Advanced Mesotrione Manufacturing: Overcoming Toxicity Barriers with Novel Rearrangement Catalysts
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate manufacturing, specifically for compounds like mesotrione, a critical HPPD inhibitor used extensively in corn cultivation. A significant technological breakthrough in this sector is detailed in Chinese Patent CN113233998B, which outlines a novel preparation method that fundamentally alters the rearrangement step of the synthesis. Unlike conventional routes that rely on hazardous reagents, this invention introduces a class of rearrangement agents characterized by unsaturated double bonds, represented by Formula (I). This strategic modification not only enhances the reaction kinetics through electronic synergy but also addresses the pressing environmental and safety concerns associated with legacy cyanide-based processes. For R&D directors and supply chain leaders, this patent represents a pivotal shift towards sustainable chemistry, offering a pathway to produce high-purity agrochemical intermediates with reduced ecological footprints and simplified downstream processing.
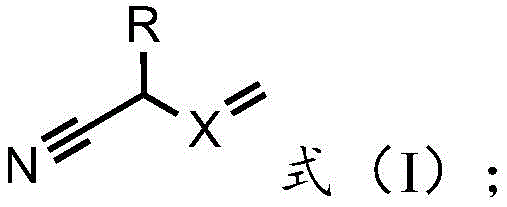
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of mesotrione has been plagued by significant safety and environmental liabilities, primarily stemming from the reliance on highly toxic rearrangement catalysts. Prior art, such as U.S. Patent No. US5886231, discloses the use of sodium cyanide, a substance that poses extreme toxicity risks to personnel and creates formidable challenges in wastewater treatment due to residual cyanide ions remaining in the aqueous phase. Similarly, other established methods utilize acetone cyanohydrin, which, while effective, carries similar virulent properties that necessitate rigorous containment and disposal protocols, thereby inflating operational costs. Alternative approaches involving alkali metal azides, as seen in TW089115906, introduce the risk of explosion, requiring specialized equipment and stringent safety measures that hinder large-scale adoption. Furthermore, methods employing bulky rearrangement agents like 6-chloropurine increase raw material costs and complicate the purification of the final product due to the difficulty in separating high molecular weight byproducts. These conventional pathways collectively suffer from poor atom economy, hazardous waste generation, and complex work-up procedures that limit their viability in a modern, regulated chemical manufacturing environment.
The Novel Approach
The methodology presented in CN113233998B offers a transformative solution by replacing these hazardous catalysts with specific unsaturated nitriles defined by Formula (I), where X is CH or N and R varies among hydrogen, hydroxyl, chlorine, or alkyl groups. This new class of rearrangement agents leverages the electronic properties of the unsaturated double bond to activate the enol ester intermediate more efficiently than saturated counterparts. Experimental data within the patent demonstrates that agents such as 2-hydroxy-3-butenenitrile or N-methyleneaminoacetonitrile facilitate the rearrangement reaction with superior rates and selectivity under mild thermal conditions. By operating at temperatures between 10-70°C, the process minimizes energy consumption while avoiding the decomposition pathways often triggered by harsher conditions. The result is a streamlined synthesis where the target mesotrione is obtained with high purity (up to 98.9%) and excellent molar yields (reaching 96.9%), effectively bypassing the toxicity and waste issues inherent in cyanide or azide-based routes.
Mechanistic Insights into Unsaturated Nitrile-Catalyzed Rearrangement
The core innovation of this technology lies in the mechanistic role of the unsaturated double bond within the rearrangement agent. In the context of the enol ester rearrangement, the presence of the alkene functionality adjacent to the nitrile group creates a conjugated system that enhances the nucleophilicity or Lewis basicity of the nitrogen atom, facilitating a more rapid attack on the carbonyl carbon of the enol ester. This synergistic effect lowers the activation energy required for the formation of the key transition state, leading to faster conversion rates compared to saturated nitriles like acetonitrile, which showed negligible activity (only 8.2% yield in comparative examples). The specific structural variations allowed in Formula (I), such as the inclusion of hydroxyl or chloro substituents on the allylic position, further fine-tune the electronic density and steric environment, optimizing the interaction with the substrate. This precise tuning ensures that the rearrangement proceeds with high regioselectivity, minimizing the formation of isomeric impurities that are difficult to remove in later stages.
Furthermore, the mechanism supports a cleaner impurity profile by avoiding the generation of inorganic salts or heavy metal complexes often associated with traditional base-catalyzed systems. The reaction mixture, post-rearrangement, can be treated through a straightforward sequence of filtration, dissolution in water, and acidification to pH 2-4. This simplicity suggests that the byproducts formed are either water-soluble or remain in the organic phase, allowing the target mesotrione to crystallize out with high fidelity. The absence of persistent toxic residues means that the wastewater generated is significantly easier to treat, aligning with increasingly strict global environmental regulations. For process chemists, this mechanistic clarity provides a robust framework for scaling the reaction, as the kinetic parameters are well-defined and less susceptible to the variability introduced by heterogeneous catalysts or unstable reagents.
