Scalable Production of Azole Fungicide Intermediates via Novel Metal Exchange Chemistry
Scalable Production of Azole Fungicide Intermediates via Novel Metal Exchange Chemistry
The global demand for high-efficacy, low-toxicity agricultural agents continues to drive innovation in synthetic chemistry, particularly within the realm of azole fungicides. A pivotal advancement in this sector is detailed in patent CN115087642B, which discloses a robust and economically superior method for producing azole derivatives represented by general formula (I). This technology addresses critical bottlenecks in traditional manufacturing, specifically targeting the high costs associated with iodine-based reagents and the environmental burdens of acidic waste streams. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize supply chains for key agrochemical intermediates. The disclosed route leverages a sophisticated metal exchange reaction followed by a streamlined azolization step, offering a pathway that is not only chemically elegant but also commercially viable for large-scale production.
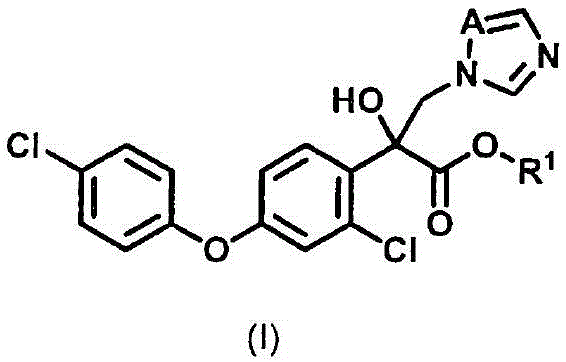
Traditional synthesis pathways for these valuable compounds have long been plagued by inefficiencies that inflate operational expenditures and complicate regulatory compliance. Conventional methods often rely on the oxidation and esterification of acetophenone derivatives, a process that notoriously requires the use of high-priced iodine or methyl iodide. Alternatively, Friedel-Crafts acylation routes generate substantial volumes of strongly acidic waste liquids containing aluminum, necessitating costly neutralization and disposal protocols. Another common approach involves the reaction of organometallic reagents with dialkyl oxalates; however, when alkyllithium reagents are employed, the reaction must be conducted at extremely low temperatures, imposing severe energy demands on the cooling infrastructure. Even the use of standard Grignard reagents in these legacy processes often results in suboptimal yields, further eroding profit margins. These limitations underscore the urgent need for a process redesign that balances chemical efficiency with economic prudence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on legacy synthetic routes creates a fragile supply chain vulnerable to raw material price volatility and stringent environmental regulations. The use of iodine species not only spikes the bill of materials but also introduces handling hazards that require specialized containment and safety measures. Furthermore, the generation of heavy metal-containing acidic waste from Friedel-Crafts reactions poses a significant liability for manufacturers aiming to meet modern green chemistry standards. The thermal inefficiency of cryogenic reactions required for organolithium species adds another layer of operational complexity, limiting the feasible batch sizes and throughput of manufacturing facilities. Consequently, producers relying on these outdated methodologies face diminishing competitiveness in a market that increasingly rewards sustainability and cost-effectiveness.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115087642B introduces a transformative two-step sequence that circumvents these historical challenges. The core innovation lies in the generation of an organometallic reagent from 1-bromo-2-chloro-4-(4-chlorophenoxy)benzene via a metal exchange reaction, which is subsequently reacted with a bromopyruvate derivative to yield a bromohydrin intermediate. This intermediate is then seamlessly converted into the target azole derivative through reaction with imidazole or 1,2,4-triazole salts. This approach completely eliminates the need for expensive iodine reagents and avoids the formation of problematic aluminum waste. By utilizing a Turbo Grignard reagent (iPrMgCl·LiCl), the process operates under much milder temperature conditions compared to organolithium alternatives, significantly reducing energy consumption while maintaining high reaction fidelity.
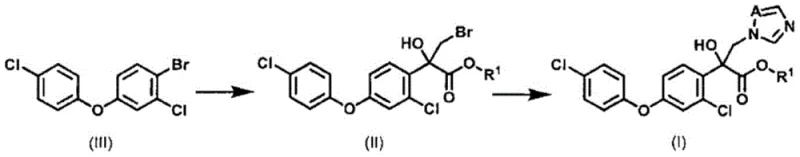
Mechanistic Insights into the Metal Exchange and Azolization Pathway
The chemical elegance of this process is rooted in the precise control of the halogen-magnesium exchange reaction. The use of an isopropyl magnesium chloride-lithium chloride complex facilitates the rapid and selective formation of the aryl magnesium species from the bromo-chloro precursor. This organometallic intermediate is highly reactive yet stable enough to be handled at temperatures ranging from -10°C to 30°C, a window that is far more accessible for industrial reactors than the cryogenic conditions demanded by lithiation. Upon addition of the bromopyruvate derivative, typically methyl 3-bromopyruvate, the nucleophilic attack occurs efficiently at the carbonyl carbon, establishing the crucial carbon-carbon bond and the tertiary alcohol center in a single operation. The presence of the lithium chloride additive is critical here, as it enhances the solubility and reactivity of the magnesium species, ensuring high conversion rates and minimizing the formation of homocoupling by-products.
Following the formation of the bromohydrin derivative, the subsequent azolization step proceeds via a classic nucleophilic substitution mechanism. The bromine atom on the side chain serves as an excellent leaving group, allowing for displacement by the nitrogen nucleophile of the triazole or imidazole ring. Conducting this reaction in polar aprotic solvents like dimethylformamide (DMF) at moderate temperatures (40°C to 80°C) ensures rapid kinetics without compromising the integrity of the sensitive ester or ether functionalities elsewhere in the molecule. Furthermore, the patent details a highly efficient method for synthesizing the key starting material, 1-bromo-2-chloro-4-(4-chlorophenoxy)benzene, via direct bromination of 1-chloro-3-(4-chlorophenoxy)benzene. This precursor synthesis can be performed solvent-free, drastically reducing waste volume, and employs a clever repulping purification strategy to remove positional isomers, ensuring the high purity required for downstream pharmaceutical or agrochemical applications.
