Advanced Synthesis of Entecavir Key Intermediates via Titanium-Catalyzed Radical Cyclization
Introduction to Novel Entecavir Intermediate Technology
The global demand for effective antiviral therapies, particularly for Hepatitis B Virus (HBV), continues to drive innovation in pharmaceutical intermediate synthesis. Patent CN103387587A discloses a groundbreaking preparation method for a critical Entecavir intermediate, specifically a methylene cyclopentane compound, which serves as the chiral core for the final active pharmaceutical ingredient. This technology represents a significant leap forward in process chemistry, addressing long-standing challenges related to stereocontrol and toxic reagent usage. By leveraging a transition metal-centered radical ring closure reaction, the disclosed method offers a streamlined pathway to high-purity intermediates essential for the production of life-saving medications. 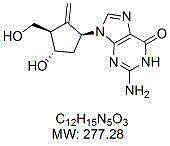
Entecavir, a potent nucleoside analogue, requires a highly specific stereochemical configuration to exhibit its antiviral activity. The synthesis of its key carbocyclic intermediate has historically been a bottleneck due to complex chirality requirements. The patented approach utilizes a precursor compound containing an epoxy and an alkyne functionality, which undergoes a sophisticated cyclization to form the desired five-membered ring system. This strategic disconnection not only simplifies the synthetic route but also aligns with modern green chemistry principles by reducing the reliance on hazardous materials. For procurement and supply chain leaders, understanding this technological shift is crucial for securing reliable entecavir intermediate supplier partnerships that prioritize both quality and sustainability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Entecavir intermediates have been plagued by significant inefficiencies and safety concerns that hinder industrial scalability. Early methods often relied on achiral cyclopentadiene as a starting material, necessitating the introduction of chirality through expensive chiral ligands or difficult chiral separation processes. These approaches typically suffer from low efficiency in establishing the chiral center, leading to substantial material loss and inflated production costs. Furthermore, alternative pathways starting from glucose derivatives frequently employed highly toxic organotin reagents for deoxygenation steps, creating severe environmental and handling hazards. The removal of these tin residues often requires specialized resins or extensive chromatography, complicating the purification process and limiting the achievable purity to levels often below 92% even after repeated column separations.
Another critical drawback of conventional methodologies is the poor selectivity during the coupling of the nucleobase. When reacting intermediate precursors with guanine derivatives, there is a tendency for nucleophilic attack at both the 9-N and 7-N positions. Although 9-N selectivity is generally preferred, the inevitable formation of 7-N isomers creates a complex mixture of steric isomers that are notoriously difficult to separate. Additionally, some prior art methods required the use of silane precursors for hydroxyl groups, which demanded very harsh oxidizing conditions for conversion in later stages. These harsh conditions often resulted in decomposition and low yields, rendering such methods unsuitable for suitability for industrialized production. The cumulative effect of these limitations is a fragile supply chain vulnerable to yield fluctuations and regulatory scrutiny regarding impurity profiles.
The Novel Approach
The innovative method described in the patent data overcomes these historical barriers by employing a transition metal-catalyzed radical cyclization strategy. Instead of struggling with post-synthetic chiral resolution, this approach builds the chiral cyclopentane ring directly from a chiral epoxy-alkyne precursor, designated as Compound 2 in the patent documentation. The core transformation involves the reductive cyclization of this precursor using a titanium species, effectively closing the ring to form the methylene cyclopentane structure, Compound 1. This reaction proceeds with high stereocontrol, leveraging the existing chirality of the starting material to dictate the configuration of the new ring system. By avoiding the use of organotin reagents entirely, the process eliminates a major source of toxicity and simplifies the workup procedure significantly. 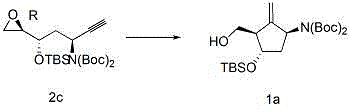
Moreover, this novel route demonstrates superior compatibility with large-scale manufacturing conditions. The reaction can be conducted at moderate temperatures, typically ranging from 20°C to 30°C, which reduces energy consumption and thermal stress on sensitive functional groups. The use of readily available chiral pool starting materials, such as N-Acetyl-D-glucosamine or L-hydroxyproline, ensures a consistent supply of enantiomerically pure inputs. This foundational purity propagates through the synthesis, minimizing the formation of difficult-to-remove isomers and enabling the production of high-purity pharmaceutical intermediates. For stakeholders focused on cost reduction in antiviral manufacturing, this method offers a compelling value proposition by merging high yield potential with a safer, more robust operational profile.
Mechanistic Insights into Titanium-Catalyzed Radical Cyclization
The heart of this synthetic breakthrough lies in the mechanism of the titanium-centered radical ring closure. The reaction initiates with the generation of a low-valent titanium species, often prepared in situ from dichlorobis(cyclopentadienyl)titanium(IV) and a reducing metal such as zinc or manganese. This active titanium complex interacts with the epoxy-alkyne precursor to generate a radical intermediate. The proximity of the alkyne moiety allows for an intramolecular radical addition, which drives the formation of the five-membered carbocyclic ring. This radical cyclization is highly exothermic and kinetically favorable, leading to the rapid construction of the core scaffold. The stereochemical outcome is rigorously controlled by the chiral centers present on the side chains of the precursor, ensuring that the newly formed stereocenters align with the required configuration for Entecavir activity.
