Advanced Palladium-Catalyzed Route to High-Purity Trifluoromethyl Imidazoles for Commercial Pharmaceutical Manufacturing Scale-Up
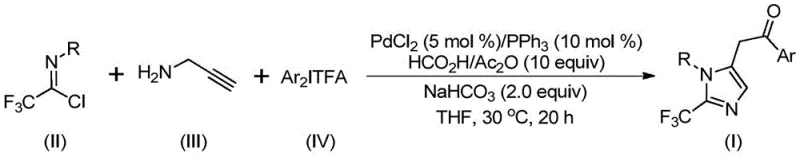
Patent CN111423381B discloses a groundbreaking methodology for synthesizing high-purity 2-trifluoromethyl substituted imidazole compounds through an innovative palladium-catalyzed carbonylation process that operates under exceptionally mild conditions of just 30°C for precisely controlled reaction times between 18 to 24 hours. This patented approach represents a significant advancement in heterocyclic chemistry with direct implications for pharmaceutical intermediate manufacturing where purity specifications are non-negotiable requirements from regulatory authorities worldwide. The methodology utilizes readily available starting materials including trifluoroethylimidoyl chloride (II), propargylamine (III), and diaryl iodonium salts (IV), all of which demonstrate excellent commercial availability through established global supply chains without requiring specialized handling protocols or hazardous storage conditions. By integrating formic acid and acetic anhydride as a safe carbon monoxide surrogate system, the process eliminates dependency on high-pressure equipment typically required for carbonylation reactions while maintaining excellent conversion efficiency across diverse substrate combinations. This innovation establishes a robust foundation for producing critical building blocks used in numerous bioactive molecules including H1 histamine receptor antagonists like Alcaftadine, demonstrating clear relevance to pharmaceutical development pipelines where structural diversity is essential for lead optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-substituted imidazole compounds frequently encounter multiple critical challenges that severely limit their commercial viability and scalability within pharmaceutical manufacturing environments. Many established methods require extreme reaction conditions such as elevated temperatures exceeding 100°C or cryogenic environments below -40°C, which substantially increase energy consumption while introducing significant safety hazards during large-scale implementation that necessitate expensive engineering controls and specialized operator training protocols. These conventional routes often depend on expensive or unstable reagents with limited commercial availability that create supply chain vulnerabilities through single-source dependencies while generating inconsistent product quality due to batch-to-batch variability in raw material specifications. Furthermore, traditional syntheses typically exhibit narrow substrate scope with poor functional group tolerance that restricts molecular diversity needed for pharmaceutical development programs requiring rapid structural modifications during lead optimization phases. Purification processes frequently involve multiple complex steps including recrystallization from hazardous solvents or preparative chromatography with low throughput capacity that significantly reduce overall yield while increasing production costs through extended processing times and specialized equipment requirements.
The Novel Approach
The patented methodology presented in CN111423381B addresses these limitations through an elegant palladium-catalyzed system operating under remarkably mild conditions that eliminate the need for extreme temperatures or specialized high-pressure equipment typically associated with carbonylation chemistry. By utilizing formic acid and acetic anhydride as a safe carbon monoxide surrogate system at precisely controlled temperature of 30°C for standard reaction duration of 20 hours, this innovation achieves efficient transformation without requiring hazardous reagents or complex engineering controls that complicate traditional manufacturing workflows. The process demonstrates exceptional substrate flexibility with broad functional group tolerance across diverse aromatic systems including methyl-, tert-butyl-, chloro-, bromo-, trifluoromethyl-, and nitro-substituted variants as evidenced by successful implementation across fifteen experimental examples yielding products I-1 through I-15 with consistent quality profiles. Commercially available catalysts including palladium chloride paired with triphenylphosphine ligand deliver excellent reaction efficiency while maintaining cost-effectiveness compared to alternative transition metal systems requiring expensive ligands or precious metal loadings.
Mechanistic Insights into Palladium-Catalyzed Imidazole Formation
The reaction mechanism initiates with intermolecular carbon-nitrogen bond formation promoted by sodium bicarbonate between trifluoroethylimidoyl chloride (II) and propargylamine (III), yielding a trifluoroacetamidine intermediate through nucleophilic substitution under mild basic conditions without requiring elevated temperatures or specialized catalysts. This key transformation step occurs spontaneously at ambient pressure while maintaining excellent chemoselectivity toward amide bond formation over competing hydrolysis pathways due to precise stoichiometric control between reactants. The resulting intermediate undergoes isomerization to form a more stable configuration that facilitates subsequent coordination with palladium(II) species generated from palladium chloride precursor and triphenylphosphine ligand through oxidative addition at the alkyne moiety of propargylamine derivative. This critical step forms an alkenyl palladium complex serving as a crucial catalytic intermediate that subsequently isomerizes through proton transfer mechanisms to generate an alkyl palladium species capable of undergoing carbonylation using carbon monoxide released in situ from formic acid/acetic anhydride system.
