Scalable Chemical Synthesis of Beta-Nicotinamide Mononucleotide for Commercial Production
Scalable Chemical Synthesis of Beta-Nicotinamide Mononucleotide for Commercial Production
The rapidly expanding market for NAD+ precursors has placed beta-nicotinamide mononucleotide (NMN) at the forefront of nutraceutical and pharmaceutical research. As detailed in patent CN112724180A, a novel chemical synthesis pathway has been developed that addresses critical bottlenecks in existing manufacturing technologies. This method utilizes a strategically protected ribose derivative to achieve high stereoselectivity and simplified purification, marking a significant advancement for industrial-scale production. By shifting away from expensive enzymatic substrates and problematic direct phosphorylation routes, this technology offers a robust framework for reliable pharmaceutical intermediate supplier operations. The following analysis dissects the technical merits and commercial implications of this innovative four-step synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, NMN production has relied heavily on enzymatic catalysis or less selective chemical routes, both of which present substantial hurdles for commercial scale-up. Enzymatic methods often require costly raw materials such as D-5-phosphoribose or nicotinamide riboside, alongside complex multi-enzyme systems like PRPPS and NAMPT, which demand precise reaction conditions and expensive equipment. Furthermore, direct chemical phosphorylation of nicotinamide ribose frequently suffers from poor regioselectivity, leading to a mixture of mono-, di-, and tri-phosphorylated impurities that are structurally similar and notoriously difficult to separate. These purification challenges not only drive up costs but also compromise the overall yield and purity required for high-grade applications.
The Novel Approach
The patented process introduces a refined chemical strategy that circumvents these issues through careful protecting group manipulation. By starting with a ribose derivative where the primary hydroxyl is protected with a tert-butyldiphenylsilyl (TBDPS) group and secondary hydroxyls are acylated, the synthesis ensures that subsequent reactions occur with high specificity. This approach prevents the formation of unwanted poly-phosphorylated byproducts and facilitates the removal of inorganic salts through organic phase extraction. The result is a streamlined workflow that maintains high yields while significantly simplifying downstream processing, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
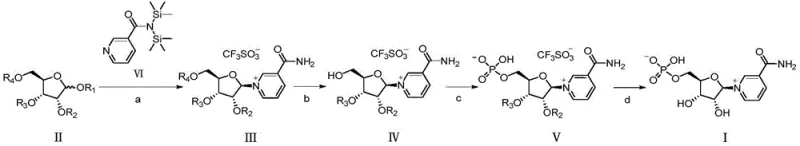
Mechanistic Insights into TMSOTf-Catalyzed Glycosylation and Selective Phosphorylation
The core of this synthesis lies in the glycosylation step, where a silylated nicotinamide derivative reacts with the protected ribose donor under the influence of a Lewis acid catalyst, specifically trimethylsilyl trifluoromethanesulfonate (TMSOTf). This catalyst activates the anomeric center of the ribose, promoting nucleophilic attack by the nitrogen of the nicotinamide ring. The presence of participating acyl groups at the C2 and C3 positions of the ribose ring assists in directing the stereochemistry, favoring the formation of the desired beta-anomer through neighboring group participation. This mechanistic feature is crucial for ensuring the biological efficacy of the final NMN product, as the alpha-anomer is inactive.
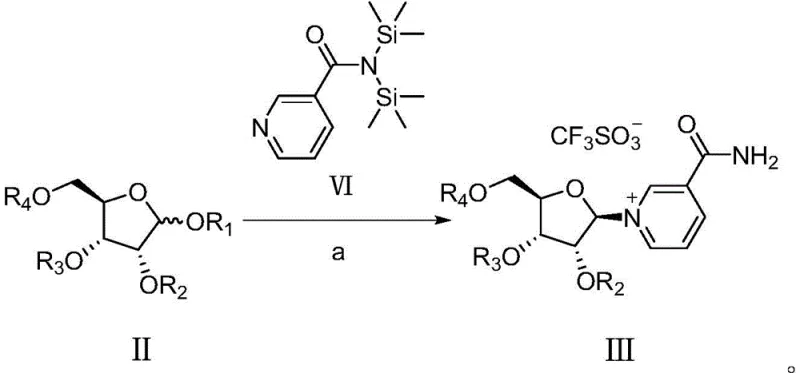
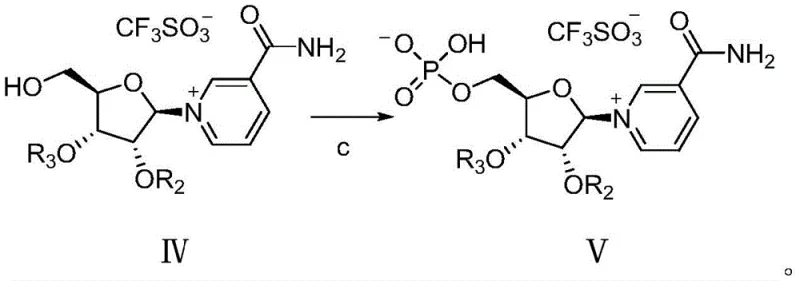
Following glycosylation and selective deprotection of the primary hydroxyl group, the process moves to the phosphorylation stage. Here, the free 5-hydroxyl group reacts with phosphorus oxychloride (POCl3). Because the secondary hydroxyls remain protected during this step, the phosphorylation is strictly regioselective for the primary position. This eliminates the generation of ectopic phosphorylation impurities (such as 2'- or 3'-phosphates) that plague other methods. The resulting phosphate ester is then subjected to global deacylation, typically using sodium methoxide, to reveal the final hydrophilic NMN structure. This sequence ensures that the complex phosphate moiety is introduced only after the glycosidic bond is securely established and purified.
