Scalable Production of Polysubstituted Isoquinolinones via Novel Copper-Catalyzed Cyclization for Commercial Applications
Scalable Production of Polysubstituted Isoquinolinones via Novel Copper-Catalyzed Cyclization for Commercial Applications
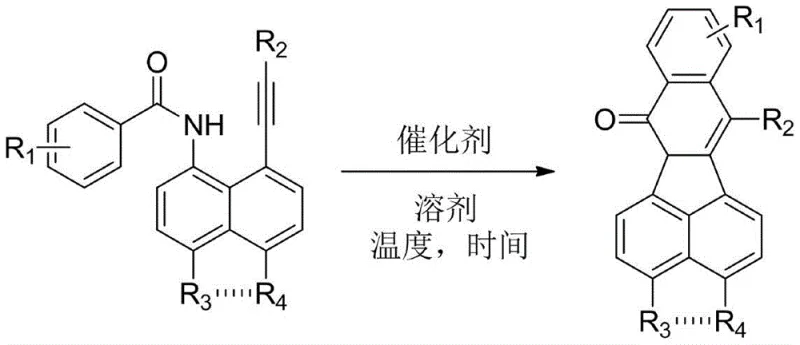
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for safer, more efficient, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN111410656A, which discloses a robust preparation method for isoquinolinone derivatives. These heterocyclic scaffolds are not merely academic curiosities; they serve as critical structural units in a vast array of bioactive molecules, including potent anti-inflammatory, anti-tumor, and vasodilatory agents. The patent introduces a transformative "one-pot" synthesis strategy that leverages copper catalysis to construct the isoquinolinone core from 8-alkynyl-1-aroyl naphthylamine precursors. This technological breakthrough addresses long-standing challenges in heterocycle synthesis, offering a pathway that combines high reaction selectivity with operational simplicity. For R&D directors and procurement specialists alike, understanding the nuances of this method is essential for optimizing supply chains and reducing the cost of goods sold (COGS) for complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isoquinolinone skeleton has been fraught with significant chemical and operational hurdles that impede efficient commercial manufacturing. Traditional methodologies often rely on the amination of isocoumarins, a process that necessitates the pre-preparation of specific substrates and the use of refluxing acetic acid, leading to energy-intensive operations and potential corrosion issues. Alternatively, classical approaches involving double lithiation reactions utilize highly hazardous reagents such as sec-butyllithium. These pyrophoric materials demand stringent cryogenic conditions and specialized handling equipment, introducing substantial safety risks and inflating capital expenditure for manufacturing facilities. Furthermore, other conventional routes, such as the condensation of 2-bromomethylbenzonitrile with phthalic anhydride, suffer from severe substrate limitations, often failing with electron-rich systems and resulting in poor reaction efficiency. These legacy methods collectively contribute to extended lead times, elevated waste generation, and inconsistent product quality, creating bottlenecks in the supply of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in patent CN111410656A presents a streamlined and versatile solution. By employing a transition metal-catalyzed cyclization strategy, specifically utilizing copper salts, the invention enables the direct conversion of readily available 8-alkynyl-1-aroyl naphthylamine compounds into polysubstituted isoquinolinones. This approach eliminates the need for dangerous organolithium reagents and harsh acidic or basic conditions, operating instead within a moderate temperature range of 80-150°C. The reaction exhibits exceptional chemoselectivity, avoiding the formation of complex mixtures that often plague alkyne-based cyclizations. Moreover, the "one-pot" nature of the synthesis drastically simplifies the workflow by removing the necessity for isolating unstable intermediates. This paradigm shift not only enhances the safety profile of the manufacturing process but also significantly improves the overall atom economy and throughput, making it an ideal candidate for the cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into CuCN-Catalyzed Cyclization
The core of this technological innovation lies in the efficient activation of the alkyne moiety by the copper catalyst, facilitating an intramolecular nucleophilic attack that closes the ring to form the isoquinolinone structure. The patent specifies a variety of effective copper sources, including CuCN, Cu(OAc)2, and CuI, with CuCN emerging as the preferred catalyst due to its superior performance in promoting the cyclization. The mechanism likely involves the coordination of the copper species to the alkyne triple bond, increasing its electrophilicity and enabling the proximal amide oxygen or nitrogen to attack, followed by proton transfer and aromatization. This catalytic cycle operates effectively in polar aprotic solvents such as N,N-dimethylformamide (DMF), which stabilize the charged intermediates and ensure complete dissolution of the reactants. The robustness of this catalytic system allows for a broad tolerance of functional groups, including halogens, methyl groups, and heterocycles, without compromising the integrity of the final product.
