Advanced Manufacturing of Long-Acting GLP-1 Analogues: Microwave-Assisted Solid-Phase Synthesis and Coumarin Modification
Advanced Manufacturing of Long-Acting GLP-1 Analogues: Microwave-Assisted Solid-Phase Synthesis and Coumarin Modification
The global landscape of diabetes treatment is undergoing a significant transformation, driven by the urgent need for therapies that offer improved patient compliance and sustained glycemic control without the risk of hypoglycemia. Natural Glucagon-like Peptide-1 (GLP-1) has emerged as a potent therapeutic candidate due to its glucose-dependent insulin secretion properties; however, its clinical utility is severely hampered by a fleeting biological half-life of approximately five minutes, primarily caused by rapid degradation via dipeptidyl peptidase IV (DPP-IV) and swift renal clearance. Addressing these critical pharmacokinetic limitations, Patent CN102180963B introduces a groundbreaking approach involving the chemical modification of GLP-1 analogues with 4-hydroxycoumarin derivatives. This innovation not only enhances the stability of the peptide against enzymatic hydrolysis but also leverages microwave-promoted solid-phase peptide synthesis (SPPS) to achieve unprecedented efficiency in manufacturing. For pharmaceutical developers and supply chain strategists, this technology represents a pivotal shift towards more viable, long-acting anti-diabetic agents that can be produced with greater speed and economic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solid-phase peptide synthesis (SPPS) has long been the industry standard for producing peptide therapeutics, yet it is plagued by inherent inefficiencies that escalate costs and extend lead times, particularly for complex sequences like the 31-amino acid GLP-1. In conventional protocols, the coupling of each amino acid residue typically requires extensive reaction times ranging from two to twenty hours to ensure complete conversion, while the removal of the Fmoc protecting group often necessitates thirty minutes to an hour of incubation. These prolonged durations not only bottleneck production capacity but also increase the likelihood of side reactions such as racemization and aggregation, which compromise the purity of the final product. Furthermore, conventional methods often demand a large excess of protected amino acids, typically four to five equivalents, to drive the reaction to completion, resulting in significant material waste and inflated raw material costs. The cumulative effect of these factors is a synthesis process that is labor-intensive, time-consuming, and economically burdensome for large-scale commercialization.
The Novel Approach
The methodology outlined in Patent CN102180963B revolutionizes this landscape by integrating microwave irradiation into the SPPS workflow, thereby accelerating reaction kinetics and enhancing overall process efficiency. By utilizing microwave energy, the coupling time for each amino acid is drastically reduced to an average of merely ten minutes, and the deprotection step is condensed to approximately five minutes, representing a magnitude of improvement in throughput. This rapid synthesis capability is complemented by a strategic reduction in reagent consumption, where the required excess of protected amino acids is lowered to just two equivalents, significantly minimizing waste generation. Moreover, the implementation of this microwave-assisted technique yields a crude peptide product with a purity exceeding 60%, a marked enhancement over traditional methods, which simplifies subsequent purification steps and improves the final recovery yield. This novel approach effectively transforms the manufacturing paradigm from a slow, resource-heavy operation into a streamlined, high-efficiency process suitable for industrial automation.
