Revolutionizing Tomoxetine Production: High-Efficiency Chemo-Enzymatic Synthesis for Global Supply Chains
Revolutionizing Tomoxetine Production: High-Efficiency Chemo-Enzymatic Synthesis for Global Supply Chains
The pharmaceutical industry is constantly seeking more efficient, sustainable, and cost-effective pathways for the production of active pharmaceutical ingredients (APIs) and their key intermediates. A significant breakthrough in this domain is detailed in patent CN112708641A, which outlines a novel chemo-enzymatic synthesis method for tomoxetine, a widely prescribed non-central excitatory therapeutic for attention deficit hyperactivity disorder (ADHD). This patent introduces a highly selective carbonyl reductase capable of catalyzing the asymmetric reduction of 3-chloropropiophenone to (S)-3-chlorophenylpropanol with exceptional stereoselectivity. By achieving an enantiomeric excess (ee) value of greater than 99.9% and supporting high substrate concentrations, this technology addresses critical bottlenecks in traditional manufacturing, offering a robust solution for reliable API intermediate supplier networks aiming to optimize their production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
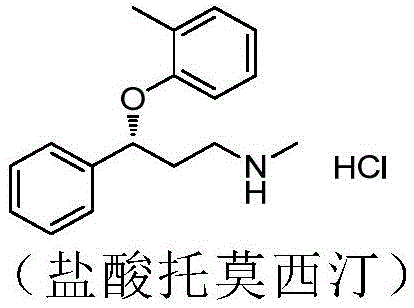
Historically, the industrial preparation of tomoxetine has relied on several distinct synthetic routes, each plagued by significant inefficiencies that hinder cost reduction in pharmaceutical manufacturing. One common approach involves the chemical reduction of 3-chlorophenyl acetone followed by resolution using mandelic acid. While chemically straightforward, this method is fundamentally limited by a theoretical maximum yield of only 50% due to the racemic nature of the chemical reduction, necessitating the disposal or recycling of the unwanted enantiomer. Another reported route utilizes Baker's yeast for chiral reduction; however, this biocatalytic step suffers from poor stereoselectivity, often resulting in an ee value of merely 70%, which requires extensive downstream purification. Furthermore, some existing pathways involve lengthy sequences including Boc protection and deprotection steps, which increase material costs, extend production timelines, and generate substantial chemical waste, making them less attractive for modern green chemistry standards.
The Novel Approach
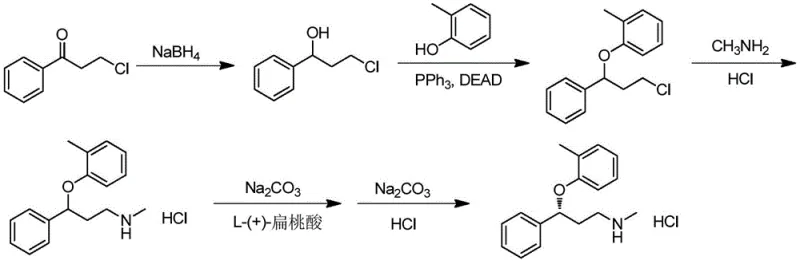
In stark contrast to these legacy methods, the technology disclosed in patent CN112708641A presents a streamlined chemo-enzymatic pathway that dramatically enhances process efficiency. The core innovation lies in the use of a specific carbonyl reductase that facilitates the direct asymmetric reduction of 3-chloropropiophenone to the chiral alcohol intermediate, (S)-3-chlorophenylpropanol. Unlike previous biocatalytic attempts that were restricted to low substrate concentrations (e.g., 2 mM), this novel enzyme system operates effectively at substrate concentrations ranging from 50 g/L to 1000 g/L. This massive increase in loading capacity translates directly to higher space-time yields and reduced solvent consumption. Following the enzymatic step, the process proceeds through a Mitsunobu reaction with o-cresol and a subsequent methylamination, culminating in salt formation. This route eliminates the need for resolution entirely, theoretically doubling the yield compared to racemic methods while ensuring superior optical purity from the outset.
