Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Advanced Pd-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
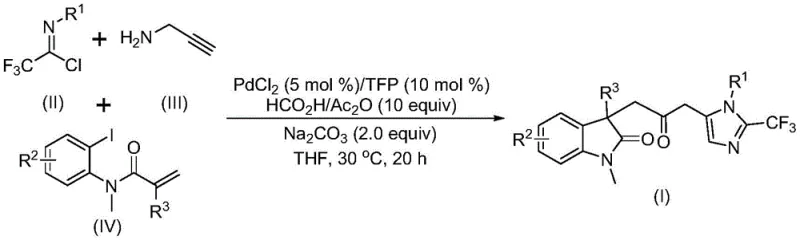
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to complex heterocyclic scaffolds, which serve as critical building blocks for bioactive molecules. Patent CN115353511A introduces a groundbreaking preparation method for carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technology represents a significant leap forward in synthetic efficiency, utilizing a transition metal palladium-catalyzed carbonylation cascade reaction. By employing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, this process eliminates the need for hazardous carbon monoxide gas while maintaining high reaction efficiency. The ability to synthesize diversified substituted double heterocyclic compounds containing both trifluoromethyl and carbonyl functionalities through simple substrate design offers immense value for drug discovery programs aiming to optimize metabolic stability and binding affinity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of biheterocyclic systems has relied on laborious multi-step sequences that often suffer from poor atom economy and harsh reaction conditions. Conventional strategies typically involve the direct coupling of two pre-formed heterocyclic substrates, which requires expensive and sensitive reagents, or oxidative cyclization reactions that generate significant stoichiometric waste. Furthermore, introducing a carbonyl bridge usually necessitates the use of toxic carbon monoxide gas under high pressure, posing severe safety risks and requiring specialized equipment that increases capital expenditure. These legacy methods often struggle with substrate compatibility, limiting the diversity of chemical space that can be explored during lead optimization phases. The cumulative effect of these limitations is a prolonged development timeline and inflated manufacturing costs, creating a bottleneck for the rapid production of high-purity pharmaceutical intermediates needed for clinical trials.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated yet operationally simple multicomponent reaction strategy that constructs the complex biheterocyclic core in a single pot. By leveraging a palladium catalyst system with trifurylphosphine (TFP) as a ligand, the method facilitates a cascade sequence involving Heck-type cyclization and carbonylation without external CO gas. Instead, it employs a safe and inexpensive formic acid/acetic anhydride mixture as an in situ carbon monoxide source. This innovation not only drastically simplifies the operational workflow but also enhances safety profiles by removing high-pressure gas handling requirements. The reaction proceeds under mild conditions at 30°C, demonstrating exceptional functional group tolerance that allows for the incorporation of sensitive moieties like nitro and halogen groups without degradation. This streamlined process transforms the synthesis of these valuable scaffolds from a challenging academic exercise into a robust, industrially viable manufacturing protocol.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis, initiating with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck reaction that generates a divalent alkyl-palladium intermediate, setting the stage for ring closure. Crucially, the subsequent carbonylation step is driven by carbon monoxide released from the decomposition of the formic acid and acetic anhydride additive mixture, forming a reactive acyl-palladium intermediate. Parallel to this metal cycle, a base-promoted intermolecular carbon-nitrogen bond formation occurs between the trifluoroethylimidoyl chloride and propargylamine, yielding a trifluoroacetamidine compound that undergoes isomerization. The convergence of these pathways sees the acyl-palladium intermediate activating the trifluoroacetamidine species, catalyzing a final intramolecular cyclization that delivers the target carbonyl-bridged biheterocyclic compound with high regioselectivity.
Understanding the impurity profile is vital for R&D directors focused on purity specifications, and this mechanism offers inherent controls against common side reactions. The use of sodium carbonate as a mild base ensures that the delicate balance between amidine formation and palladium cycling is maintained, minimizing hydrolysis of the imidoyl chloride starting material. The specific choice of the TFP ligand stabilizes the palladium center throughout the catalytic cycle, preventing premature catalyst deactivation which often leads to incomplete conversion and difficult-to-remove metal residues. Furthermore, the mild temperature of 30°C suppresses thermal decomposition pathways that could generate polymeric byproducts or degrade the sensitive trifluoromethyl group. This precise control over the reaction environment ensures that the crude reaction mixture is cleaner, thereby reducing the burden on downstream purification processes like column chromatography and facilitating the isolation of high-purity intermediates suitable for stringent pharmaceutical applications.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis effectively, operators must adhere to strict stoichiometric ratios and solvent choices as defined in the patent embodiments. The process begins with the preparation of a catalyst solution containing palladium chloride and TFP in an aprotic solvent such as tetrahydrofuran (THF), which has been identified as optimal for solubility and conversion rates. The sequential addition of reagents, particularly the timing of the formic acid/acetic anhydride mixture, is critical to ensure the steady release of carbon monoxide matches the rate of the palladium cycle. Detailed standardized synthesis steps see the guide below for exact procedural parameters.
