Revolutionizing Ezetimibe Production: A Technical Deep Dive into Streamlined Synthetic Pathways
Revolutionizing Ezetimibe Production: A Technical Deep Dive into Streamlined Synthetic Pathways
The pharmaceutical industry is constantly seeking more efficient routes for the production of high-value active pharmaceutical ingredients (APIs), and the cholesterol absorption inhibitor Ezetimibe is no exception. A recent technological breakthrough detailed in patent CN115850144A introduces a significantly optimized preparation process that addresses long-standing inefficiencies in synthetic design. This innovation is particularly relevant for R&D directors and procurement managers looking to enhance supply chain resilience while maintaining stringent purity standards. By fundamentally rethinking the reaction sequence, specifically regarding the handling of key intermediates, this new methodology offers a compelling case for adoption in large-scale manufacturing environments where margin optimization is critical.
The core of this advancement lies in the strategic elimination of redundant protection and deprotection cycles that have historically plagued Ezetimibe synthesis. Traditional pathways often require the isolation of stable intermediates, which, while safe, introduce significant yield losses due to mechanical transfer and purification steps. The new process leverages a telescoped approach where the reaction proceeds from raw materials directly through to the final cyclization and deprotection phases with minimal intervention. This not only reduces the overall processing time but also minimizes the consumption of auxiliary chemicals and solvents, presenting a clear pathway for cost reduction in API manufacturing without compromising the structural integrity of the final molecule.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ezetimibe has been burdened by a multi-step protocol that necessitates the isolation of Intermediate 2, a chemically stable but operationally cumbersome species. As illustrated in prior art such as CN112441959A, the conventional route involves reacting Raw Material A and Raw Material B to form an initial adduct, which must then be quenched, extracted, purified, and dried before proceeding to the next stage. This isolation step is particularly detrimental because it requires the use of additional solvents for recrystallization, such as isopropanol, and generates substantial waste streams. Furthermore, the subsequent conversion of Intermediate 2 to Ezetimibe requires another round of silylation protection followed by deprotection, adding layers of complexity that increase the risk of impurity formation and lower the overall molar yield.
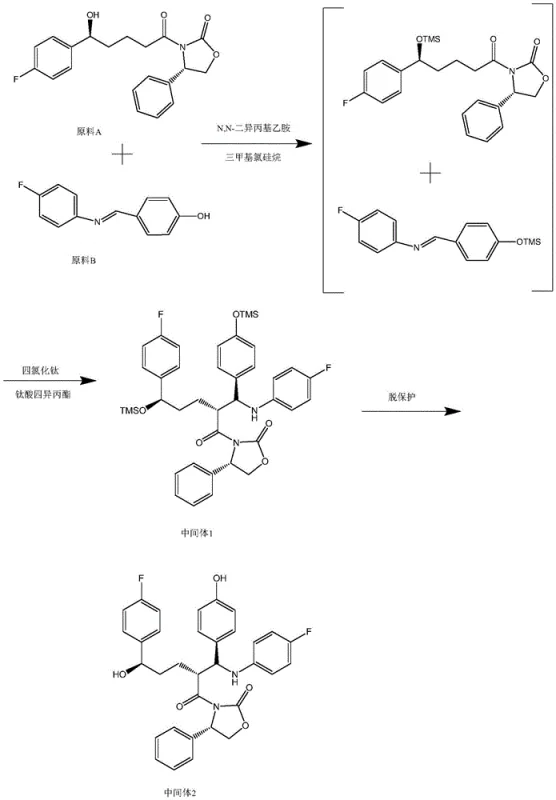
The operational drawbacks of the conventional method extend beyond mere yield loss. The requirement to handle Intermediate 2 as a solid introduces potential variability in particle size and dissolution rates during the subsequent reaction steps, which can lead to inconsistent batch quality. Additionally, the repeated use of harsh quenching agents and multiple aqueous workups increases the load on wastewater treatment facilities and extends the production cycle time significantly. For a reliable ezetimibe intermediate supplier, these inefficiencies translate directly into higher production costs and longer lead times, making the conventional route less attractive in a competitive market where speed-to-market and cost-efficiency are paramount drivers of success.