How to Synthesize Beta-Nicotinamide Mononucleotide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of solvent selection and temperature control to maximize yield. The use of acetonitrile as a solvent for the glycosylation step and tetrahydrofuran for deprotection highlights the need for polarity management throughout the sequence. Detailed operational parameters, including molar ratios of catalysts and specific reaction temperatures, are critical for reproducing the high stereoselectivity reported in the examples. For a comprehensive breakdown of the exact experimental conditions and workup procedures required to implement this route, please refer to the standardized guide below.
- Condensation reaction between protected ribose derivative (Formula II) and silylated nicotinamide (Formula VI) using TMSOTf catalyst to form Formula III.
- Selective deprotection of the primary hydroxyl group in Formula III using fluoride reagents to obtain Formula IV.
- Phosphorylation of the free primary hydroxyl in Formula IV using phosphorus oxychloride to generate Formula V.
- Final deacylation of Formula V using sodium methoxide to yield the target beta-nicotinamide mononucleotide (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this chemical route offers distinct advantages over both fermentation and legacy chemical methods by decoupling production from biological variability and expensive specialty substrates. The reliance on commodity chemicals like ribose, acetic anhydride, and benzoyl chloride ensures a stable and predictable supply chain, reducing the risk of raw material shortages that often affect enzymatic processes dependent on niche biocatalysts. Furthermore, the ability to purify intermediates using standard extraction and chromatography techniques rather than complex enzymatic separations translates to lower capital expenditure on specialized equipment.
- Cost Reduction in Manufacturing: The elimination of expensive enzymes and costly starting materials like nicotinamide riboside drastically lowers the bill of materials. Additionally, the high selectivity of the phosphorylation step reduces the burden on purification resources, minimizing solvent consumption and waste disposal costs associated with removing closely related phosphate impurities. This efficiency gain allows for a more competitive pricing structure for bulk NMN production without compromising quality standards.
- Enhanced Supply Chain Reliability: By utilizing a fully chemical synthesis pathway, manufacturers are not subject to the batch-to-batch variability inherent in biological fermentation or the shelf-life limitations of enzymatic reagents. The robust nature of the protected intermediates allows for potential storage and stockpiling, providing greater flexibility in production scheduling and ensuring consistent delivery timelines for downstream clients requiring high-purity API intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from glassware to large-scale reactors. The improved purification profile means fewer recycling loops and less energy-intensive distillation steps are required to meet purity specifications. This streamlined approach not only accelerates time-to-market for commercial scale-up of complex pharmaceutical intermediates but also aligns better with environmental regulations by reducing the overall chemical footprint per kilogram of product.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this specific NMN synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on reaction mechanisms and process optimization strategies.
Q: How does this chemical method improve upon enzymatic synthesis for NMN?
A: Unlike enzymatic methods that require expensive substrates like D-5-phosphoribose or Nicotinamide Riboside and complex enzyme systems, this chemical route utilizes cost-effective protected ribose derivatives. It eliminates the need for specialized bioreactors and offers superior control over phosphorylation selectivity, avoiding complex poly-phosphorylated impurities common in direct chemical phosphorylation.
Q: What ensures the high stereoselectivity of the beta-anomer in this process?
A: The process employs a specific glycosylation strategy using silylated nicotinamide and a Lewis acid catalyst (TMSOTf) with protected ribose donors. The steric hindrance provided by the 2,3-di-O-acyl protecting groups and the 5-O-TBDPS group directs the nucleophilic attack to favor the formation of the beta-glycosidic bond, ensuring high stereochemical purity essential for biological activity.
Q: Why is the purification of intermediates considered easier in this new method?
A: The introduction of lipophilic protecting groups like tert-butyldiphenylsilyl (TBDPS) and acetyl/benzoyl groups significantly alters the solubility profile of the intermediates. This allows for effective separation of inorganic salts and polar impurities via standard organic extraction and washing steps, overcoming the difficulty of purifying highly water-soluble ammonium salts encountered in prior art methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nicotinamide Mononucleotide Supplier
As the demand for NAD+ boosters continues to surge, securing a supply partner with proven expertise in advanced chemical synthesis is paramount. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver NMN that meets stringent purity specifications. Our rigorous QC labs ensure that every batch adheres to the highest standards, minimizing impurities and maximizing bioavailability for your final formulations.
We invite you to discuss how this optimized synthesis route can benefit your specific product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments to support your transition to a more efficient and reliable supply chain.