- Combine palladium chloride catalyst, TFP ligand, sodium carbonate base, and a formic acid/acetic anhydride CO source in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide substrate to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12 to 20 hours, followed by filtration and column chromatography purification to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this patented methodology are profound, primarily driven by the simplification of the raw material portfolio and the elimination of hazardous reagents. The shift away from toxic carbon monoxide gas removes the need for specialized high-pressure reactors and associated safety infrastructure, leading to significant capital expenditure savings and reduced regulatory compliance burdens. Moreover, the reliance on commercially available and inexpensive starting materials like propargylamine and acrylamide derivatives ensures a stable and resilient supply chain, mitigating the risks associated with sourcing custom-synthesized precursors. The robustness of the reaction conditions allows for flexible scheduling and reduces the likelihood of batch failures due to sensitive parameter fluctuations, thereby enhancing overall supply continuity for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are anchored in the use of a palladium catalyst system that operates efficiently at low loading levels, combined with the substitution of expensive or hazardous carbonyl sources with cheap formic acid derivatives. By consolidating multiple bond-forming events into a single one-pot operation, the method significantly reduces solvent consumption, energy usage for heating and cooling, and labor hours compared to traditional stepwise syntheses. The elimination of toxic gas handling also lowers insurance and waste disposal costs, contributing to a leaner overall cost structure. These factors collectively drive down the cost of goods sold (COGS), making the production of these complex biheterocycles economically viable even at early development stages.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the high availability of the core building blocks, which are commodity chemicals rather than bespoke intermediates subject to long lead times. The reaction's tolerance for various functional groups means that a single robust platform can be adapted to produce a wide library of analogues without needing to requalify entirely new synthetic routes for each derivative. This flexibility allows manufacturers to respond rapidly to changing demand signals from R&D departments or clinical partners. Additionally, the simplified workup procedure involving filtration and standard chromatography reduces the dependency on specialized purification equipment, ensuring that production can be maintained across multiple manufacturing sites with varying levels of technological sophistication.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process aligns well with green chemistry principles by avoiding toxic reagents and minimizing waste generation. The ability to scale the reaction to gram levels, as demonstrated in the patent examples, provides a clear pathway to kilogram and ton-scale production without encountering the heat transfer or mixing issues often associated with exothermic cascades. The use of THF as a solvent, while requiring recovery systems, is well-established in the industry, and the absence of heavy metal contaminants in the final product simplifies the validation process for API manufacturing. This combination of scalability and environmental safety makes the technology highly attractive for long-term commercial partnerships focused on sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational boundaries and potential applications of the method for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines.
Q: What are the key advantages of this Pd-catalyzed method over traditional coupling?
A: This method avoids toxic carbon monoxide gas by using a formic acid/acetic anhydride surrogate, operates at mild temperatures (30°C), and achieves high atom economy through a one-pot multicomponent design.
Q: Is this synthesis scalable for industrial production?
A: Yes, the patent explicitly demonstrates scalability to gram-level reactions with simple post-treatment procedures like filtration and silica gel chromatography, indicating strong potential for commercial scale-up.
Q: What is the substrate scope for the R1 and R3 groups?
A: The reaction tolerates a wide range of substituents including alkyl, substituted aryl (methyl, methoxy, halogens, nitro, trifluoromethyl), and benzyl groups, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN115353511A for accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with rigorous safety protocols, and our stringent purity specifications guarantee that every batch meets the highest quality standards required by global regulatory bodies. With our rigorous QC labs and dedicated process chemistry team, we are uniquely positioned to optimize this multicomponent reaction for your specific target molecules, delivering high-purity intermediates that empower your research and commercial success.
We invite you to leverage our technical expertise to evaluate the feasibility of this advanced synthesis for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce overall development costs. Let us be your partner in transforming innovative chemistry into commercial reality.