The Novel Approach
In stark contrast, the novel approach outlined in the present patent data streamlines the entire synthesis into a more cohesive and fluid operation. Instead of isolating Intermediate 2, the process maintains the reaction mixture in a liquid state, transitioning directly from the formation of Intermediate 1 to the final cyclization and deprotection steps. This is achieved by carefully controlling the reaction environment, specifically by managing the addition of reagents like N,O-bis(trimethylsilyl)acetamide (BSA) and tetrabutylammonium fluoride (TBAF) directly to the filtrate. By bypassing the solid-state isolation of the intermediate, the process effectively 'telescopes' multiple chemical transformations into a single continuous flow, thereby preserving the momentum of the reaction and minimizing material hold-up.
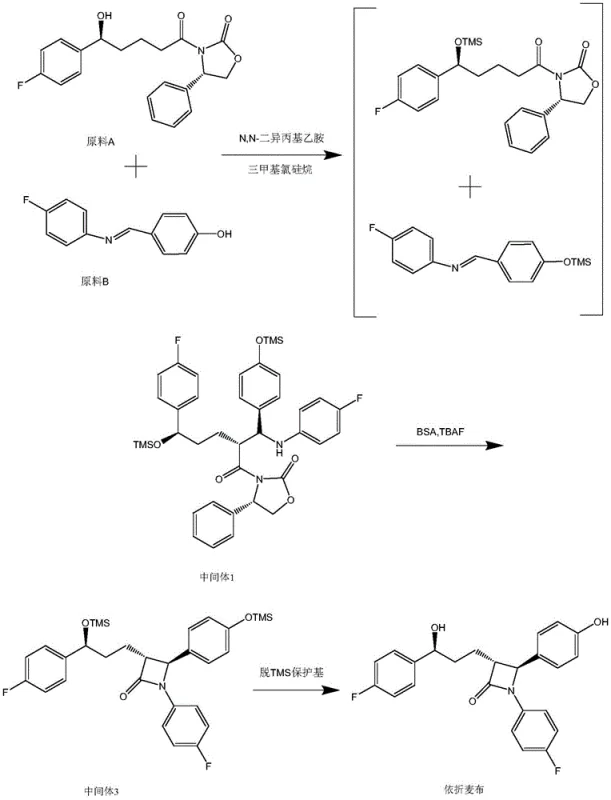
The benefits of this novel approach are quantifiable and significant. Data indicates that the yield of Ezetimibe prepared via this optimized route is approximately 15-20% higher than that achieved through the conventional isolation method. This improvement is not merely a result of better chemistry but is fundamentally driven by the reduction of unit operations. Fewer filtration, drying, and transfer steps mean less product is lost to equipment surfaces or mother liquors. Moreover, the simplified workflow reduces the demand on reactor capacity, allowing manufacturers to produce more batches in the same timeframe, which is a crucial advantage for enhancing supply chain reliability and meeting the growing global demand for cardiovascular medications.
Mechanistic Insights into TiCl4-Catalyzed Cyclization and Silylation
The success of this optimized synthesis relies heavily on the precise orchestration of Lewis acid catalysis and silyl protection strategies. The reaction initiates with the activation of the carbonyl group in Raw Material A by titanium tetrachloride (TiCl4), a potent Lewis acid that facilitates the nucleophilic attack by the imine nitrogen of Raw Material B. This step is critical for establishing the stereochemistry of the azetidinone ring, which is essential for the biological activity of Ezetimibe. The presence of trimethylchlorosilane (TMSCl) serves to protect the hydroxyl groups in situ, preventing unwanted side reactions such as polymerization or elimination that could occur under the acidic conditions generated by TiCl4. The careful addition of glacial acetic acid further modulates the acidity of the medium, ensuring that the reaction proceeds with high selectivity towards the desired (3R, 4S) configuration.
Following the initial coupling, the transition to the final product involves a sophisticated deprotection sequence mediated by fluoride ions. The addition of tetrabutylammonium fluoride (TBAF) triggers the cleavage of the trimethylsilyl (TMS) ether bonds, revealing the free hydroxyl groups necessary for the final structure. Simultaneously, N,O-bis(trimethylsilyl)acetamide (BSA) acts as a silylating agent that assists in the cyclization process by activating the amide nitrogen for nucleophilic attack on the ketone carbonyl. This dual role of silyl reagents—first as protectors and then as activators—is a hallmark of modern fine chemical synthesis. The mechanism ensures that the sensitive beta-lactam ring is formed under mild conditions, preserving the integrity of the fluorophenyl moieties which are susceptible to nucleophilic aromatic substitution under harsher conditions.