Mechanistic Insights into Carbonyl Reductase Catalyzed Asymmetric Reduction
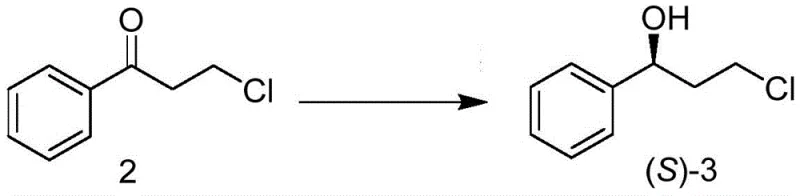
The heart of this technological advancement is the specialized carbonyl reductase, identified by amino acid sequence SEQ ID NO. 2, which exhibits remarkable specificity for the prochiral ketone substrate. Mechanistically, the enzyme facilitates the transfer of a hydride ion from a reduced coenzyme, such as NADH or NADPH, to the carbonyl carbon of 3-chloropropiophenone. The chiral environment of the enzyme's active site strictly controls the trajectory of this hydride attack, ensuring the exclusive formation of the (S)-enantiomer of the alcohol product. To make this process economically viable on an industrial scale, the patent describes a coupled coenzyme regeneration system. By employing enzymes like glucose dehydrogenase alongside a cosubstrate such as glucose, the expensive oxidized coenzymes (NAD+ or NADP+) are continuously recycled in situ. This regeneration loop ensures that only catalytic amounts of the cofactor are required, significantly lowering the raw material costs associated with the biotransformation step.
Beyond the primary reduction, the integrity of the chiral center is maintained throughout the subsequent chemical transformations. The downstream processing involves a Mitsunobu etherification, where the chiral alcohol reacts with o-cresol in the presence of triphenylphosphine and DEAD (diethyl azodicarboxylate). This reaction proceeds with inversion of configuration, which is a critical consideration in the overall stereochemical design, ultimately leading to the desired (R)-configuration in the final tomoxetine molecule after amination. The entire sequence is designed to minimize impurity formation; the high specificity of the initial enzymatic step ensures that the crude intermediate already possesses an ee value exceeding 99.9%, thereby simplifying the purification burden in later stages and ensuring the final high-purity pharmaceutical intermediate meets stringent regulatory specifications without complex chromatographic separations.
How to Synthesize Tomoxetine Efficiently
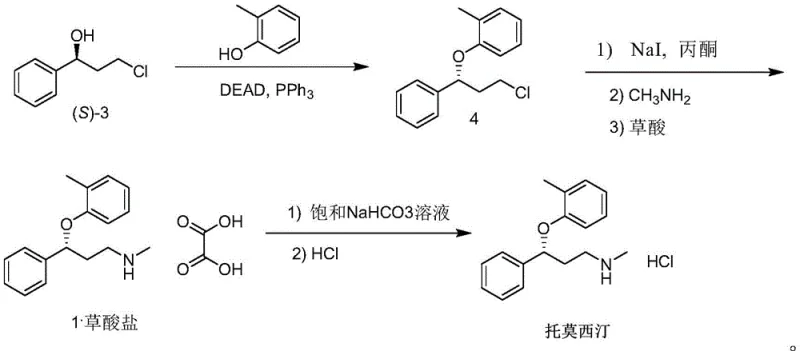
The implementation of this chemo-enzymatic strategy requires precise control over reaction parameters to maximize the benefits of the biocatalyst. The process begins with the preparation of the enzymatic reaction system, where the substrate concentration is carefully optimized to balance solubility and reaction rate, often utilizing cosolvents like dimethyl sulfoxide or isopropanol to enhance substrate tolerance. Following the biotransformation, the chiral alcohol is isolated and subjected to the Mitsunobu conditions under anhydrous environments to prevent side reactions. The subsequent amination and salt formation steps are standard organic transformations but are rendered more efficient due to the high purity of the incoming intermediate. For a detailed breakdown of the specific reagents, stoichiometry, and operational conditions required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Perform asymmetric reduction of 3-chloropropiophenone using a specific carbonyl reductase and coenzyme system to obtain (S)-3-chlorophenylpropanol with >99.9% ee.
