Revolutionizing Telmisartan Production: Advanced Intermediate Synthesis for Commercial Scale
The global demand for Angiotensin II receptor blockers (ARBs) continues to drive significant innovation in the pharmaceutical intermediates sector, with Telmisartan remaining a cornerstone molecule for cardiovascular therapy. Recent intellectual property developments, specifically Chinese Patent CN101024631A, have unveiled a transformative approach to synthesizing the critical precursor 4'-((4-methyl-6-(2-methylamino)carbanilino)-2-propyl-1H-benzimidazole-1-yl)methyl)biphenyl-2-carboxylic acid. This technical breakthrough addresses long-standing inefficiencies in the legacy manufacturing workflows that have plagued the industry for decades. By re-engineering the synthetic pathway to focus on stable acyl chloride intermediates and optimized alkylation conditions, this new methodology offers a robust framework for producing high-purity telmisartan intermediates. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a competitive edge in the supply chain. The shift away from unstable tert-butyl protecting groups towards more manageable ester functionalities represents a paradigm shift in process chemistry, promising enhanced operational safety and reduced environmental footprint without compromising the structural integrity required for final API cyclization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Telmisartan has been hindered by reliance on synthetic routes that are inherently inefficient and costly at scale, as illustrated in the traditional Scheme 1 found in earlier literature. The conventional pathway typically necessitates the use of trifluoroacetic acid (TFA) for the critical deprotection step, a reagent that is not only prohibitively expensive but also poses significant corrosion challenges to standard stainless steel reactor equipment. Furthermore, the purification of key intermediates in the old process frequently relies on column chromatography, a technique that is notoriously difficult to translate from laboratory benchtop to multi-ton commercial production due to excessive solvent consumption and low throughput. The instability of the tert-butyl ester intermediates under slightly acidic conditions often leads to premature deprotection, resulting in complex impurity profiles that require extensive downstream processing to resolve. These factors collectively contribute to extended lead times and inflated manufacturing costs, creating bottlenecks for reliable pharmaceutical intermediates supplier networks aiming to meet global demand.
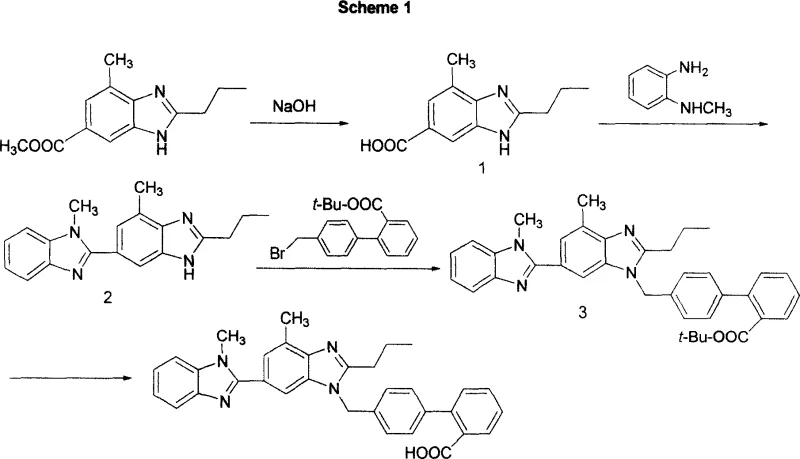
The Novel Approach
In stark contrast to the legacy methodologies, the novel approach detailed in the patent data introduces a streamlined sequence that prioritizes crystallization-driven purification over chromatographic separation. By converting the benzimidazole carboxylic acid into an acyl chloride prior to amidation, the process achieves higher reaction specificity and minimizes the formation of hard-to-remove side products. The strategic selection of alkyl esters or benzyl esters as protecting groups allows for milder deprotection conditions, such as alkaline hydrolysis or catalytic hydrogenolysis, which are far more compatible with large-scale reactor setups. This redesigned workflow eliminates the need for hazardous TFA handling and removes the throughput limitations associated with silica gel columns. Consequently, the novel approach facilitates a more continuous and predictable manufacturing cadence, enabling cost reduction in API manufacturing through simplified unit operations and reduced solvent recovery loads. The result is a chemically robust pathway that maintains high fidelity to the target molecular structure while drastically simplifying the physical processing requirements.