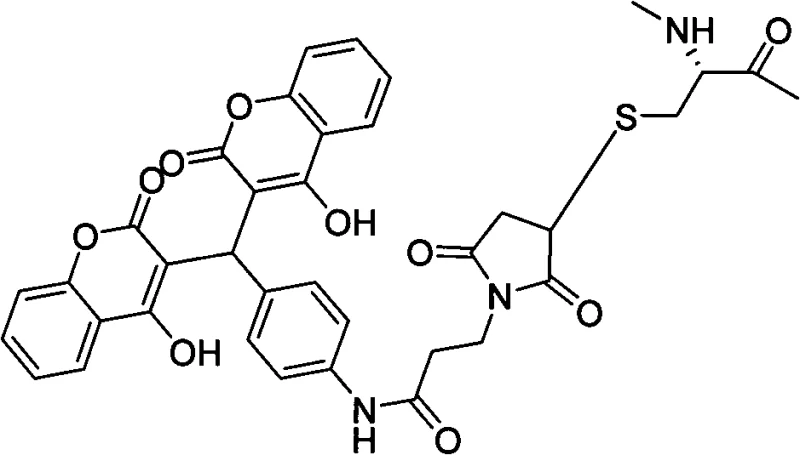
Mechanistic Insights into Coumarin-Mediated Peptide Stabilization
The core innovation of this technology lies in the precise chemical modification of the GLP-1 peptide chain using a 4-hydroxycoumarin analog, specifically designed to interact with cysteine residues introduced at key positions within the sequence. The synthesis involves reacting the thiol group of a chemically modified cysteine residue with a maleimide-functionalized 4-hydroxycoumarin derivative, forming a stable thioether linkage. This covalent attachment serves a dual purpose: firstly, the bulky and hydrophobic nature of the coumarin moiety sterically hinders the access of DPP-IV enzymes to the N-terminal cleavage site, thereby conferring resistance to enzymatic degradation. Secondly, the 4-hydroxycoumarin scaffold possesses a high affinity for serum albumin, a abundant plasma protein. Upon administration, the modified GLP-1 analogue binds reversibly to serum albumin, creating a circulating reservoir of the drug that is protected from glomerular filtration in the kidneys. This protein-binding mechanism effectively extends the biological half-life of the peptide, allowing for a sustained release of the active drug over an extended period, which is crucial for achieving long-acting therapeutic effects with less frequent dosing intervals.
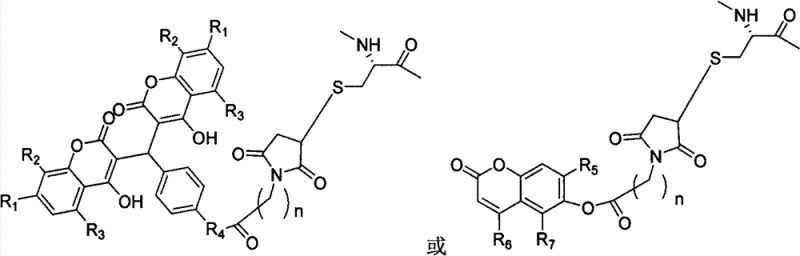
Furthermore, the structural integrity of the peptide is meticulously preserved through the use of orthogonal protection strategies, such as the Fmoc/tBu scheme, which ensures that side-chain functional groups remain inert during the chain elongation process. The microwave irradiation not only accelerates the formation of peptide bonds but also helps in overcoming solvation issues that often lead to incomplete couplings in difficult sequences. By maintaining a controlled temperature of 50°C during the microwave cycles, the process avoids thermal degradation while ensuring sufficient energy for the activation of carboxyl groups by coupling reagents like HBTU or HATU. The result is a highly pure intermediate that retains the specific conformational requirements necessary for binding to the GLP-1 receptor (GLP-1R) on pancreatic beta cells. This precise engineering ensures that while the pharmacokinetic profile is enhanced for longevity, the pharmacodynamic activity—specifically the glucose-dependent stimulation of insulin secretion—remains potent and effective, avoiding the hypoglycemic risks associated with non-glucose-dependent insulin secretagogues.
How to Synthesize Long-Acting GLP-1 Analogues Efficiently
The synthesis protocol described in the patent offers a robust framework for producing these advanced therapeutics, beginning with the swelling of Fmoc-Rink amide-MBHA resin and proceeding through iterative cycles of microwave-assisted deprotection and coupling. The process culminates in the cleavage of the peptide from the resin and a final conjugation step with the coumarin derivative in a Tris buffer system. This standardized approach ensures reproducibility and scalability, making it an ideal candidate for technology transfer to commercial manufacturing facilities.
- Swelling of Fmoc-Rink amide-MBHA Resin in DCM and NMP, followed by microwave-assisted removal of the Fmoc protecting group using piperidine/NMP solution.
- Sequential coupling of protected amino acids using HBTU/HOBt/DIPEA activation under microwave irradiation (50°C, 25W) to rapidly extend the peptide chain.