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic thermal cyclizations. The presence of the catalyst lowers the activation energy barrier, allowing the reaction to proceed at lower temperatures where competing decomposition pathways are minimized. The high selectivity observed in the patent examples, with yields reaching up to 84% in optimized conditions, suggests that side reactions such as polymerization of the alkyne or hydrolysis of the amide are effectively suppressed. This level of control is paramount for R&D teams focused on purity specifications, as it reduces the burden on downstream purification processes like column chromatography or recrystallization. Consequently, the final isoquinolinone derivatives exhibit cleaner impurity profiles, which is a critical factor for regulatory compliance in the production of active pharmaceutical ingredients (APIs).
How to Synthesize Polysubstituted Isoquinolinones Efficiently
The practical implementation of this synthesis route is designed for ease of execution, requiring standard laboratory or plant equipment without the need for exotic hardware. The process begins with the precise weighing of the 8-alkynyl-1-aroyl naphthylamine substrate and the copper catalyst, typically in a molar ratio ranging from 1:0.1 to 1:0.2, although ratios up to 1:1.5 are permissible depending on the specific substrate reactivity. These components are introduced into a reaction vessel, followed by the addition of a suitable aprotic solvent such as DMF, toluene, or DMSO, ensuring the complete dissolution of the starting materials to create a homogeneous reaction mixture. The detailed standardized synthesis steps, including specific workup procedures and purification protocols validated for commercial viability, are outlined below.
- Weigh 8-alkynyl-1-aroyl naphthylamine compounds and catalyst (e.g., CuCN) in a molar ratio of 1: 0.1-1.5 and place them in a reaction vessel.
- Add an aprotic solvent such as N,N-dimethylformamide (DMF) until the starting material is completely dissolved.
- Heat the reaction mixture to 80-150°C (optimally 120°C) and stir for 0-36 hours, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift from precious metal catalysts or hazardous organometallic reagents to abundant copper salts represents a fundamental optimization of the raw material cost structure. By utilizing catalysts like CuCN, which are significantly less expensive than palladium or rhodium alternatives, manufacturers can achieve substantial cost savings without sacrificing reaction efficiency. Furthermore, the elimination of cryogenic conditions and the use of common industrial solvents reduce the energy consumption and infrastructure requirements of the production facility. This simplification of the process parameters directly correlates to a more resilient supply chain, as the reliance on specialized, hard-to-source reagents is minimized, thereby mitigating the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of high-cost reagents with economical copper catalysts and the reduction of processing steps. Traditional lithiation methods require expensive reagents and rigorous temperature control, whereas this method operates at moderate temperatures with catalytic loading. The "one-pot" design eliminates the need for intermediate isolation and purification, which are often the most labor and time-intensive stages of chemical manufacturing. By consolidating the synthesis into a single reaction vessel, labor costs are reduced, and solvent usage is optimized, leading to a leaner and more cost-effective production model that enhances overall profit margins for high-volume intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commercially available and stable starting materials. The 8-alkynyl-1-aroyl naphthylamine precursors are synthesized from readily accessible building blocks, ensuring a steady flow of raw materials even during market fluctuations. Additionally, the robustness of the copper catalyst means that the process is less sensitive to minor variations in reagent quality or environmental conditions, resulting in consistent batch-to-batch reproducibility. This reliability allows for more accurate forecasting and inventory management, reducing the need for excessive safety stock and freeing up working capital for other strategic investments within the organization.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this methodology is inherently scalable due to its simple thermal requirements and lack of hazardous gas evolution. The absence of pyrophoric reagents simplifies the safety protocols required for large-scale reactors, lowering the barrier for technology transfer to manufacturing sites. From an environmental standpoint, the use of copper instead of heavy metals like palladium simplifies waste treatment and metal residue removal, aiding in compliance with increasingly stringent environmental regulations. The high atom economy and reduced solvent waste further contribute to a greener manufacturing footprint, aligning with global sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isoquinolinone synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of this copper-catalyzed method over traditional lithiation routes?
A: Unlike traditional methods requiring dangerous sec-butyllithium reagents and cryogenic conditions, this novel approach utilizes stable copper catalysts at moderate temperatures (80-150°C), significantly enhancing operational safety and reducing equipment costs.
Q: What is the substrate scope for this isoquinolinone synthesis?
A: The method demonstrates broad substrate universality, successfully accommodating 8-substituted aromatic hydrocarbon ethynyl, 8-heterocycloethynyl, and 8-alkane ethynyl groups, allowing for diverse functionalization of the isoquinolinone core.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed as a 'one-pot' synthesis using inexpensive catalysts like CuCN and common solvents like DMF, eliminating complex intermediate isolation steps and making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoquinolinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in driving innovation within the pharmaceutical and fine chemical industries. Our team of expert chemists has thoroughly analyzed the potential of the copper-catalyzed cyclization route described in CN111410656A and is prepared to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of isoquinolinone derivatives we deliver meets the highest standards of quality required for downstream drug development.
We invite you to collaborate with us to unlock the full commercial potential of these valuable intermediates. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in catalytic cyclization can optimize your supply chain and accelerate your time to market.
