Advanced Iridium-Catalyzed Asymmetric Hydrogenation for Scalable Pharmaceutical Intermediate Production
The landscape of asymmetric synthesis for nitrogen-containing heterocycles has been fundamentally shifted by the innovations detailed in patent CN1468852A, which introduces a highly enantioselective iridium-catalyzed hydrogenation method. This technology addresses a long-standing challenge in organic chemistry: the efficient reduction of aromatic pyridine and pyrazine rings without compromising stereochemical integrity. For R&D directors and process chemists, this patent represents a pivotal advancement, offering a robust catalytic system capable of achieving up to 96% asymmetric induction under relatively mild conditions. By utilizing a specialized combination of iridium chiral complexes and specific adducts, this method overcomes the thermodynamic stability of aromatic systems that typically necessitates extreme pressure and temperature in conventional processes. The implications for the supply chain of high-purity pharmaceutical intermediates are profound, as it enables the production of chiral building blocks with unprecedented efficiency and purity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of aromatic compounds has been plagued by significant thermodynamic and kinetic barriers. Traditional catalytic systems, such as Wilkinson-type rhodium catalysts or early ruthenium bisphosphine complexes, often struggle with the inherent stability of the aromatic ring, requiring harsh reaction conditions to force hydrogenation. Literature precedents indicate that breaking aromaticity usually demands high temperatures and elevated pressures, which can lead to catalyst decomposition and poor selectivity. Furthermore, the presence of heteroatoms like nitrogen and sulfur in these substrates frequently acts as a catalyst poison, deactivating the metal center and resulting in negligible conversion rates. Previous attempts, such as the HRh(DIOP) system developed by Murata, yielded dismal enantiomeric excess values as low as 3%, rendering them commercially non-viable for producing high-value chiral intermediates. These limitations have forced manufacturers to rely on resolution techniques or multi-step synthetic routes that generate substantial waste and increase overall production costs.
The Novel Approach
The novel approach described in CN1468852A circumvents these historical bottlenecks through a sophisticated iridium-based catalytic architecture. By employing a catalytic system composed of iridium chiral complexes paired with specific adducts like iodine, amines, or quaternary ammonium salts, the method achieves remarkable reactivity at temperatures ranging from 0°C to 80°C. This system demonstrates exceptional tolerance for various functional groups and does not require the nitrogen atom to be protected, a significant advantage over earlier rhodium-based methods that necessitated N-Ac or N-Boc protection. The ability to operate at substrate-to-catalyst ratios as high as 5000:1 indicates a turnover number that drastically reduces the reliance on expensive precious metals. This innovation transforms the synthesis of substituted quinolines and quinoxalines from a difficult, low-yield transformation into a streamlined, high-efficiency process suitable for industrial application.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the unique interaction between the iridium metal precursor and the diverse array of chiral ligands employed. The catalyst preparation involves reacting neutral or ionic iridium compounds, such as [Ir(COD)Cl]2 or Ir(COD)2BF4, with chiral ligands including bisphosphines, nitrogen-phosphorus ligands, or sulfur-phosphorus ligands. This pre-activation step, followed by the addition of critical adducts like iodine or tetrabutylammonium iodide, generates a highly active cationic iridium species. This active species is uniquely capable of coordinating with the electron-deficient aromatic rings of pyridines and pyrazines, facilitating the hydride transfer necessary for reduction. The mechanistic pathway likely involves the formation of an iridium-dihydride complex that selectively delivers hydrogen to the prochiral face of the substrate, driven by the steric and electronic environment created by the chiral ligand backbone.
Crucially, this catalytic system exhibits broad substrate scope, accommodating a wide variety of substituents on the aromatic ring without significant loss in enantioselectivity. As illustrated in the structural diversity below, the method is effective for methyl-substituted quinolines, ethyl-substituted derivatives, and even more complex fused ring systems.
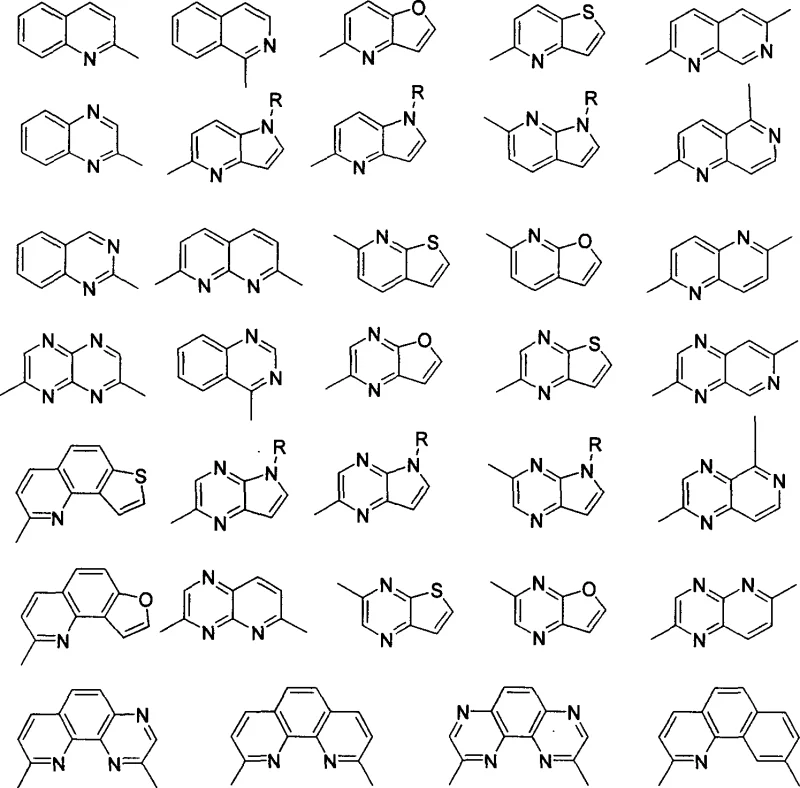
The impurity profile control is inherently superior in this method due to the high specificity of the iridium catalyst. Unlike non-selective hydrogenation which can lead to over-reduction or ring-opening side reactions, this system maintains strict control over the reduction of the specific nitrogen-containing ring. The use of mild solvents such as dichloromethane, toluene, or isopropanol further minimizes the formation of solvent-derived byproducts. For procurement managers, this high level of selectivity translates directly into simplified downstream processing, as the crude reaction mixtures contain fewer impurities that require costly chromatographic separation. The ability to achieve greater than 90% enantiomeric excess for most substrates ensures that the final API intermediates meet the stringent purity specifications required by global regulatory bodies.
