Advanced Synthesis of Voglibose Intermediates for Commercial Scale-Up
The global pharmaceutical landscape is continuously evolving to meet the demands of chronic disease management, with Type 2 diabetes representing a critical area of focus for medicinal chemistry innovation. Within this sector, Voglibose stands out as a potent alpha-glucosidase inhibitor, essential for regulating postprandial blood glucose levels. However, the commercial viability of such life-saving medications is often hindered by complex and inefficient manufacturing processes. The patent CN111747857B introduces a transformative approach to synthesizing key amino sugar intermediates, addressing long-standing bottlenecks in yield and operational safety. This technical insight report analyzes the proprietary methodology disclosed in the patent, highlighting its potential to redefine supply chain standards for reliable pharmaceutical intermediates supplier networks. By shifting away from multi-step protection strategies towards a direct epoxidation mechanism, this technology offers a pathway to more robust and economically viable production systems.
The significance of this patent extends beyond mere chemical novelty; it represents a strategic asset for procurement and supply chain leaders seeking to optimize their API sourcing strategies. Traditional methods for producing Voglibose have historically suffered from low atom economy and the use of hazardous reagents, creating vulnerabilities in the supply chain. The disclosed invention mitigates these risks by streamlining the synthetic route, thereby enhancing the reliability of supply. For R&D directors, the detailed mechanistic insights provided in the patent offer a clear understanding of how stereoselectivity is achieved without cumbersome chiral auxiliaries. This report serves as a comprehensive guide for decision-makers evaluating the integration of this advanced synthesis route into their existing manufacturing portfolios, ensuring alignment with both quality standards and cost-efficiency goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Voglibose has been plagued by inefficiencies that drive up costs and complicate regulatory compliance. As illustrated in the prior art, conventional routes often involve more than ten chemical steps, starting from glucose or requiring fermentation followed by extensive chemical modification. These legacy processes frequently rely on the use of dangerous reagents such as bromine and benzyl chloroformate, which pose significant safety hazards and environmental disposal challenges. Furthermore, the necessity for multiple hydroxyl protection and deprotection cycles results in substantial material loss and increased solvent consumption. The cumulative effect of these factors is a low overall yield and a high production cost, making the final API expensive and difficult to source consistently. The complexity of these routes also introduces multiple points of failure, where impurities can accumulate, necessitating rigorous and costly purification protocols to meet pharmacopeial standards.
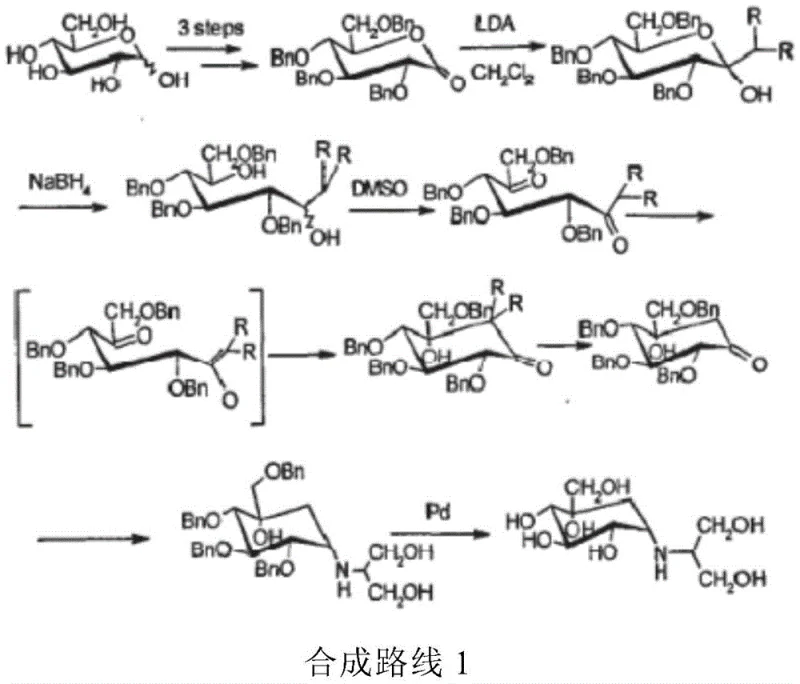
The Novel Approach
In stark contrast to the convoluted legacy pathways, the method disclosed in CN111747857B offers a streamlined alternative that fundamentally simplifies the molecular construction of the target intermediates. The core innovation lies in the direct epoxidation of valienamine or its derivatives, bypassing the need for extensive protecting group manipulation. By utilizing a specific combination of polar solvents and organic acids, the process facilitates the in situ formation of a quaternary ammonium salt. This intermediate state is crucial as it protects the amino group from unwanted oxidation while simultaneously directing the stereochemical outcome of the epoxidation. The result is a significant reduction in the number of synthetic steps and a marked improvement in operational simplicity. This approach not only enhances the overall yield but also drastically reduces the environmental footprint of the manufacturing process by minimizing waste generation and solvent usage.