How to Synthesize Mesotrione Efficiently
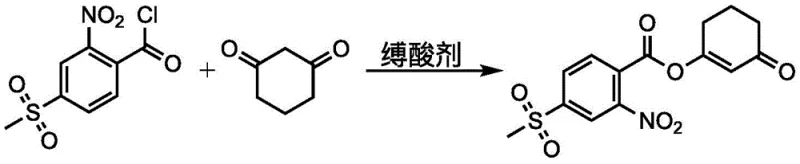
The synthesis protocol described in the patent is designed for operational simplicity and high throughput, making it an ideal candidate for technology transfer. The process begins with the preparation of the acyl chloride using solid phosgene, followed by condensation with 1,3-cyclohexanedione to form the enol ester, and concludes with the critical rearrangement step. Each stage is optimized for solvent compatibility and temperature control to ensure maximum yield and safety. The following guide outlines the standardized approach derived from the patent embodiments, highlighting the critical control points for successful implementation.
- Prepare 2-nitro-4-methylsulfonylbenzoyl chloride by reacting the corresponding benzoic acid with solid phosgene in an organic solvent at 20-100°C.
- React the acyl chloride intermediate with 1,3-cyclohexanedione in the presence of an acid-binding agent to form the enol ester reaction liquid.
- Add a rearrangement agent containing unsaturated double bonds (Formula I) to the enol ester liquid at 10-70°C to effect rearrangement into mesotrione.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond mere chemical efficiency. The elimination of highly regulated and dangerous reagents like sodium cyanide drastically reduces the compliance burden and insurance costs associated with handling hazardous materials. Moreover, the switch to solid phosgene for the initial chlorination step eliminates the release of sulfur dioxide gas, a common byproduct of thionyl chloride usage, thereby simplifying scrubber requirements and reducing capital expenditure on pollution control equipment. The overall process design favors continuous or large-batch operations due to its robust thermal profile and simple work-up, ensuring a reliable supply of high-purity mesotrione to meet market demand without the bottlenecks typical of older, more fragile chemistries.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the reduction in waste treatment costs and the improved yield of the final product. By avoiding the use of expensive and toxic catalysts that require specialized disposal, manufacturers can achieve substantial cost savings in their operational expenditure. The high purity of the intermediate acyl chloride, achieved through the solid phosgene route, reduces the load on downstream purification steps, further lowering solvent and energy consumption. Additionally, the ability to recycle the filtrate from the chlorination step for subsequent batches contributes to a more circular and cost-effective material flow, minimizing raw material wastage.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including the specific unsaturated nitriles and standard solvents like dichloromethane or acetonitrile, are commercially available and do not suffer from the supply constraints often seen with controlled precursors like cyanides. This accessibility ensures a stable supply chain, reducing the risk of production stoppages due to regulatory delays in sourcing hazardous chemicals. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in utility supplies, enhancing the overall reliability of the manufacturing schedule and ensuring consistent delivery to customers.
- Scalability and Environmental Compliance: From a scalability perspective, the process is inherently safer and easier to manage at large volumes. The absence of explosive azides or highly toxic cyanides simplifies the safety case for plant expansion, allowing for faster regulatory approval of new production lines. The green nature of the post-treatment, involving simple acidification and filtration, aligns perfectly with modern sustainability goals, making the final product more attractive to environmentally conscious end-users. This compliance advantage future-proofs the supply chain against tightening environmental legislation, securing long-term market access for the manufactured herbicide.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented mesotrione synthesis route. These answers are derived directly from the experimental data and technical specifications provided in CN113233998B, offering clarity on the practical aspects of adopting this technology.
Q: Why are unsaturated nitriles preferred over traditional cyanide catalysts for mesotrione synthesis?
A: Traditional catalysts like sodium cyanide or acetone cyanohydrin are extremely toxic and create difficult wastewater treatment challenges. The novel unsaturated nitriles described in CN113233998B offer superior reactivity due to the synergistic effect of the double bond, allowing for milder conditions and a greener post-treatment process without compromising yield.
Q: What are the advantages of using solid phosgene for the acyl chlorination step?
A: Using solid phosgene instead of thionyl chloride avoids the generation of sulfur dioxide tail gas, significantly improving environmental compliance. Furthermore, it allows for mild reaction conditions that enhance the purity and yield of the acyl chloride intermediate, and the filtrate can be directly recycled for subsequent batches.
Q: How does this process impact the scalability of mesotrione production?
A: The process simplifies the workflow by eliminating complex purification steps associated with toxic heavy metals or azides. The rearrangement proceeds with high selectivity at moderate temperatures (40-60°C), and the final product is isolated via simple filtration and acidification, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mesotrione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN113233998B for the global agrochemical market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel chemistry are fully realized in a commercial setting. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of validating the high-quality standards required for herbicide intermediates, guaranteeing that every batch meets the exacting demands of international regulatory bodies.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this greener route can optimize your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and efficient supply of high-purity mesotrione.
