Scalable Manufacturing of High-Purity Nicotinamide Riboside via Mild Deacetylation Technology
Scalable Manufacturing of High-Purity Nicotinamide Riboside via Mild Deacetylation Technology
The global demand for Nicotinamide Riboside (NR), a critical precursor for the essential coenzyme NAD+, has surged due to its profound implications in cellular metabolism and anti-aging research. As industries seek reliable nicotinamide riboside suppliers capable of delivering pharmaceutical-grade intermediates, the focus shifts to synthesis methods that balance efficiency, safety, and environmental compliance. Patent CN112028954B introduces a groundbreaking preparation method that addresses the longstanding limitations of conventional NR synthesis. This technology utilizes a mild deacetylation strategy involving nicotinamide triacetyl nucleoside and inorganic strong base weak acid salts in a methanol solvent system. Unlike traditional routes that rely on hazardous gaseous reagents, this approach operates under controlled low-temperature conditions ranging from -20°C to 30°C, ensuring high product yield and exceptional purity. The structural integrity of the final molecule, as depicted below, is preserved through gentle reaction conditions that prevent degradation of the sensitive glycosidic bond.
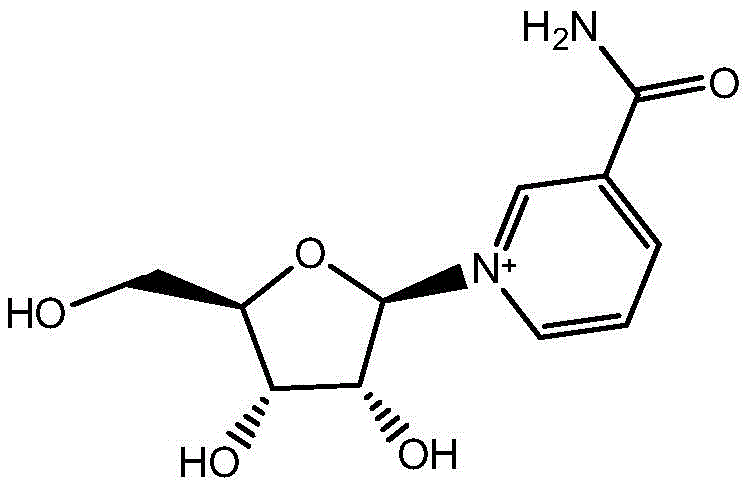
This patent represents a significant leap forward in the commercial scale-up of complex pharmaceutical intermediates, offering a pathway that is not only chemically robust but also economically viable for mass production. By eliminating the need for toxic raw materials and simplifying the purification workflow, the process aligns perfectly with modern green chemistry principles. For R&D directors and procurement managers alike, understanding the nuances of this synthetic route is essential for securing a stable supply chain of high-value NAD+ boosters. The following analysis dissects the technical advantages, mechanistic insights, and commercial implications of adopting this novel methodology over legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Nicotinamide Riboside has been plagued by significant operational hazards and process inefficiencies, primarily stemming from the reliance on ammonia gas for the deprotection of acetyl groups. In comparative studies referenced within the patent data, the introduction of ammonia into the reaction system presents severe safety challenges, including the risk of leaks from pressurized cylinders and the potential for dangerous suck-back phenomena during gas flow regulation. Furthermore, the stoichiometry of gaseous ammonia is notoriously difficult to control with precision, often leading to incomplete reactions or the formation of unwanted byproducts due to fluctuating concentrations. This lack of controllability directly impacts the reproducibility of the batch, resulting in variable yields that can destabilize supply chains for downstream manufacturers. Additionally, the handling of ammonia requires specialized infrastructure and rigorous safety protocols, which inherently drives up the capital expenditure and operational costs associated with the manufacturing facility.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112028954B revolutionizes the deacetylation step by substituting hazardous ammonia gas with solid inorganic strong base weak acid salts, such as sodium carbonate, potassium carbonate, or trisodium phosphate. This substitution fundamentally alters the reaction dynamics, allowing for precise stoichiometric control and eliminating the safety risks associated with high-pressure gas handling. The reaction proceeds smoothly in methanol at mild temperatures, typically between -10°C and 0°C, which minimizes thermal stress on the sensitive nucleoside structure. This novel approach not only enhances the safety profile of the manufacturing process but also significantly simplifies the operational workflow, as solid reagents are easier to weigh, handle, and dose accurately compared to gaseous counterparts. The result is a robust, reproducible process that delivers consistent high yields, with experimental data indicating yields reaching up to 80%, thereby offering a compelling solution for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Inorganic Salt-Mediated Deacetylation