Mechanistic Insights into Direct Epoxidation of Valienamine Derivatives
The chemical elegance of this patented process is rooted in its sophisticated control over reaction kinetics and thermodynamics. The mechanism begins with the protonation of the amino group in valienamine using a strong organic acid, such as p-toluenesulfonic acid or methanesulfonic acid. This protonation converts the nucleophilic amine into a stable quaternary ammonium salt, effectively masking it from the oxidizing environment. Subsequently, the addition of a peroxide oxidant, such as m-chloroperoxybenzoic acid (m-CPBA) or dimethyl ketone peroxide, initiates the epoxidation of the intra-ring double bond. The presence of the positively charged ammonium species creates a hydrogen bonding network with the transition state of the peroxide. This interaction is pivotal as it locks the conformation of the molecule, ensuring high stereoselectivity during the oxygen transfer. This level of control eliminates the formation of unwanted diastereomers, which are common pitfalls in carbohydrate chemistry, thereby simplifying downstream purification.
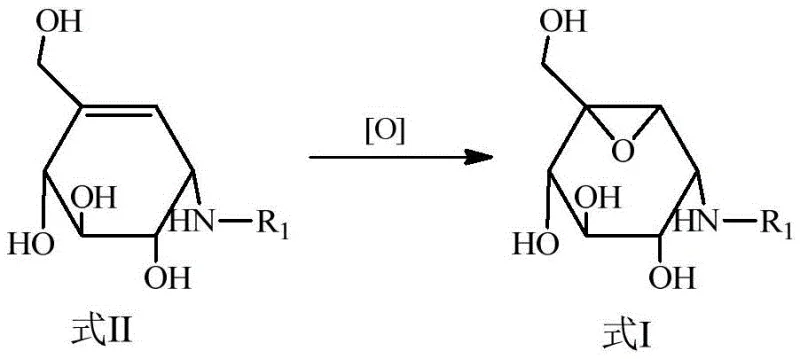
Impurity control is another critical aspect where this mechanism excels, providing significant value for quality assurance teams. In traditional synthesis, the exposure of free amino groups to oxidizing conditions often leads to N-oxide formation or other degradation products that are difficult to remove. By maintaining the amino group in a protonated salt form throughout the oxidation step, the patented method effectively suppresses these side reactions. Furthermore, the choice of polar solvents like DMF or methanol ensures excellent solubility of the ionic intermediates, preventing precipitation that could lead to inconsistent reaction rates. The subsequent workup involves a simple neutralization and resin chromatography, which is far more scalable than the crystallization techniques required for protected intermediates. This robustness in impurity profiling ensures that the resulting intermediates meet the stringent purity specifications required for subsequent API synthesis, reducing the risk of batch rejection.
How to Synthesize Voglibose Intermediates Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize efficiency and yield. The process begins by dissolving the valienamine starting material in a selected polar solvent system, followed by the careful addition of an organic acid to generate the reactive salt species. Once the salt formation is complete, the peroxide oxidant is introduced at a controlled temperature, typically ranging from -20°C to 80°C depending on the specific reagents used. The reaction is allowed to proceed for a defined period, after which it is quenched and neutralized. The crude product is then purified using ion-exchange resin chromatography to isolate the high-purity epoxy intermediate. This intermediate can then be subjected to reductive ring-opening to yield Voglibose. Detailed standardized synthesis steps see the guide below.