How to Synthesize Chiral Quinolines Efficiently
The implementation of this synthesis route requires precise control over catalyst activation and reaction parameters to maximize yield and optical purity. The patent outlines a standardized protocol where the iridium precursor and chiral ligand are stirred under inert atmosphere before the introduction of the substrate and hydrogen gas. Detailed standard operating procedures regarding stoichiometry, pressure ramping, and workup protocols are essential for reproducibility at scale.
- Prepare the catalyst by reacting an iridium metal precursor, such as [Ir(COD)Cl]2, with a chiral ligand like (R)-MeO-Biphep in a solvent such as toluene or dichloromethane.
- Add specific adducts, such as iodine or tetrabutylammonium iodide, to the mixture to activate the catalytic species and enhance enantioselectivity.
- Introduce the substrate (e.g., 2-substituted quinoline) and subject the mixture to hydrogen pressure (1-100 atm) at temperatures between 0-80°C to achieve high conversion and ee.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement strategists, the adoption of this iridium-catalyzed hydrogenation technology offers tangible economic and operational benefits. The shift from multi-step, low-yield synthetic routes to a direct, catalytic hydrogenation process fundamentally alters the cost structure of producing chiral heterocyclic intermediates. By eliminating the need for protecting group manipulation and reducing the number of unit operations, manufacturers can achieve significant reductions in raw material consumption and waste generation. This process intensification aligns perfectly with modern green chemistry initiatives, reducing the environmental footprint associated with pharmaceutical manufacturing while simultaneously improving margin potential.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the exceptionally high turnover number of the catalyst system. With substrate-to-catalyst ratios reaching up to 5000:1, the consumption of expensive iridium metal and chiral ligands is minimized on a per-kilogram basis. Furthermore, the elimination of protecting group steps removes the cost of additional reagents and the associated waste disposal fees. The mild reaction conditions also imply lower energy consumption for heating and pressurization compared to traditional high-temperature hydrogenation methods, contributing to a leaner overall cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against catalyst poisoning by nitrogen and sulfur heteroatoms ensures consistent batch-to-batch performance. In traditional processes, trace impurities in starting materials could deactivate catalysts, leading to stalled reactions and delayed shipments. This new method's tolerance allows for the use of commercially available starting materials with standard purity grades, reducing the risk of supply disruptions caused by raw material quality fluctuations. Additionally, the broad applicability of the technology means that a single manufacturing platform can produce a wide variety of intermediates, increasing asset utilization and flexibility.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of common industrial solvents like toluene and isopropanol, which are readily available and easy to recover. The high selectivity of the reaction reduces the load on wastewater treatment facilities by minimizing the generation of complex organic byproducts. As regulatory pressure on pharmaceutical effluents increases, adopting a cleaner, catalytic process provides a strategic advantage in maintaining compliance with environmental standards without incurring excessive remediation costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities.
Q: What are the advantages of this iridium-catalyzed method over traditional rhodium systems?
A: Unlike traditional rhodium systems which often require harsh conditions or specific protecting groups, this iridium-based method operates under milder temperatures and pressures while achieving superior enantiomeric excess (up to 96% ee) for unfunctionalized aromatic heterocycles.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent highlights a high substrate-to-catalyst ratio of up to 5000:1, indicating high turnover numbers that significantly reduce catalyst loading costs, making it highly viable for commercial scale-up.
Q: What types of substrates are compatible with this hydrogenation technique?
A: The system is versatile and effective for a broad range of substituted aromatic pyridine and pyrazine rings, including quinolines and quinoxalines, which are critical scaffolds in pharmaceutical and agrochemical synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enantioselective iridium-catalyzed hydrogenation route for producing high-value chiral building blocks. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market supply is seamless. Our facility is equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that meet or exceed patent benchmarks.
We invite you to collaborate with our technical team to evaluate the feasibility of applying this advanced catalytic method to your specific pipeline projects. By leveraging our expertise in process optimization, we can help you achieve substantial cost savings and accelerate your time to market. Please contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your target molecules.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