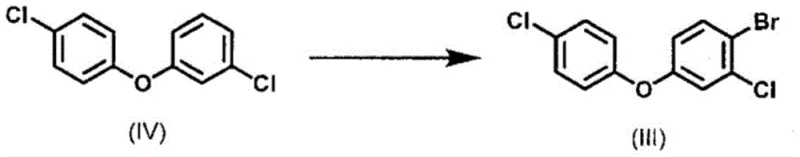
How to Synthesize Azole Derivative (I) Efficiently
The implementation of this synthetic route requires careful attention to reagent stoichiometry and temperature control to maximize yield and purity. The process begins with the activation of the aryl bromide using the Turbo Grignard reagent in tetrahydrofuran, followed by the controlled addition of the bromopyruvate electrophile. Once the bromohydrin intermediate is isolated, it is subjected to nucleophilic substitution with a triazole salt in DMF. The detailed standardized operating procedures, including specific quenching methods, extraction protocols, and purification parameters such as repulping with cold 2-propanol, are critical for reproducing the high success rates reported in the patent literature. Manufacturers looking to adopt this technology should focus on optimizing the workup of the metal exchange step to minimize magnesium salt contamination, which can interfere with the subsequent azolization reaction.
- Perform a metal exchange reaction on 1-bromo-2-chloro-4-(4-chlorophenoxy)benzene using iPrMgCl·LiCl in THF at -10°C to 30°C, then react with methyl 3-bromopyruvate to form the bromohydrin intermediate.
- React the resulting bromohydrin derivative with sodium triazolate in DMF at 40°C to 80°C to effect nucleophilic substitution and yield the final azole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers compelling strategic advantages that extend beyond simple unit cost reductions. By shifting away from iodine-dependent chemistry, organizations can insulate themselves from the volatile pricing trends of halogen commodities and reduce the regulatory burden associated with hazardous reagent storage. The ability to utilize 1-chloro-3-(4-chlorophenoxy)benzene, a commodity chemical available at a fraction of the cost of specialized halogenated benzenes, fundamentally alters the cost structure of the final active ingredient. Moreover, the solvent-free nature of the precursor bromination step aligns perfectly with corporate sustainability goals, reducing the carbon footprint associated with solvent manufacture, recovery, and incineration. This process intensification allows for higher throughput in existing reactor trains without the need for capital-intensive expansion.
- Cost Reduction in Manufacturing: The elimination of expensive iodine and methyl iodide reagents directly lowers the raw material bill, which is often the largest component of COGS in fine chemical synthesis. Additionally, the avoidance of cryogenic conditions reduces utility costs related to refrigeration, while the simplified waste profile decreases expenditure on effluent treatment and hazardous waste disposal. The use of cheaper starting materials further compounds these savings, creating a robust margin buffer against market fluctuations.
- Enhanced Supply Chain Reliability: Sourcing strategies are strengthened by the reliance on widely available bulk chemicals rather than niche, custom-synthesized intermediates. The robustness of the Turbo Grignard chemistry ensures consistent batch-to-bquality, reducing the risk of production delays caused by failed reactions or difficult purifications. Furthermore, the patented recycling method for positional isomers via hydrogenation ensures that raw material utilization is maximized, securing supply continuity even during periods of tight market availability.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as liquid-liquid extraction and crystallization that are well-understood in industrial settings. The reduction in acidic and heavy metal waste simplifies environmental permitting and compliance reporting, facilitating faster approval for new manufacturing lines. The ability to operate at near-ambient temperatures for key steps also enhances process safety, lowering insurance premiums and operational risks associated with exothermic runaways.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this azole derivative synthesis. These insights are derived directly from the experimental data and embodiments described in the patent documentation, providing a clear picture of the process capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements.
Q: How does this new process reduce production costs compared to conventional methods?
A: The novel process eliminates the need for expensive iodine or methyl iodide reagents required in traditional acetophenone oxidation routes. Additionally, it utilizes 1-chloro-3-(4-chlorophenoxy)benzene, a significantly cheaper starting material compared to halogenated benzenes used in prior art.
Q: What is the key advantage of the solvent-free bromination step?
A: Performing the bromination of 1-chloro-3-(4-chlorophenoxy)benzene without a solvent reduces waste generation and simplifies downstream processing. The reaction mixture can be directly concentrated and purified via repulping, avoiding complex solvent recovery systems.
Q: How are positional isomers managed during the synthesis?
A: Positional isomers formed during bromination are effectively removed through repulping washing with cold alcohol solvents like 2-propanol. Furthermore, the wash filtrate containing isomers can be recycled back to the starting material via catalytic hydrogenation, minimizing raw material loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN115087642B for the production of high-value agrochemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this chemistry are fully realized in a manufacturing environment. Our state-of-the-art facilities are equipped to handle the specific requirements of organometallic chemistry, including inert atmosphere processing and precise temperature control, while our rigorous QC labs enforce stringent purity specifications to meet the exacting standards of the global crop protection industry.
We invite forward-thinking partners to collaborate with us on optimizing their supply chains through the adoption of this advanced manufacturing technology. By leveraging our expertise, you can achieve significant operational efficiencies and secure a competitive edge in the market. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Let us provide you with specific COA data and route feasibility assessments to demonstrate how we can support your long-term growth objectives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