The core chemical transformation in this patent involves the selective removal of acetyl protecting groups from the ribose sugar moiety of nicotinamide triacetyl nucleoside. The mechanism relies on the nucleophilic attack facilitated by the basic environment created by the inorganic salts in the methanol solvent. When salts like trisodium phosphate are introduced, they generate a mildly alkaline medium that promotes the hydrolysis or methanolysis of the ester bonds without compromising the stability of the N-glycosidic linkage connecting the sugar to the nicotinamide base. Comparative Example 1 in the patent data critically demonstrates the necessity of these basic conditions; in the absence of anhydrous sodium carbonate, the reaction fails to proceed even after extended periods of 30 hours, yielding zero product. This confirms that the base is not merely a catalyst but an essential reactant that drives the equilibrium towards the deprotected Nicotinamide Riboside. The choice of a "strong base weak acid salt" is particularly ingenious, as it provides sufficient basicity to cleave the acetyl groups while avoiding the harsh conditions of strong hydroxides that could degrade the vitamin B3 derivative.
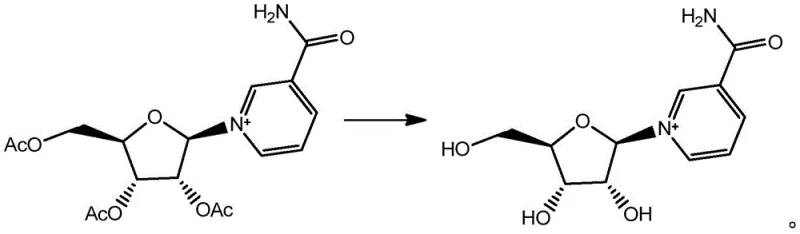
Furthermore, the purification mechanism is elegantly designed to leverage the solubility differences between the product and impurities. Following the reaction, the mixture is neutralized with hydrochloric acid, converting excess base into soluble salts that can be easily filtered off. The subsequent concentration and crystallization steps utilize a water-alcohol solvent system, which is highly effective at excluding organic impurities and residual starting materials. The patent highlights that this recrystallization process is so efficient that it obviates the need for column chromatography, a technique that is often costly, time-consuming, and difficult to scale in an industrial setting. By achieving purity levels greater than 99% through simple crystallization, the process ensures that the final high-purity nicotinamide riboside meets stringent quality specifications required for human consumption and clinical applications, all while maintaining a streamlined production timeline.
How to Synthesize Nicotinamide Riboside Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent ratios to maximize yield and purity. The process begins with the dissolution of the protected nucleoside in methanol, followed by cooling to sub-zero temperatures to manage the exothermic nature of the deacetylation. The addition of the inorganic base must be performed under an inert atmosphere, such as nitrogen or argon, to prevent moisture uptake and oxidation. Detailed standard operating procedures for this transformation, including specific molar ratios and crystallization parameters, are outlined in the patent examples to ensure reproducibility. For technical teams looking to adopt this method, the following guide summarizes the critical operational steps derived from the most successful embodiments of the invention.
- Dissolve nicotinamide triacetyl nucleoside in methanol and cool the solution to a temperature range of -20°C to -10°C under a protective nitrogen atmosphere.
- Add an inorganic strong base weak acid salt, such as trisodium phosphate or sodium carbonate, and maintain the reaction temperature between -10°C and 0°C for 6 to 20 hours.