Impurity control is inherently built into this mechanistic pathway. Unlike methods that rely on late-stage differentiation of isomers, this route establishes the correct stereochemistry early in the sequence. The radical nature of the cyclization minimizes side reactions that typically arise from ionic pathways, such as elimination or rearrangement, which can lead to complex impurity profiles. Furthermore, the choice of protecting groups, such as Boc or TBS, is optimized to withstand the reaction conditions while remaining orthogonal for subsequent deprotection steps. This strategic selection prevents premature degradation or unwanted side reactions at the hydroxyl or amine functionalities. The result is a cleaner reaction mixture that requires less intensive purification, directly translating to higher overall recovery rates and reduced solvent consumption in the downstream processing stages.
How to Synthesize Methylene Cyclopentane Compound Efficiently
The synthesis of the target methylene cyclopentane compound involves a meticulously designed sequence of transformations that prioritize yield and purity. The process begins with the protection of chiral starting materials to mask reactive hydroxyl groups, followed by the installation of the epoxy and alkyne functionalities required for the cyclization. Once the precursor is assembled, the key ring-closing step is executed under inert atmosphere conditions to prevent oxidation of the sensitive titanium catalyst. Detailed standard operating procedures for each step, including precise stoichiometry, temperature controls, and workup protocols, are essential for replicating the high performance reported in the patent literature.
- Start with chiral pool materials like N-Acetyl-D-glucosamine or L-hydroxyproline and perform selective hydroxyl protection to establish the necessary stereochemical framework.
- Convert the protected sugar or proline derivative into an epoxy-alkyne precursor through activation of hydroxyl groups and subsequent elimination or alkynylation reactions.
- Execute the key ring-closing step using a transition metal catalyst such as dichlorobis(cyclopentadienyl)titanium(IV) reduced in situ to induce radical cyclization, forming the methylene cyclopentane core.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced synthetic route offers profound commercial benefits for organizations managing the supply of antiviral ingredients. The elimination of toxic organotin reagents not only enhances workplace safety but also drastically reduces the costs associated with hazardous waste disposal and environmental compliance. Traditional methods often incur hidden expenses related to the removal of heavy metal traces, which this new technology effectively bypasses. Furthermore, the reliance on abundant chiral pool starting materials mitigates the risk of supply disruptions associated with specialized chiral ligands or rare catalysts. This stability is critical for maintaining continuous production schedules and meeting the rigorous delivery timelines expected by global pharmaceutical partners.
- Cost Reduction in Manufacturing: The streamlined nature of the titanium-catalyzed process leads to substantial cost savings by reducing the number of purification steps required. By avoiding the use of expensive chiral ligands and toxic tin reagents, the raw material costs are significantly lowered. Additionally, the milder reaction conditions decrease energy consumption and extend the lifespan of processing equipment. The higher selectivity of the reaction minimizes the loss of valuable intermediates to side products, thereby improving the overall mass balance and economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Utilizing commercially available starting materials like glucose derivatives or proline ensures a robust and resilient supply chain. These feedstocks are produced on a massive scale for various industries, guaranteeing consistent availability and price stability. The simplified synthetic route reduces the dependency on custom-synthesized reagents that may have long lead times. This reliability allows procurement managers to forecast demand more accurately and secure long-term contracts with confidence, knowing that the production capacity is not constrained by niche raw material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The reaction parameters are well-suited for large reactor vessels, and the absence of highly exothermic or hazardous steps facilitates safe operation at multi-ton scales. From an environmental perspective, the reduction in toxic waste generation aligns with increasingly stringent global regulations on pharmaceutical manufacturing. This compliance advantage reduces the regulatory burden on manufacturers and enhances the marketability of the final product to environmentally conscious clients and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Entecavir intermediates using this patented methodology. The answers are derived from the specific technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is vital for evaluating the feasibility of integrating this technology into existing production frameworks.
Q: What are the primary advantages of the titanium-catalyzed route over traditional organotin methods?
A: The titanium-catalyzed radical cyclization eliminates the need for highly toxic organotin reagents used in traditional deoxygenation steps. This significantly improves the environmental profile of the synthesis and simplifies downstream purification by removing heavy metal residues, thereby enhancing the overall safety and regulatory compliance of the manufacturing process.
Q: How does this method address the issue of stereoisomer contamination?
A: By utilizing chiral pool starting materials such as N-Acetyl-D-glucosamine or L-hydroxyproline, the stereochemistry is inherently controlled from the beginning of the synthesis. This avoids the low-efficiency chiral separation or expensive chiral ligand strategies required when starting from achiral cyclopentadiene, resulting in a product with superior optical purity and reduced isomeric impurities.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process operates under relatively mild reaction conditions, typically between 20°C and 30°C, and utilizes reagents that are manageable on an industrial scale. The avoidance of harsh oxidizing conditions and the use of robust protection group strategies facilitate scalability, making it a viable option for cost reduction in antiviral manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entecavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and production of antiviral therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trials and full-scale market supply. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity and assay. Our expertise in transition metal catalysis and chiral synthesis positions us as a leader in the field, capable of executing complex chemistries with precision and consistency.
We invite you to collaborate with us to leverage this advanced synthetic technology for your Entecavir projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments that demonstrate how our optimized processes can enhance your supply chain efficiency. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable entecavir intermediate supplier dedicated to driving innovation and value in the pharmaceutical industry.
