Revolutionizing Hydrocortisone Production: A Green Synthetic Route for Commercial Scale-up
Revolutionizing Hydrocortisone Production: A Green Synthetic Route for Commercial Scale-up
The global demand for corticosteroids remains steadfast, driven by their critical role in treating inflammatory and autoimmune conditions. However, the manufacturing landscape for key intermediates like hydrocortisone has long been plagued by environmental hazards and complex purification challenges. Patent CN102367262A introduces a transformative methodology that redefines the synthesis of this vital API intermediate. By shifting away from traditional oxidative pathways that rely on hazardous chromium reagents and carcinogenic nitrites, this invention establishes a new benchmark for green chemistry in steroid manufacturing. For R&D directors and procurement specialists alike, understanding this technological pivot is essential for securing a sustainable and cost-effective supply chain. This report dissects the technical merits of this novel route, highlighting its potential to drastically reduce operational risks while maintaining superior product quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of hydrocortisone has relied heavily on semi-synthetic routes derived from microbial fermentation intermediates, such as those produced by the Rhizopus niger method. While effective, these legacy processes are fraught with significant safety and environmental liabilities. A primary bottleneck involves the protection and deprotection of ketone functionalities at the C3 and C20 positions. Conventional strategies often employ semicarbazone protection, which subsequently requires sodium nitrite for removal—a substance classified as toxic and carcinogenic. Furthermore, the oxidation steps frequently necessitate the use of chromium anhydride, generating heavy metal waste that demands expensive and energy-intensive remediation protocols. These factors not only inflate the cost of goods sold but also introduce substantial regulatory compliance burdens for manufacturers aiming to meet modern environmental standards.
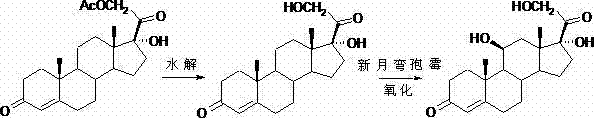
The Novel Approach
In stark contrast, the methodology disclosed in CN102367262A leverages a sophisticated ketal protection strategy that circumvents the need for hazardous deprotection agents. By utilizing ethylene glycol and triethyl orthoformate, the process effectively masks the C3 and C20 ketones, creating a stable intermediate that allows for highly selective downstream transformations. This approach eliminates the reliance on sodium nitrite entirely, thereby removing a major toxicological risk from the production line. Moreover, the subsequent steps utilize mild iodination and acetate substitution conditions that operate under standard atmospheric pressure and moderate temperatures. This shift not only simplifies the engineering requirements—removing the need for specialized cryogenic equipment found in other competing patents—but also significantly enhances the overall atom economy of the synthesis, presenting a compelling value proposition for cost-conscious procurement teams.
Mechanistic Insights into Ketal-Protection Mediated Steroid Synthesis
The core innovation of this patent lies in the precise orchestration of functional group protection to enable selective reduction. The process initiates with the conversion of 17α-hydroxy-4-pregnene-3,11,20-trione into its 3,20-diketal derivative. This structural modification is critical because it prevents the unwanted reduction of the C3 and C20 carbonyls during the subsequent hydride treatment. When borohydride reagents, such as sodium borohydride or potassium borohydride, are introduced, they selectively target the C11 ketone due to steric and electronic differentiation provided by the ketal groups. This selectivity is paramount for achieving the correct 11β-hydroxyl stereochemistry required for biological activity. Following reduction, the ketal groups are cleaved via mild acid hydrolysis, regenerating the ketones without affecting the newly formed hydroxyl group, thus demonstrating a high degree of chemoselectivity that is often difficult to achieve in complex steroid scaffolds.
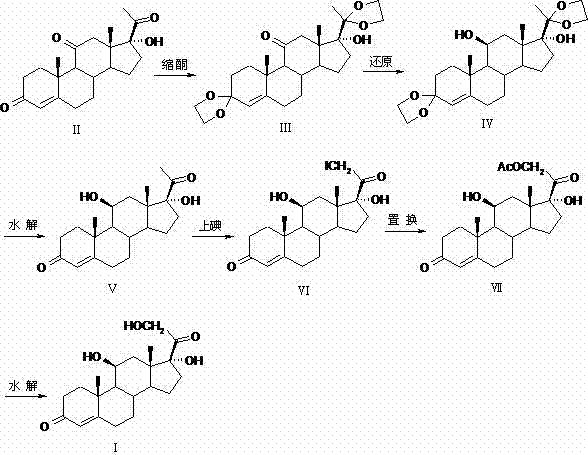
Following the restoration of the ketone functionalities, the synthesis proceeds through a unique iodination sequence at the C21 position. Unlike traditional halogenation methods that might require aggressive conditions, this protocol employs a calcium oxide and iodine system in a methanol-calcium chloride medium. This specific reagent combination facilitates the formation of the 21-iodo intermediate with high regioselectivity, minimizing the formation of di-iodinated byproducts. The subsequent nucleophilic substitution with potassium acetate efficiently installs the acetoxy group, yielding hydrocortisone acetate. Finally, a gentle saponification step removes the acetate moiety to deliver the final hydrocortisone API. Each step in this cascade is designed to maximize yield and minimize impurity generation, ensuring that the final product meets stringent pharmacopoeial specifications with minimal downstream purification effort.
