Advanced Synthesis of Aztreonam Intermediates: A Cost-Effective Route for Commercial Scale-Up
Advanced Synthesis of Aztreonam Intermediates: A Cost-Effective Route for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of critical antibiotic intermediates, particularly for monocyclic beta-lactams like Aztreonam. Patent CN100586930C introduces a groundbreaking preparation method for (3S-trans)-3-amino-4-methyl-2-oxo-1-sulfoazetidine, a pivotal building block in the synthesis of this life-saving medication. This innovative approach leverages strong acid ion exchange resin catalysis to streamline what was traditionally a cumbersome multi-step sequence into a highly efficient one-pot reaction. By integrating amidation, cyclization, and sulfonation under controlled conditions, this technology addresses long-standing challenges in yield optimization and process simplicity. For global supply chain leaders, this represents a significant opportunity to enhance production reliability while maintaining stringent quality standards required for active pharmaceutical ingredients.
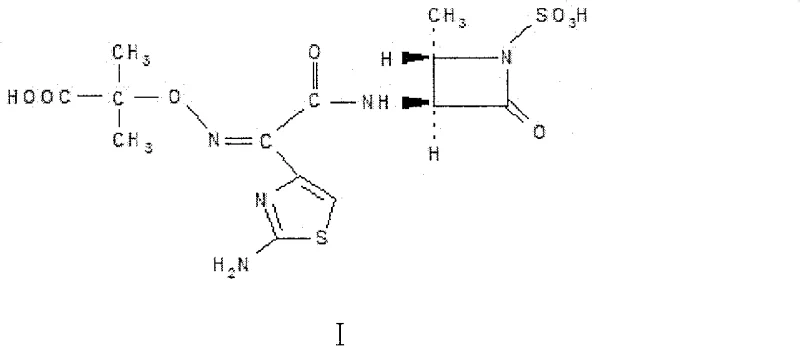
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Aztreonam main ring has been plagued by complex reaction schemes that hinder efficient commercial manufacturing. Traditional methods, such as those disclosed in earlier patents like EP86556, often rely on difficult-to-obtain raw materials or require harsh reaction conditions that are unsafe for large-scale operations. One common route involves the nitrene derivation of azetidinone, which presents significant safety hazards and scalability issues due to the instability of intermediates. Another approach utilizes chirality Threonine protection but suffers from an excessively long reaction cycle and numerous purification steps. These conventional pathways not only inflate production costs through high material consumption but also introduce multiple points of failure in the supply chain, leading to inconsistent batch quality and extended lead times for pharmaceutical manufacturers seeking reliable partners.
The Novel Approach
In stark contrast, the novel methodology described in CN100586930C revolutionizes the synthesis landscape by condensing three distinct chemical transformations into a single, cohesive operational flow. This approach utilizes N-protected L-Threonine as a readily available starting material, reacting it with ammonia to form a hydroxy butyramide intermediate before proceeding directly to cyclization. The core innovation lies in the use of sulfonic acid ion exchange resin, such as Dowex-50, which acts as both a catalyst and a reagent carrier during the introduction of SO3 gas. This eliminates the need for hazardous liquid acids and simplifies the workup process to mere filtration and concentration. The result is a drastic reduction in the production cycle, effectively halving the time required compared to prior art, while simultaneously boosting the overall yield to commercially viable levels suitable for metric-ton production.
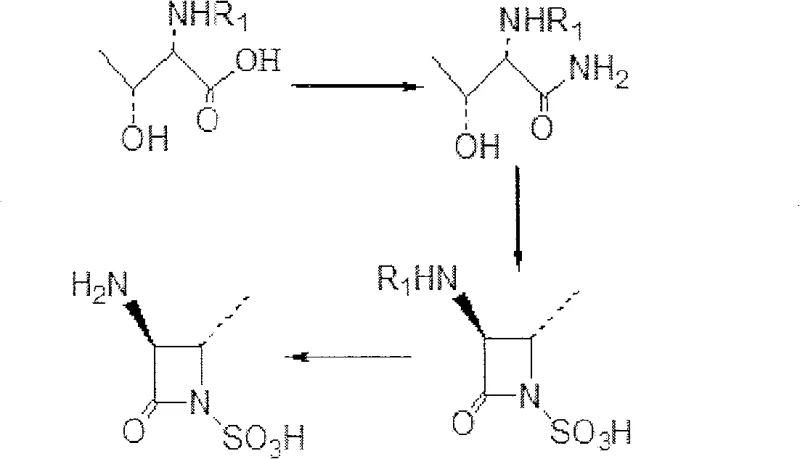
Mechanistic Insights into Resin-Catalyzed Cyclization and Sulfonation
The chemical elegance of this process is rooted in the synergistic interaction between the substrate and the solid-phase acid catalyst. When the (2S, 3R)-2-(R1-amino)-3-hydroxyl butyramide intermediate is dissolved in anhydrous methanol, the addition of strong acid ion exchange resin creates a localized acidic environment conducive to intramolecular cyclization. As SO3 gas is introduced into the system at a controlled temperature of 30°C, the resin facilitates the activation of the hydroxyl group, promoting nucleophilic attack by the amide nitrogen to form the four-membered beta-lactam ring. Simultaneously, the sulfonic acid groups on the resin assist in the sulfonation of the nitrogen atom, securing the 1-sulfoazetidine structure in a single operational step. This mechanism avoids the formation of unstable sulfonic acid chlorides or other aggressive sulfonating agents, thereby preserving the stereochemical integrity of the chiral centers which is critical for the biological activity of the final antibiotic.