Mechanistic Insights into Benzimidazole Acylation and Alkylation
The core chemical innovation lies in the precise control of the acylation and subsequent N-alkylation steps, which dictate the overall purity profile of the final intermediate. The process initiates with the activation of 7-methyl-2-propyl-3H-benzimidazole-5-carboxylic acid using thionyl chloride, generating a highly reactive acyl chloride species that readily couples with N-methyl-o-phenylenediamine hydrochloride. This reaction is conducted under strictly controlled low-temperature conditions, typically between -10°C and 10°C, to suppress potential thermal degradation and ensure selective amide bond formation. The use of mineral alkalis or organic bases like triethylamine serves to scavenge the generated hydrochloric acid, driving the equilibrium towards the desired amide product while keeping the reaction mixture homogeneous. Following isolation, the amide intermediate undergoes N-alkylation with biphenyl methyl bromide derivatives in the presence of strong bases such as sodium hydride. This step is critical for installing the biphenyl moiety, and the choice of anhydrous polar aprotic solvents ensures complete solubility of the ionic intermediates, thereby maximizing conversion rates and minimizing unreacted starting materials that could complicate downstream purification.
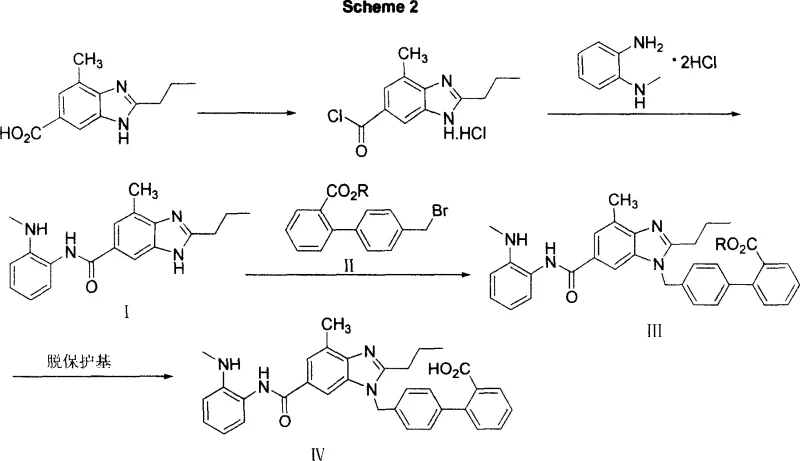
Impurity control within this mechanistic framework is achieved primarily through the physical properties of the intermediates rather than complex chemical scavenging. The patent highlights that the target amide and ester intermediates possess distinct solubility characteristics that allow them to precipitate as adularescent solids upon pH adjustment or solvent addition. This phenomenon is exploited to purge soluble impurities, such as excess amines or salt byproducts, directly during the workup phase. By adjusting the pH to specific ranges, such as 8 to 14 for precipitation or 1 to 5 for acidification, operators can selectively crystallize the product while leaving contaminants in the mother liquor. This crystallization-induced purity enhancement is far superior to chromatographic methods in terms of scalability and cost-efficiency. Furthermore, the stability of the ester protecting group throughout the alkylation step prevents the formation of free carboxylic acid impurities that could otherwise interfere with the final cyclization to Telmisartan. This rigorous control over the reaction environment ensures that the impurity spectrum remains narrow and manageable, meeting the stringent quality standards required for high-purity telmisartan intermediate production.
How to Synthesize Telmisartan Intermediate Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and thermal management to ensure consistent batch-to-batch quality. The detailed standardized synthesis steps involve the initial activation of the benzimidazole acid, followed by controlled amidation, alkylation with biphenyl derivatives, and final deprotection to yield the target carboxylic acid. Operators must adhere to strict anhydrous conditions during the acyl chloride formation and alkylation phases to prevent hydrolysis of sensitive intermediates. The purification strategy relies heavily on pH-controlled crystallization, necessitating precise monitoring of acidity levels during the quench and isolation stages. For a comprehensive breakdown of the specific reaction parameters, solvent volumes, and workup procedures, please refer to the technical guide below.