How to Synthesize Hydrocortisone Efficiently
Implementing this synthesis route requires careful attention to reaction stoichiometry and temperature control, particularly during the ketalization and iodination phases. The patent outlines a robust protocol where the ratio of ethylene glycol to the starting trione is maintained between 3:1 and 5:1 to drive the equilibrium towards the protected ketal. Similarly, the iodination step benefits from a controlled addition of the iodine-calcium chloride solution at low temperatures (0-5°C) to prevent side reactions. While the specific laboratory-scale parameters are detailed in the examples, scaling this process requires a thorough understanding of heat transfer dynamics during the exothermic ketal formation and the precise dosing of the iodinating agent. For a comprehensive breakdown of the standardized operating procedures and critical process parameters required for GMP manufacturing, please refer to the technical guide below.
- Perform ketal protection on 17α-hydroxy-4-pregnene-3,11,20-trione using ethylene glycol and triethyl orthoformate to protect C3 and C20 ketones.
- Execute selective reduction of the C11 ketone to 11β-hydroxyl using borohydride reagents under reflux conditions.
- Conduct acid hydrolysis to remove protecting groups, followed by mild iodination at C21 and acetate substitution to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the transition to this novel synthetic route offers profound strategic advantages beyond mere technical elegance. The elimination of toxic reagents like sodium nitrite and chromium salts directly translates to a simplified waste management infrastructure. Facilities no longer need to invest in specialized heavy metal scrubbing systems or incur the high costs associated with the disposal of hazardous chemical waste. This reduction in environmental overhead significantly lowers the total cost of manufacturing, making the final API more price-competitive in the global market. Furthermore, the reliance on common, commercially available auxiliary materials ensures that raw material sourcing remains stable and resilient against market fluctuations, mitigating the risk of supply disruptions that often plague specialty chemical supply chains.
- Cost Reduction in Manufacturing: By removing the need for expensive heavy metal catalysts and toxic deprotection agents, the process inherently reduces raw material costs. Additionally, the avoidance of complex purification steps required to remove trace heavy metals from the final product streamlines the downstream processing workflow. This efficiency gain means less solvent consumption and reduced energy usage per kilogram of product, driving down the operational expenditure significantly without compromising on the quality or potency of the pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The reagents utilized in this pathway, such as ethylene glycol, triethyl orthoformate, and potassium acetate, are commodity chemicals with robust global supply networks. This contrasts sharply with older methods that may depend on niche or regulated precursors. Consequently, manufacturers adopting this route can secure long-term supply contracts with greater confidence, ensuring consistent production schedules and reliable delivery timelines for their downstream pharmaceutical clients who depend on uninterrupted API availability.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, operating at near-ambient pressures and moderate temperatures that are easily managed in standard stainless steel reactors. This compatibility with existing infrastructure reduces the capital expenditure required for technology transfer and scale-up. Moreover, the green nature of the chemistry aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against potential regulatory crackdowns on hazardous chemical usage and ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrocortisone synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for evaluating the feasibility of integrating this route into your existing production portfolio.
Q: How does this new process improve safety compared to traditional Rhizopus niger methods?
A: Traditional methods often require sodium nitrite for deprotection, which is carcinogenic, and chromium salts for oxidation. This new patent utilizes a ketal protection strategy and mild iodination, completely eliminating the need for these highly toxic and corrosive reagents.
Q: What is the expected purity profile of hydrocortisone produced via this route?
A: The patent data indicates that this synthetic pathway consistently achieves HPLC purity levels exceeding 98.5%, with overall yields surpassing 77%, ensuring a robust impurity profile suitable for pharmaceutical grade applications.
Q: Is this synthesis route scalable for industrial manufacturing?
A: Yes, the process utilizes common auxiliary materials and avoids cryogenic equipment or deep cold conditions required by other methods. The reaction conditions, such as reflux temperatures and ambient pressure iodination, are highly conducive to large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydrocortisone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies is key to maintaining competitiveness in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale discoveries like the one in CN102367262A can be successfully translated into industrial reality. We are committed to delivering high-purity hydrocortisone and its intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to green chemistry principles aligns with this patent's ethos, allowing us to offer products that are not only cost-effective but also environmentally responsible.
We invite pharmaceutical manufacturers and contract research organizations to collaborate with us on optimizing this synthesis route for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can enhance your supply chain efficiency and product quality.