How to Synthesize Ezetimibe Efficiently
Implementing this optimized synthesis requires strict adherence to temperature controls and reagent stoichiometry to maximize yield and purity. The process begins with the preparation of a feed liquid containing Raw Material A and Raw Material B in dichloromethane, cooled to a range of -30°C to -10°C. Sequential addition of bases and silylating agents must be performed with precision to maintain the reaction exotherm within safe limits. Following the formation of the intermediate feed liquid, the workup involves a gentle aqueous quench using EDTA to chelate residual titanium species, followed by a direct treatment with BSA and TBAF. The final crystallization is induced by the addition of dilute sulfuric acid to an isopropanol-water mixture, which promotes the formation of high-purity crystals suitable for pharmaceutical applications.
- Mix Raw Material A and Raw Material B in dichloromethane at -30°C to -10°C, then sequentially add DIPEA, TMSCl, TiCl4, and glacial acetic acid.
- Quench the reaction with EDTA solution, separate the organic layer, and wash with sodium carbonate and water to obtain Intermediate 1 feed liquid.
- Treat the filtrate with BSA and TBAF, concentrate, and crystallize using isopropanol/water/sulfuric acid mixture to isolate pure Ezetimibe.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this streamlined Ezetimibe preparation process offers tangible strategic advantages that go beyond simple chemistry. The primary benefit is the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures (OPEX). By eliminating the isolation and purification of Intermediate 2, manufacturers can significantly reduce the consumption of solvents, filtration media, and energy required for drying operations. This reduction in resource intensity translates into a lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing in the global market while maintaining healthy profit margins. Furthermore, the reduced complexity minimizes the risk of batch failures, ensuring a more consistent and reliable supply of high-purity intermediates.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps removes the need for extensive solvent recovery and solid handling equipment, leading to substantial capital expenditure (CAPEX) savings and lower utility costs. The process inherently reduces the volume of chemical waste generated per kilogram of product, which lowers disposal costs and environmental compliance burdens. Additionally, the higher overall yield means that less raw material is required to produce the same amount of final API, optimizing the utilization of expensive starting materials and reagents.
- Enhanced Supply Chain Reliability: A shorter synthesis route with fewer unit operations inherently reduces the total production cycle time, enabling faster turnaround from order to delivery. This agility is crucial for responding to sudden spikes in demand or supply disruptions in the raw material market. The robustness of the new process, which tolerates slight variations in reaction conditions better than the multi-step isolation method, ensures consistent quality across different production batches, thereby reducing the risk of supply interruptions due to out-of-specification results.
- Scalability and Environmental Compliance: The use of common solvents like dichloromethane and isopropanol, combined with the avoidance of exotic catalysts, makes this process highly scalable from pilot plant to commercial production volumes. The simplified waste stream, characterized by lower levels of heavy metal residues and organic byproducts, facilitates easier treatment and compliance with increasingly stringent environmental regulations. This 'green chemistry' aspect not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optimized Ezetimibe synthesis. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a transparent view into the feasibility and benefits of the new technology. Understanding these details is essential for technical teams evaluating the potential for technology transfer or licensing.
Q: How does the new process improve yield compared to conventional methods?
A: The novel process described in CN115850144A eliminates the isolation and purification of Intermediate 2, reducing material loss and operational complexity, resulting in a yield increase of approximately 15-20% over previous methods.
Q: What are the critical control parameters for the Lewis acid catalysis step?
A: Temperature control between -30°C and -10°C during the addition of titanium tetrachloride and trimethylchlorosilane is critical to prevent side reactions and ensure high stereoselectivity.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the simplification of unit operations and the use of standard reagents like dichloromethane and isopropanol make this route highly amenable to commercial scale-up with reduced equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay ahead in the competitive pharmaceutical landscape. Our team of expert chemists has extensively evaluated the process described in CN115850144A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Ezetimibe intermediate we supply meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized process can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this streamlined route for your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify the superior quality and consistency of our products. Let us help you secure a reliable source of high-purity Ezetimibe intermediates that drives both your scientific success and commercial growth.