- Convert 7-methyl-2-propylbenzimidazole-5-carboxylic acid to its corresponding acyl chloride using thionyl chloride.
- React the acyl chloride with N-methyl-o-phenylenediamine hydrochloride under controlled alkaline conditions to form the amide intermediate.
- Perform N-alkylation using sodium hydride and biphenyl methyl bromide derivatives in anhydrous organic solvents.
- Execute final deprotection via hydrolysis or hydrogenolysis to isolate the target carboxylic acid intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers substantial benefits that extend beyond mere chemical yield improvements. The elimination of column chromatography represents a massive reduction in operational expenditure, as it removes the need for large quantities of silica gel and the associated solvent disposal costs that typically burden fine chemical manufacturing budgets. Additionally, the replacement of trifluoroacetic acid with standard mineral acids or catalytic hydrogenation methods significantly lowers raw material costs and reduces the corrosive load on plant infrastructure, extending the lifespan of capital equipment. These process simplifications translate directly into a more resilient supply chain, as the reliance on specialized, hard-to-source reagents is minimized in favor of commodity chemicals that are readily available in the global market. For procurement managers, this means reduced risk of supply disruption and greater flexibility in vendor negotiations, ensuring a steady flow of critical materials for downstream API production.
- Cost Reduction in Manufacturing: The structural redesign of the synthetic pathway fundamentally alters the cost basis of production by removing high-cost purification steps. By shifting from chromatography to crystallization, the process drastically reduces solvent consumption and waste treatment volumes, leading to significant operational savings. The avoidance of expensive fluorinated reagents further contributes to a leaner cost structure, allowing for more competitive pricing models without sacrificing margin. This efficiency gain is compounded by the higher throughput achievable with filtration-based isolation compared to batch-wise column loading, enabling facilities to maximize asset utilization and reduce the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the new chemical route enhances supply security by mitigating the risks associated with unstable intermediates and complex purification bottlenecks. Since the process utilizes stable ester protecting groups and standard reaction conditions, the likelihood of batch failures due to unexpected side reactions is significantly diminished. This predictability allows for more accurate production planning and inventory management, ensuring that delivery schedules can be met consistently even during periods of high market demand. Furthermore, the use of common industrial solvents and reagents reduces dependency on niche suppliers, diversifying the supply base and strengthening the overall resilience of the procurement network against external shocks.
- Scalability and Environmental Compliance: The inherent design of this synthesis method aligns perfectly with the principles of green chemistry and industrial scalability. The reduction in solvent intensity and the elimination of solid waste from chromatography media contribute to a lower environmental footprint, facilitating easier compliance with increasingly stringent environmental regulations. The process is readily transferable from pilot scale to commercial production without the need for specialized equipment modifications, supporting rapid capacity expansion. This scalability ensures that the manufacturing partner can grow alongside the client's needs, providing a long-term solution for the commercial scale-up of complex benzimidazoles that is both sustainable and economically viable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel telmisartan intermediate synthesis. These insights are derived directly from the patent specifications and practical manufacturing considerations, aimed at clarifying the operational benefits and chemical feasibility for potential partners. Understanding these details is crucial for making informed decisions about technology transfer and supply agreements.
Q: How does this new route improve upon conventional telmisartan synthesis?
A: The patented method eliminates the need for expensive trifluoroacetic acid (TFA) deprotection and avoids column chromatography purification, significantly reducing solvent waste and processing time while improving overall yield stability.
Q: What are the critical purity controls in this benzimidazole alkylation?
A: Critical controls involve maintaining strict temperature ranges during acylation to prevent side reactions and utilizing crystallization-induced precipitation rather than chromatographic separation to remove unreacted starting materials and salts.
Q: Is this process suitable for multi-ton commercial manufacturing?
A: Yes, the reliance on standard unit operations like reflux, filtration, and crystallization, combined with the avoidance of hazardous column chromatography, makes this route highly adaptable for large-scale industrial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent concept to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of telmisartan intermediate meets the highest international standards. We understand the critical nature of cardiovascular supply chains and are committed to delivering consistent quality and reliability.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering excellence can optimize your supply chain for high-purity ARB intermediates.