- Dissolve valienamine or its derivative in a polar solvent and add acid to form a quaternary ammonium salt in situ.
- Introduce a peroxide oxidant at controlled temperatures between -20°C and 80°C to initiate the epoxidation reaction.
- Quench the reaction, neutralize the mixture, and purify the resulting epoxy intermediate using resin chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond simple unit cost calculations. The primary advantage lies in the drastic simplification of the manufacturing workflow. By eliminating the need for multiple protection and deprotection steps, the process reduces the total number of unit operations required. This reduction directly correlates to lower labor costs, decreased energy consumption, and reduced equipment occupancy time. Furthermore, the avoidance of high-risk reagents like bromine simplifies the safety compliance burden, lowering insurance premiums and reducing the need for specialized containment infrastructure. These factors collectively contribute to a more resilient supply chain capable of withstanding market fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by improved atom economy and reduced material throughput. Since the process avoids the use of expensive protecting groups and the reagents required to install and remove them, the raw material cost per kilogram of product is significantly lowered. Additionally, the reduction in solvent volume and the simplification of waste streams lead to substantial savings in disposal and recycling costs. The higher overall yield means that less starting material is required to produce the same amount of final product, further enhancing the cost efficiency. These savings can be passed down the supply chain, offering a competitive pricing advantage for the final API without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes that rely on niche or hazardous reagents. This new method utilizes commercially available and stable chemicals, reducing the risk of supply disruptions. The simplified process flow also means that production cycles are shorter, allowing for faster turnaround times and more responsive inventory management. For a reliable pharmaceutical intermediates supplier, this reliability is crucial for maintaining long-term contracts with major pharmaceutical companies. The robustness of the chemistry ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed quality control tests or out-of-specification results.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden complexities, but this route is inherently designed for scale-up. The reaction conditions are mild, and the exothermicity is manageable, reducing the engineering challenges associated with large-scale reactors. Moreover, the environmental profile of the process is superior to traditional methods. By generating less hazardous waste and consuming fewer resources, the process aligns with modern green chemistry principles. This alignment facilitates easier regulatory approval and supports corporate sustainability goals, making it an attractive option for companies committed to reducing their carbon footprint in cost reduction in API manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation. Understanding these aspects is vital for stakeholders evaluating the feasibility of adopting this route for commercial production. The answers provide clarity on how the chemical innovations translate into practical benefits for manufacturing and quality control teams.
Q: How does the new epoxidation method improve upon traditional Voglibose synthesis?
A: The new method eliminates the need for multiple hydroxyl protection and deprotection steps required in conventional routes. By forming a quaternary ammonium salt in situ, it prevents amino group oxidation and controls stereoselectivity, significantly shortening the synthetic sequence and reducing solvent consumption.
Q: What are the safety advantages of this process for industrial production?
A: Traditional routes often utilize hazardous reagents such as bromine and benzyl chloroformate. This novel approach avoids these high-risk chemicals, relying instead on safer peroxide oxidants and common organic acids, thereby enhancing operational safety and simplifying waste treatment protocols.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the process is designed for scalability. It reduces the total number of unit operations and avoids complex purification steps associated with protecting groups. The use of readily available starting materials like valienamine and standard polar solvents facilitates easy technology transfer from laboratory to commercial plant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Voglibose Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical industry. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN111747857B and is fully prepared to leverage these insights for your project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Voglibose intermediate meets the highest international standards. We are committed to delivering high-purity Voglibose intermediates that support the uninterrupted manufacture of life-saving diabetes medications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to our deep expertise in commercial scale-up of complex amino sugars and our commitment to reducing lead time for high-purity pharmaceutical intermediates. Please contact us today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-effective supply of critical pharmaceutical ingredients for your global operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