- Neutralize the reaction mixture with hydrochloric acid, filter to remove salts, concentrate the filtrate, and perform recrystallization using water and ethanol to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this salt-mediated synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of ammonia gas cylinders removes a major logistical and safety burden, reducing the regulatory compliance overhead and insurance costs associated with storing hazardous compressed gases. This shift significantly enhances the overall safety culture of the manufacturing site, minimizing the risk of workplace accidents and ensuring uninterrupted production schedules. Moreover, the reliance on commodity chemicals like methanol, sodium carbonate, and hydrochloric acid ensures that raw material sourcing is stable and resistant to market volatility, unlike specialized catalysts or reagents that may face supply bottlenecks. These factors collectively contribute to a more resilient supply chain capable of meeting the growing global demand for NAD+ precursors without compromising on delivery reliability or product quality.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the downstream processing workflow. By removing the requirement for column chromatography, manufacturers save substantial amounts on silica gel, solvents, and labor hours typically associated with purification. The high yield of up to 80% means less starting material is wasted, directly improving the atom economy and reducing the cost per kilogram of the final API intermediate. Additionally, the use of inexpensive inorganic salts instead of costly organic bases or enzymes further lowers the direct material costs. These cumulative savings allow for a more competitive pricing structure in the marketplace, providing a distinct advantage for companies aiming to capture market share in the nutraceutical and pharmaceutical sectors.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a highly reliable production schedule. Since the reaction tolerates a reasonable range of temperatures (-20°C to 30°C) and times (6-30 hours), minor fluctuations in process parameters do not lead to batch failures, ensuring consistent output. The simplicity of the workup, involving basic filtration and crystallization, reduces the turnaround time between batches, allowing for higher throughput in existing reactor vessels. This operational flexibility ensures that suppliers can respond quickly to surges in demand, reducing lead times for high-purity nicotinamide riboside and strengthening partnerships with downstream formulators who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with increasingly strict global regulations regarding waste disposal and emissions. The process generates no toxic byproducts, and the solvent system (methanol/ethanol/water) is relatively benign and easy to recover and recycle. The absence of heavy metal catalysts or chlorinated solvents simplifies the wastewater treatment process, lowering the environmental compliance costs for the manufacturer. This "green" profile is not only ethically sound but also commercially advantageous, as it appeals to eco-conscious consumers and corporate buyers who prioritize sustainability in their supply chain audits. The ease of scaling from gram to ton quantities makes this technology ideal for meeting the massive volume requirements of the global anti-aging and metabolic health markets.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled a set of frequently asked questions based on the specific technical disclosures and experimental data found in patent CN112028954B. These answers address common concerns regarding process safety, product quality, and scalability, providing a clear picture of what manufacturers can expect when implementing this synthesis route. Understanding these details is crucial for conducting a thorough feasibility assessment and for communicating the value proposition of this method to internal stakeholders and external partners.
Q: Why is the inorganic salt method superior to the traditional ammonia gas method for NR synthesis?
A: The traditional method utilizing ammonia gas introduces significant safety hazards due to the handling of pressurized cylinders and potential suck-back issues. Furthermore, controlling the airflow of ammonia is difficult, leading to inconsistent reaction rates. The patented method replaces gaseous ammonia with solid inorganic salts like trisodium phosphate, which eliminates these safety risks and provides precise stoichiometric control, resulting in higher reproducibility and yields up to 80%.
Q: What purity levels can be achieved with this deacetylation process?
A: This synthesis route is designed to achieve exceptional purity without the need for complex purification techniques like column chromatography. Through a optimized recrystallization process involving water and alcohol, the final Nicotinamide Riboside product consistently achieves purity levels exceeding 99% as verified by HPLC analysis, making it suitable for high-grade pharmaceutical and nutraceutical applications.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its reliance on common, inexpensive reagents like methanol and sodium carbonate rather than specialized catalysts or hazardous gases. The post-treatment involves simple filtration and crystallization steps, which are easily adaptable to industrial reactors. The elimination of toxic byproducts and the use of mild temperatures (-20°C to 30°C) further enhance its viability for green, large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicotinamide Riboside Supplier
The technological advancements detailed in patent CN112028954B underscore the immense potential of Nicotinamide Riboside as a cornerstone molecule in the future of metabolic health and longevity therapeutics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative laboratory methods into robust, industrial-scale realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of Nicotinamide Riboside we produce, adhering to the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your product pipeline. Our team of expert chemists is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient route can optimize your bottom line. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing and delivering high-quality NAD+ precursors to the global market.
