Scalable Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates for Pharma
Scalable Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates for Pharma
The rapid evolution of medicinal chemistry demands increasingly complex molecular scaffolds, particularly those containing biheterocyclic motifs which are prevalent in bioactive natural products and modern pharmaceuticals. A recent technological breakthrough, documented in patent CN115353511A, introduces a highly efficient, multi-component strategy for constructing carbonyl-bridged biheterocyclic compounds. This methodology specifically targets the synthesis of indolinone-imidazole fused systems, a structural class known for its broad-spectrum biological activity and significance in drug discovery pipelines. By leveraging a transition metal palladium-catalyzed cascade reaction, this innovation addresses critical bottlenecks in traditional heterocycle synthesis, offering a pathway that is not only chemically robust but also operationally streamlined for industrial application. The ability to construct multiple chemical bonds in a single pot represents a paradigm shift towards atom-economical and sustainable manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic frameworks has relied on laborious multi-step sequences that often suffer from poor atom economy and harsh reaction conditions. Traditional approaches typically involve the direct coupling of two pre-formed heterocyclic substrates, which frequently requires expensive activating agents and results in low overall yields due to cumulative losses at each step. Alternatively, oxidative cyclization strategies utilizing activated methyl-substituted heterocycles often necessitate strong oxidants and high temperatures, leading to significant safety hazards and the generation of toxic waste streams. Furthermore, conventional carbonylation reactions to introduce the crucial carbonyl bridge have traditionally depended on the use of pressurized carbon monoxide gas, posing severe safety risks and requiring specialized high-pressure equipment that limits scalability in standard pharmaceutical manufacturing facilities.
The Novel Approach
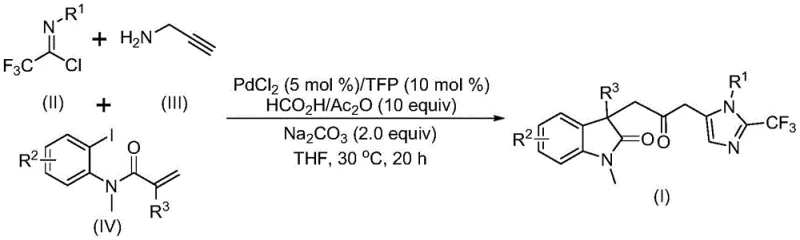
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a sophisticated palladium-catalyzed multicomponent reaction that seamlessly integrates three distinct building blocks: trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives. This one-pot protocol eliminates the need for toxic carbon monoxide gas by employing a safe formic acid and acetic anhydride mixture as an in situ CO source, thereby drastically simplifying the reactor setup and enhancing workplace safety. The reaction operates under remarkably mild conditions, typically at 30°C, which preserves sensitive functional groups and reduces energy consumption significantly. As illustrated in the general reaction scheme below, this cascade process efficiently constructs the complex carbonyl-bridged core with high regioselectivity and excellent yields, representing a substantial advancement in synthetic efficiency.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic elegance of this transformation lies in its intricate catalytic cycle, which orchestrates multiple bond-forming events with precision. The reaction is initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This species subsequently undergoes an intramolecular Heck-type cyclization to form a divalent alkyl-palladium complex, setting the stage for the crucial carbonylation step. Unlike traditional methods requiring external CO tanks, the carbon monoxide required for this step is released gently from the decomposition of the formic acid and acetic anhydride additive mixture. This in situ generated CO inserts into the palladium-carbon bond to form an acyl-palladium intermediate, which is the key precursor for the final ring closure.
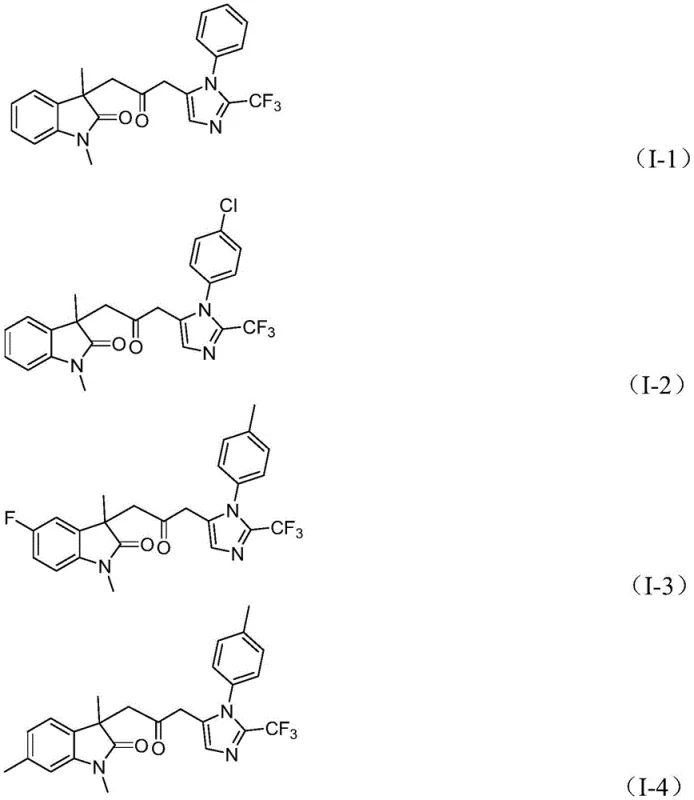
Concurrently, the trifluoroethylimidoyl chloride and propargylamine undergo a base-promoted intermolecular reaction to form a trifluoroacetamidine intermediate, which subsequently isomerizes to a more reactive form. The acyl-palladium species then activates this amidine compound, catalyzing an intramolecular cyclization that forge the final imidazole ring and completes the construction of the carbonyl-bridged biheterocyclic architecture. The versatility of this mechanism is evidenced by the broad substrate scope tolerated by the catalytic system, as shown in the specific examples below, where various substituents including halogens, alkyl groups, and electron-withdrawing moieties are accommodated without inhibiting the catalytic turnover. This robustness ensures that the process can be adapted to synthesize a diverse library of analogues for structure-activity relationship studies.