- Execute a Mitsunobu reaction between the chiral alcohol intermediate and o-cresol to form the ether linkage, followed by activation with sodium iodide.
- Conduct methylamination using methylamine, followed by oxalate salt formation and subsequent conversion to the hydrochloride salt to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages that go beyond simple technical metrics. The shift from resolution-based or low-efficiency biocatalytic methods to this high-loading enzymatic process fundamentally alters the cost structure of tomoxetine production. By eliminating the 50% yield loss inherent in resolution processes and avoiding the multi-step protection strategies of older routes, the overall material throughput is significantly improved. This efficiency gain allows manufacturers to produce more API from the same amount of starting material, directly impacting the cost of goods sold (COGS) and enabling more competitive pricing structures in the global market for CNS therapeutics.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive resolving agents and the reduction of solvent volumes. Because the enzymatic step operates at high substrate concentrations (up to 1000 g/L), the volume of water and buffer required per kilogram of product is drastically lower than in traditional fermentation or dilute biocatalysis. Furthermore, the removal of Boc protection and deprotection steps reduces the consumption of reagents like di-tert-butyl dicarbonate and acids, simplifying the bill of materials. The patent explicitly notes that the enzyme cost for producing 1 kg of product is remarkably low, contributing to a leaner manufacturing budget without compromising on quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex synthetic routes that rely on scarce reagents or have low success rates. This chemo-enzymatic route utilizes recombinant enzymes that can be produced consistently via fermentation, ensuring a stable supply of the biocatalyst. The robustness of the enzyme, which tolerates organic cosolvents and high substrate loads, reduces the risk of batch failures due to solubility issues or inhibition. This reliability translates to more predictable production schedules and shorter lead times for high-purity pharmaceutical intermediates, allowing downstream formulation partners to plan their inventory with greater confidence and reduced safety stock requirements.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles. The high atom economy of the asymmetric reduction, combined with the avoidance of heavy metal catalysts often used in chemical hydrogenation, results in a cleaner waste profile. The process generates less organic waste and wastewater, simplifying effluent treatment and reducing compliance costs. Additionally, the simplicity of the work-up procedures—often requiring only extraction and concentration rather than complex chromatography—facilitates easier commercial scale-up of complex chiral intermediates from pilot plant to multi-ton production scales, ensuring that the technology remains viable as market demand grows.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is crucial for stakeholders evaluating its potential for integration into existing supply chains. The following questions address common inquiries regarding the performance, scalability, and regulatory implications of the chemo-enzymatic route described in patent CN112708641A. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What is the chiral purity achieved by the new enzymatic method compared to traditional routes?
A: The patented carbonyl reductase method achieves an enantiomeric excess (ee) value of greater than 99.9%, significantly surpassing traditional Baker's yeast methods which often yield around 70% ee, and eliminating the need for costly resolution steps.
Q: How does the substrate concentration in this process impact industrial scalability?
A: The process supports exceptionally high substrate concentrations ranging from 50 g/L to 1000 g/L. This high loading capacity drastically reduces solvent usage and reactor volume requirements, making it highly suitable for large-scale commercial manufacturing compared to previous biocatalytic methods limited to millimolar concentrations.
Q: Does this synthesis route require expensive metal catalysts or protecting groups?
A: No, the chemo-enzymatic route avoids the use of transition metal catalysts and eliminates the need for Boc protection and deprotection steps found in older synthetic pathways, thereby simplifying the workflow and reducing environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tomoxetine Supplier
As the global demand for ADHD medications continues to rise, the ability to source high-quality intermediates efficiently is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced chemo-enzymatic technologies to deliver superior value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN112708641A are fully realized in practical manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tomoxetine intermediate meets the highest international standards, providing our partners with the consistency required for regulatory filings and commercial success.
We invite procurement leaders and R&D directors to explore how our optimized synthesis routes can enhance your project economics. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your pharmaceutical supply chain.