Impurity control is achieved through multiple built-in mechanisms within this catalytic cycle that prevent common side reactions observed in alternative synthetic approaches including hydrolysis products from moisture exposure or dimerization byproducts from uncontrolled radical pathways. The mild reaction temperature of exactly 30°C minimizes thermal decomposition pathways that typically generate byproducts in higher temperature processes while maintaining optimal catalyst activity throughout the entire reaction duration. Precise stoichiometric balance between reactants prevents over-reaction or incomplete conversion issues that could lead to impurity formation through residual starting materials or partially transformed intermediates. Sodium bicarbonate functions as a mild base avoiding strong alkaline conditions that might cause hydrolysis or other unwanted transformations of sensitive functional groups present in diverse substrate combinations tested across fifteen experimental examples.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
This patented methodology provides a streamlined approach to producing high-purity pharmaceutical intermediates through carefully optimized reaction parameters that ensure consistent product quality across diverse substrate combinations while maintaining operational simplicity suitable for industrial implementation. The synthesis leverages commercially available starting materials with established global supply chains eliminating dependency on specialty chemicals that create procurement bottlenecks in traditional manufacturing workflows. By operating at ambient pressure under precisely controlled temperature conditions without requiring cryogenic cooling or high-pressure reactors, this process significantly reduces capital expenditure requirements while enhancing operational safety profiles compared to conventional approaches used in heterocyclic chemistry manufacturing environments.
- Prepare reaction mixture by combining palladium chloride (5 mol%), triphenylphosphine (10 mol%), sodium bicarbonate (2.0 equiv), formic acid/acetic anhydride (10 equiv), trifluoroethylimidoyl chloride (II), propargylamine (III), and diaryl iodonium salt (IV) in tetrahydrofuran solvent under inert atmosphere.
- Stir reaction mixture at precisely controlled temperature of 30°C for duration of 20 hours to ensure complete conversion while maintaining optimal catalyst activity and minimizing side reactions.
- Execute post-reaction processing through filtration to remove solids followed by silica gel mixing and standard column chromatography purification to isolate high-purity target imidazole compound meeting pharmaceutical specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis route addresses critical pain points in pharmaceutical intermediate supply chains by delivering a robust manufacturing solution that enhances reliability while reducing operational complexity across multiple dimensions essential for procurement decision-making processes within multinational pharmaceutical organizations seeking dependable partners for critical intermediate sourcing requirements.
- Cost Reduction in Manufacturing: Elimination of expensive transition metal catalysts requiring complex removal procedures substantially reduces downstream processing costs while minimizing waste generation associated with metal scavenging operations required by alternative methodologies employing precious metal systems needing extensive purification steps before final product isolation can occur.
- Enhanced Supply Chain Reliability: Utilization of widely available starting materials with multiple commercial sources ensures consistent raw material availability regardless of regional market fluctuations or geopolitical disruptions affecting specialty chemical procurement channels while providing flexibility through vendor diversification strategies that mitigate single-source dependency risks common in traditional synthetic routes.
- Scalability and Environmental Compliance: Streamlined workup procedures requiring only basic filtration followed by standard column chromatography significantly reduce processing time compared to multi-step purification methods employed in conventional syntheses while generating substantially less hazardous waste aligning with increasingly stringent environmental regulations governing chemical manufacturing operations worldwide.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation feasibility and technical advantages based on comprehensive analysis of patent CN111423381B's experimental data and process parameters specifically relevant to procurement teams evaluating reliable pharmaceutical intermediate suppliers capable of delivering consistent quality at commercial scale.
Q: How does this patented method overcome limitations of conventional imidazole synthesis routes?
A: This methodology operates under mild conditions (30°C) without requiring extreme temperatures or pressures characteristic of traditional approaches. It utilizes cost-effective starting materials with broad commercial availability while demonstrating exceptional substrate tolerance across diverse functional groups through its innovative palladium-catalyzed carbonylation mechanism.
Q: What specific advantages does this process offer for pharmaceutical purity requirements?
A: The built-in impurity control mechanisms prevent common side reactions through precise temperature management (30°C) and optimized stoichiometry. Simplified workup procedures involving basic filtration followed by standard column chromatography ensure consistent production of high-purity compounds meeting stringent pharmaceutical specifications without requiring additional purification steps.
Q: How does this synthesis route enhance supply chain reliability for pharmaceutical manufacturers?
A: By leveraging widely available starting materials with established global supply chains and eliminating dependency on specialized equipment or hazardous reagents, this method significantly reduces vulnerability to single-point failures while ensuring consistent raw material availability regardless of regional market fluctuations or geopolitical disruptions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
Our patented methodology represents a significant advancement in heterocyclic chemistry with direct applications in pharmaceutical intermediate manufacturing where purity specifications are paramount concerns requiring rigorous analytical validation at every production stage. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through state-of-the-art manufacturing facilities equipped with advanced analytical instrumentation operating under ISO-certified quality management systems ensuring consistent product quality meeting global regulatory requirements including FDA and EMA standards.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your particular application needs while taking advantage of our Customized Cost-Saving Analysis service which identifies potential efficiency improvements without compromising product quality or regulatory compliance requirements essential for pharmaceutical manufacturing operations worldwide.
