wastewater treatment, facilitating easier environmental permitting and sustainable operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within CN1097419A, providing clarity for technical decision-makers. Understanding these aspects helps in evaluating the feasibility of integrating this route into existing manufacturing portfolios. For more specific technical data, our team is prepared to provide detailed route feasibility assessments.
Q: What are the key advantages of this sulfurized imidazole synthesis route?
A: The process described in CN1097419A utilizes readily available starting materials and avoids complex transition metal catalysis, ensuring a more robust and scalable manufacturing process suitable for commercial production.
Q: Can this method produce optically active isomers?
A: Yes, while the primary synthesis yields racemic mixtures, the patent explicitly covers subsequent resolution steps to obtain optically active isomers, which are critical for specific biological activities in cardiovascular therapeutics.
Q: Is this process suitable for large-scale API intermediate production?
A: Absolutely. The reaction conditions described, such as room temperature acylations and standard reflux cyclizations, are highly amenable to scale-up from kilogram to multi-ton annual production capacities without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial reactor. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize advanced analytical techniques to verify the identity and purity of every batch. We understand the critical nature of cardiovascular intermediates and the zero-tolerance policy for impurities in this therapeutic area. Our engineering team is adept at optimizing the cyclization and purification steps described in this patent to maximize throughput while maintaining the highest quality standards.
We invite you to contact our technical procurement team to discuss your specific requirements and request a Customized Cost-Saving Analysis for your project. By partnering with us, you gain access to specific COA data and route feasibility assessments tailored to your volume needs. Let us help you secure a stable and cost-effective supply of these critical sulfurized imidazole derivatives, empowering your drug development pipeline with reliable chemistry.
