Advanced [2.2]Paracyclophane Organophosphorus Ligands For High Efficiency Catalysis
Advanced [2.2]Paracyclophane Organophosphorus Ligands For High Efficiency Catalysis
The landscape of asymmetric catalysis is continuously evolving, driven by the relentless demand for higher enantioselectivity and broader substrate tolerance in the synthesis of complex pharmaceutical intermediates. A significant breakthrough in this domain is documented in patent CN109666044B, which introduces a novel class of organophosphorus compounds built upon a rigid [2.2]paracyclophane alkyl skeleton. Unlike traditional ligands that may suffer from conformational flexibility or limited steric bulk, these new structures leverage the inherent planar chirality and structural stability of the paracyclophane core. This innovation provides a powerful toolkit for chemists aiming to optimize metal-catalyzed additions, hydrogenations, and couplings, offering a reliable pathway to access optically active molecules with industrial relevance.
For procurement managers and supply chain directors, the implications of this technology extend beyond mere academic interest. The synthesis described utilizes cost-effective and readily available starting materials, specifically enantiomers or racemates of 4,12-dihalo[2.2]paracyclophane. This foundational accessibility addresses a critical bottleneck in the supply of high-performance chiral ligands. By establishing a robust manufacturing protocol for these advanced organophosphorus compounds, we enable the production of high-purity pharmaceutical intermediates that meet the stringent quality standards required by global regulatory bodies. The ability to source such specialized catalysts reliably is paramount for maintaining continuity in the production of active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral ligands has faced significant hurdles regarding universality and adaptability. Many established ligand families, while effective for specific transformations, often exhibit high dependence on particular reaction substrates, limiting their utility across diverse synthetic pathways. Furthermore, the synthesis of complex chiral backbones frequently involves multi-step sequences with low overall yields or requires expensive chiral pool starting materials that are subject to market volatility. In industrial settings, these factors translate to increased costs and extended lead times. Additionally, conventional ligands may lack the necessary rigidity to maintain a consistent chiral environment under varying reaction conditions, leading to inconsistent enantiomeric excess (ee) values that complicate downstream purification processes.
The Novel Approach
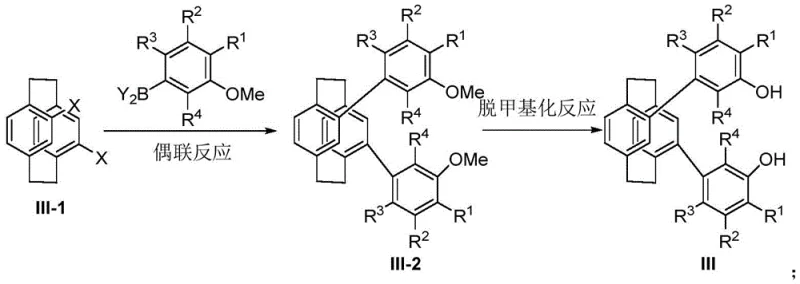
The methodology presented in the patent overcomes these challenges by introducing a modular synthetic strategy centered on the [2.2]paracyclophane framework. This approach allows for the systematic variation of substituents (R1-R4) on the aromatic rings, enabling fine-tuning of the electronic and steric properties of the ligand without altering the core chiral scaffold. The synthetic route is notably concise, proceeding through a key diol intermediate (Formula III) that serves as a versatile precursor for phosphonites, phosphites, phosphoramidites, and phosphoric acids. This modularity means that a single intermediate can be diversified into a library of catalysts, drastically reducing the time and resources needed for ligand screening. Moreover, the rigid skeleton ensures that the chiral information is effectively transferred to the substrate, promising high selectivity even in challenging transformations.
Mechanistic Insights into [2.2]Paracyclophane Functionalization
The construction of these sophisticated molecules begins with a palladium-catalyzed Suzuki coupling reaction, a cornerstone of modern organic synthesis. In this critical step, 4,12-dihalo[2.2]paracyclophane reacts with substituted arylboronic acids in the presence of a base such as potassium carbonate or cesium fluoride. The choice of solvent system, ranging from toluene/ethanol/water mixtures to dioxane/water, is optimized to ensure solubility and reaction efficiency. Following the coupling, a demethylation step utilizing reagents like boron tribromide converts the methoxy groups into free hydroxyl groups. This transformation is pivotal as it generates the nucleophilic sites necessary for subsequent phosphorylation. The precision required in these steps underscores the importance of rigorous process control to minimize impurities and maximize the yield of the key intermediate.