How to Synthesize Carbonyl-Bridged Biheterocycles Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The protocol is designed to be user-friendly, utilizing commercially available starting materials such as propargylamine and acryloyl chloride derivatives, which ensures a reliable supply chain for raw materials. The reaction is typically conducted in tetrahydrofuran (THF), an aprotic solvent that effectively dissolves all components and promotes the catalytic cycle, although other solvents like acetonitrile may also be viable depending on specific substrate solubility profiles. For detailed procedural instructions regarding stoichiometry, addition rates, and workup procedures, please refer to the standardized synthesis guide provided below.
- Combine palladium chloride, TFP ligand, sodium carbonate, and the formic acid/acetic anhydride CO source in THF solvent.
- Add the trifluoroethylimidoyl chloride, propargylamine, and substituted acrylamide substrates to the reaction mixture.
- Stir the reaction at 30°C for 12 to 20 hours, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers compelling advantages that directly impact the bottom line and supply chain resilience. By shifting from multi-step linear syntheses to a convergent multicomponent reaction, manufacturers can significantly reduce the number of unit operations required, leading to lower labor costs and reduced equipment occupancy time. The elimination of hazardous carbon monoxide gas removes the need for specialized high-pressure reactors and complex gas handling infrastructure, allowing production to occur in standard glass-lined or stainless steel vessels commonly found in multipurpose pharmaceutical plants. This flexibility enhances asset utilization and allows for faster campaign turnover, enabling suppliers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of inexpensive, commodity-grade starting materials such as propargylamine and simple acrylamides, which are readily available in bulk quantities from global chemical suppliers. The catalytic system employs palladium chloride, a relatively cost-effective precious metal catalyst compared to more exotic alternatives, and operates at low loadings, minimizing the financial burden of metal recovery and recycling. Furthermore, the mild reaction temperature of 30°C translates to substantial energy savings compared to high-temperature reflux conditions, reducing utility costs associated with heating and cooling cycles throughout the production batch.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on stable, shelf-stable reagents that do not require cryogenic storage or special transportation permits associated with toxic gases. The robustness of the reaction against moisture and air, facilitated by the specific ligand and additive choices, reduces the risk of batch failures due to minor environmental deviations during scale-up. This reliability ensures consistent delivery schedules for downstream customers, mitigating the risk of production delays that can plague more sensitive synthetic routes involving unstable intermediates or pyrophoric reagents.
- Scalability and Environmental Compliance: The process has been validated at the gram scale with high efficiency, indicating a clear path towards kilogram and tonne-scale commercial production without fundamental changes to the chemistry. The use of formic acid as a CO surrogate generates benign byproducts like carbon dioxide and water, aligning with green chemistry principles and simplifying waste treatment protocols. This environmental compatibility reduces the regulatory burden associated with hazardous waste disposal and supports corporate sustainability goals, making the technology attractive for companies aiming to minimize their ecological footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and embodiments described in the patent literature, providing a factual basis for process evaluation. Understanding these nuances is critical for process chemists and engineers tasked with translating this laboratory-scale innovation into a robust commercial manufacturing process.
Q: What is the primary safety advantage of this carbonylation method?
A: Unlike traditional carbonylation reactions that require toxic carbon monoxide gas cylinders, this method utilizes a formic acid and acetic anhydride mixture to generate CO in situ, significantly enhancing operational safety and reducing regulatory burdens.
Q: Does this process tolerate diverse functional groups on the substrate?
A: Yes, the patent demonstrates excellent substrate compatibility, successfully accommodating various substituents such as halogens, alkyl groups, alkoxy groups, and nitro groups on the aromatic rings without compromising reaction efficiency.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction proceeds under mild conditions, typically requiring a temperature of 30°C in THF solvent for 12 to 20 hours, using a low loading of palladium chloride catalyst and trifurylphosphine ligand.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN115353511A for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to marketplace is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of carbonyl-bridged biheterocyclic intermediates meets the highest standards required for pharmaceutical applications. We are committed to delivering quality and consistency, leveraging our technical expertise to optimize yields and minimize impurities.
We invite you to collaborate with us to explore how this innovative synthesis route can enhance your project economics and timeline. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data for related intermediates and to discuss comprehensive route feasibility assessments for your next generation of therapeutic candidates. Let us be your partner in turning complex chemical challenges into commercial successes.