Furthermore, the impurity profile of the resulting intermediate is significantly improved due to the specificity of the resin-catalyzed reaction. In conventional liquid-phase acid catalysis, side reactions such as over-sulfonation or ring-opening hydrolysis are common, leading to complex impurity spectra that are difficult and costly to remove. The solid-state nature of the ion exchange resin limits the mobility of reactive species, effectively suppressing these side pathways and ensuring a cleaner reaction mixture. The weight ratio of the substrate to the resin is meticulously optimized between 1:1.8 and 1:3.5, with a preferred ratio of 1:2, to maximize conversion efficiency without generating excessive waste. This precise control over the reaction environment ensures that the final product meets the rigorous purity specifications demanded by regulatory bodies for pharmaceutical intermediates, reducing the burden on downstream purification processes.
How to Synthesize (3S-trans)-3-amino-4-methyl-2-oxo-1-sulfoazetidine Efficiently
Implementing this synthesis route requires careful attention to solvent dryness and gas flow rates to ensure optimal performance. The process begins with the dissolution of N-protected L-Threonine in anhydrous polar solvents like methanol, followed by the introduction of ammonia gas to generate the amide precursor. Once this intermediate is formed, it is directly subjected to the resin-catalyzed cyclization step without isolation, showcasing the efficiency of the one-pot design. The reaction is maintained at a mild temperature range of 10°C to 60°C, preferably at 30°C, for a duration of 12 to 24 hours to ensure complete conversion. Detailed standardized synthetic steps, including specific reagent grades and safety protocols for handling SO3 gas, are outlined in the technical guide below for process engineers.
- Dissolve N-protected L-Threonine in anhydrous methanol and introduce ammonia gas to form the hydroxy butyramide intermediate.
- Add sulfonic acid ion exchange resin (e.g., Dowex-50) and feed SO3 gas at 30°C for 20 hours to effect cyclization and sulfonation in a one-pot reaction.
- Remove the amino protecting group via catalytic hydrogenolysis to yield the final (3S-trans)-3-amino-4-methyl-2-oxo-1-sulfoazetidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this resin-catalyzed synthesis route offers tangible strategic advantages that extend beyond mere technical feasibility. The simplification of the process flow directly translates to a reduction in operational expenditures, as fewer unit operations mean lower energy consumption and reduced labor requirements. By eliminating the need for expensive transition metal catalysts or hazardous liquid acids, the raw material costs are significantly lowered, and the safety profile of the manufacturing facility is greatly enhanced. This cost structure allows for more competitive pricing models in the global market, providing a buffer against raw material price volatility while maintaining healthy margins for all stakeholders involved in the supply chain.
- Cost Reduction in Manufacturing: The elimination of multiple isolation and purification steps drastically reduces solvent usage and waste disposal costs. By utilizing a reusable ion exchange resin and avoiding expensive reagents, the overall cost of goods sold is substantially decreased. This qualitative efficiency gain ensures that the manufacturing process remains economically viable even at lower production volumes, offering flexibility in meeting market demand without compromising profitability.
- Enhanced Supply Chain Reliability: The robustness of the one-pot reaction minimizes the risk of batch failures associated with complex multi-step syntheses. Since the starting materials like L-Threonine are commodity chemicals with stable global availability, the risk of supply disruption is mitigated. The simplified post-treatment process, involving only filtration and concentration, accelerates the turnover time for production vessels, thereby increasing the overall throughput capacity and ensuring consistent on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reactor equipment and avoiding extreme pressure or temperature conditions. The use of solid-phase catalysts reduces the generation of acidic wastewater, aligning with increasingly stringent environmental regulations. This green chemistry approach not only simplifies regulatory compliance but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical procurement strategies focused on long-term viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent data and practical process understanding to assist decision-makers in evaluating the feasibility of this route for their specific production needs. Understanding these details is crucial for aligning technical capabilities with commercial objectives and ensuring a smooth technology transfer process.
Q: How does the ion exchange resin method improve yield compared to conventional synthesis?
A: The use of strong acid ion exchange resin facilitates a one-pot cyclization and sulfonation process, eliminating multiple isolation steps. This reduces material loss and improves overall yield to approximately 80.0% for the cyclization step, significantly higher than multi-step conventional routes.
Q: What are the critical reaction conditions for the sulfonation step?
A: The reaction requires feeding SO3 gas into a methanol solution containing the substrate and resin. The optimal temperature is maintained at 30°C for 20 hours, with a resin-to-substrate weight ratio of 1:2 to ensure complete conversion without excessive byproduct formation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It utilizes commercially available resins and simple gas-liquid reactions. The post-treatment involves basic filtration and concentration, avoiding complex extraction or chromatography, which makes it ideal for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aztreonam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the global antibiotic supply chain. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the resin-catalyzed route are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (3S-trans)-3-amino-4-methyl-2-oxo-1-sulfoazetidine meets the highest international standards. Our commitment to technical excellence ensures that your production schedules are met with consistent, high-quality materials that facilitate seamless downstream API manufacturing.
We invite you to collaborate with us to optimize your supply chain for Aztreonam production. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical data and our proven track record in delivering complex pharmaceutical intermediates on a global scale.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