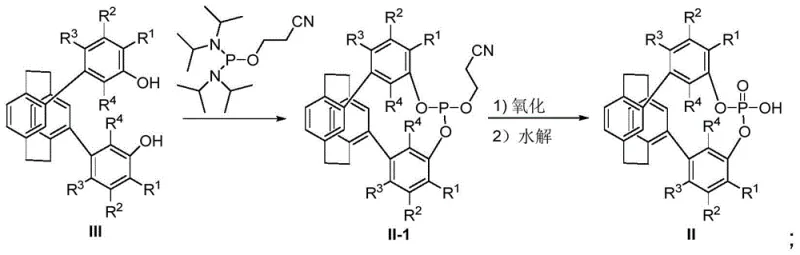
Following the formation of the diol intermediate, the installation of the phosphorus moiety is achieved through nucleophilic substitution. For the synthesis of phosphoric acids (Formula II), the diol reacts with reagents such as bis(diisopropylamino)(2-cyanoethoxy)phosphine. This is followed by a two-stage oxidation and hydrolysis sequence. First, tert-butyl hydroperoxide oxidizes the trivalent phosphorus to the pentavalent state. Subsequently, hydrolysis using a base like DBU removes the protecting group to reveal the free phosphoric acid. This specific pathway is advantageous because it avoids the use of highly moisture-sensitive phosphorus chlorides in the final steps, thereby enhancing the operational safety and scalability of the process. The resulting compounds possess a defined P-O-H acidic proton, which is essential for their function as Brønsted acid catalysts in asymmetric organocatalysis.
How to Synthesize [2.2]Paracyclophane Organophosphorus Compounds Efficiently
The synthesis of these high-value ligands follows a logical progression designed to maximize yield and purity while minimizing waste. The process initiates with the assembly of the carbon skeleton, followed by functional group manipulation to install the reactive phosphorus center. Each stage requires careful monitoring of reaction parameters such as temperature and stoichiometry to ensure the integrity of the chiral backbone is maintained. For detailed operational protocols including specific molar ratios, solvent volumes, and workup procedures, please refer to the standardized synthesis guide below.
- Perform a palladium-catalyzed Suzuki coupling reaction between 4,12-dihalo[2.2]paracyclophane and substituted arylboronic acids to construct the functionalized skeleton.
- Execute a demethylation reaction using boron tribromide or similar reagents to convert methoxy groups into hydroxyl groups, yielding the key diol intermediate.
- React the diol intermediate with phosphorus reagents such as bis(diisopropylamino)phosphine followed by oxidation and hydrolysis to finalize the organophosphorus structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this [2.2]paracyclophane-based technology offers substantial strategic benefits for chemical manufacturing operations. The primary advantage lies in the simplification of the supply chain for chiral catalysts. By utilizing a common intermediate that can be derivatized into multiple ligand types, manufacturers can reduce inventory complexity and mitigate the risk of stockouts for specific catalyst variants. Furthermore, the reliance on standard coupling chemistry means that production can be easily scaled using existing infrastructure without the need for specialized equipment. This flexibility translates directly into cost reduction in fine chemical manufacturing, as it lowers the barrier to entry for producing complex chiral building blocks.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive chiral resolution steps often associated with traditional ligand synthesis, as the chirality is inherent in the commercially available [2.2]paracyclophane starting material. This intrinsic chirality significantly lowers the raw material costs and reduces the number of processing steps required. Additionally, the high yields reported in the patent examples suggest that material throughput is optimized, further driving down the cost per gram of the final catalyst. By streamlining the synthesis, companies can achieve significant economic efficiencies without compromising on the quality of the catalytic performance.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, particularly the halogenated paracyclophanes and common arylboronic acids, are produced by multiple suppliers globally, ensuring a stable and competitive supply base. This diversification reduces dependency on single-source vendors and protects against market fluctuations. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, also means that production is less susceptible to minor variations in utility supply or environmental conditions. Consequently, lead times for high-purity organophosphorus compounds can be consistently met, supporting just-in-time manufacturing models for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The processes described utilize standard organic solvents and reagents that are well-understood in terms of waste management and recycling. The avoidance of heavy metal contaminants in the final ligand structure simplifies the purification process, reducing the burden on wastewater treatment facilities. Moreover, the high atom economy of the coupling reactions aligns with green chemistry principles, minimizing the generation of hazardous byproducts. This environmental compatibility facilitates easier regulatory approval for large-scale production facilities, ensuring long-term operational sustainability and compliance with increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the application and handling of these novel organophosphorus compounds. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What distinguishes the [2.2]paracyclophane skeleton from traditional ligand backbones?
A: The [2.2]paracyclophane skeleton offers exceptional rigidity and a unique planar chirality that differs from axial or central chirality found in BINAP or phosphine ligands. This structural rigidity creates a well-defined chiral environment around the metal center, often leading to superior enantioselectivity and broader substrate scope in asymmetric transformations.
Q: Can these organophosphorus compounds be utilized in gold-catalyzed reactions?
A: Yes, the patent explicitly demonstrates the formation of stable gold complexes using these ligands. These gold complexes have been successfully applied in cycloisomerization reactions, proving their versatility beyond traditional transition metals like rhodium or palladium.
Q: Is the synthetic route scalable for commercial production?
A: The synthesis relies on widely available starting materials like 4,12-dihalo[2.2]paracyclophane and utilizes standard cross-coupling and substitution chemistry. The absence of exotic reagents and the use of robust reaction conditions suggest high potential for scale-up from laboratory to multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organophosphorus Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral ligands play in accelerating drug discovery and process development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Whether you require custom synthesis of specific derivatives or bulk supply of standard catalog items, our infrastructure is designed to support your most demanding projects.
We invite you to collaborate with us to explore how these [2.2]paracyclophane-based catalysts can enhance your synthetic workflows. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions about integrating these high-performance materials into your supply chain.