- Cleavage of the peptide from the resin using Reagent K, followed by reaction with 3,3'-(4-(3-dimaleoyl imino propionamido) phenylmethylene)-bis-4-hydroxy coumarin to form the final long-acting analogue.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microwave-promoted synthesis technology presents a compelling value proposition centered on cost optimization and operational agility. The drastic reduction in synthesis cycle time directly translates to increased equipment utilization rates, allowing manufacturers to produce larger batches within the same timeframe without requiring additional capital investment in reactor hardware. This efficiency gain is further amplified by the significant reduction in the consumption of expensive protected amino acids and coupling reagents, which constitute a major portion of the variable costs in peptide manufacturing. By lowering the stoichiometric excess required for coupling reactions, the process minimizes raw material waste and reduces the environmental burden associated with solvent disposal and hazardous waste treatment. These factors collectively contribute to a leaner, more sustainable production model that aligns with modern green chemistry principles while delivering substantial cost savings.
- Cost Reduction in Manufacturing: The transition to microwave-assisted synthesis eliminates the need for prolonged reaction times and excessive reagent usage, which are primary cost drivers in traditional peptide production. By reducing the amino acid excess from four-to-five equivalents down to two equivalents, the direct material costs are significantly lowered. Additionally, the higher crude purity achieved (>60%) reduces the load on downstream purification processes such as preparative HPLC, leading to lower solvent consumption and higher final yields. This holistic reduction in resource intensity ensures a more competitive cost structure for the final API, enabling better pricing strategies in the global market.
- Enhanced Supply Chain Reliability: The speed and efficiency of the microwave synthesis method enhance the responsiveness of the supply chain to market demands. Shorter production cycles mean that lead times for custom peptide synthesis can be drastically compressed, allowing for quicker turnaround on pilot batches and faster scale-up to commercial volumes. The robustness of the process, characterized by high coupling efficiencies and minimal side reactions, ensures consistent batch-to-batch quality, reducing the risk of production failures or delays due to out-of-specification results. This reliability is critical for maintaining a steady supply of essential diabetes medications, mitigating the risks of stockouts and ensuring continuity of care for patients.
- Scalability and Environmental Compliance: The methodology is inherently designed for automation and large-scale production, addressing the common bottleneck of scaling peptide synthesis from laboratory to industrial levels. The reduced solvent usage and minimized waste generation simplify compliance with increasingly stringent environmental regulations regarding hazardous waste disposal. The ability to automate the microwave reactors further reduces the reliance on manual labor, decreasing the potential for human error and enhancing operator safety. These attributes make the process highly scalable, capable of meeting the growing global demand for GLP-1 analogues without compromising on environmental stewardship or operational safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these long-acting GLP-1 analogues, providing clarity for stakeholders evaluating this technology for their development pipelines.
Q: How does the 4-hydroxycoumarin modification improve GLP-1 stability?
A: The 4-hydroxycoumarin moiety significantly increases the binding rate to serum proteins, specifically serum albumin. This binding creates a balance between bound and free drug in vivo, allowing for slow release and preventing rapid renal filtration. Additionally, the modification protects the peptide from DPP-IV enzymatic degradation, extending the biological half-life from minutes to hours.
Q: What are the efficiency gains of using microwave promotion in SPPS compared to conventional methods?
A: Microwave-promoted synthesis drastically reduces reaction times. While conventional coupling often requires 2 to 20 hours per amino acid, the microwave method averages only about 10 minutes. Similarly, Fmoc deprotection is reduced from 30-60 minutes to approximately 5 minutes, significantly shortening the overall synthesis cycle and improving throughput.
Q: Does this synthesis method impact the purity of the crude peptide?
A: Yes, the microwave-promoted solid-phase synthesis yields a crude peptide purity greater than 60%, which is a substantial improvement over conventional solid-phase processes. This higher initial purity facilitates downstream purification work, reducing the burden on preparative HPLC and increasing overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GLP-1 Analogue Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in Patent CN102180963B and are fully equipped to bring these advanced long-acting GLP-1 analogues from concept to commercial reality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project benefits from our state-of-the-art microwave synthesis capabilities and rigorous QC labs. Our commitment to excellence is reflected in our ability to meet stringent purity specifications, delivering high-quality peptide intermediates and APIs that adhere to the highest international regulatory standards. We understand the complexities of peptide chemistry and are dedicated to optimizing every step of the synthesis process to maximize yield and minimize impurities.
We invite you to collaborate with us to leverage these cutting-edge synthesis strategies for your next-generation diabetes therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in microwave-promoted solid-phase synthesis can accelerate your development timeline and enhance your competitive advantage in the global pharmaceutical market.
