Advanced Chiral Copper Catalysis for High-Purity Optically Active Indole Intermediates
Introduction to Breakthrough Indole Synthesis Technology
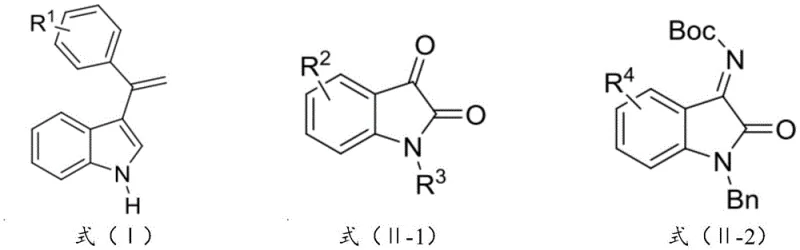
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex chiral scaffolds, particularly those found in bioactive molecules. Patent CN114160206B introduces a transformative technology for the catalytic synthesis of optically active indole compounds, specifically targeting the challenging construction of 3-alkenyl-3-substituted oxindole motifs. These structural units are prevalent in numerous natural products and drug candidates, such as spirotryprostatin B alkaloids which exhibit cytotoxic activity, and potential therapeutics for cancer and inflammation. The core innovation lies in a novel chiral copper-based catalyst system that facilitates a direct asymmetric olefination reaction with unprecedented control over stereochemistry. This technology addresses the long-standing difficulty in achieving high enantioselectivity and diastereoselectivity simultaneously in the functionalization of the C3 position of indoles, offering a robust solution for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric alkenylation of isatin and its analogues has been a formidable challenge in organic synthesis due to the poor reactivity of bare alkenyl groups and the difficulty in controlling stereochemistry at the quaternary carbon center. Prior art, such as the work by Zhou's group in 2010 using cinchona alkaloids or Zhao's group in 2016 utilizing chiral CoI2-bisphosphine complexes, often required harsh conditions, such as elevated temperatures up to 70°C, or suffered from limited substrate scope. Conventional Lewis acid catalysts frequently struggle to differentiate between enantiotopic faces of the substrate effectively, leading to mediocre enantiomeric excess and significant formation of unwanted diastereomers. Furthermore, many existing methods necessitate complex multi-step sequences or expensive transition metals that complicate downstream purification and increase the environmental footprint of the manufacturing process, making them less attractive for large-scale commercial adoption.
The Novel Approach
The methodology disclosed in patent CN114160206B represents a paradigm shift by employing a specially designed chiral copper catalyst that operates under remarkably mild conditions, typically between 0°C and 25°C. This new approach utilizes 3-alkenyl indole compounds and isatin-like derivatives as readily available starting materials, streamlining the synthetic route significantly. The reaction proceeds with high efficiency, delivering product yields exceeding 81% and purity levels greater than 99.9%, which drastically reduces the burden on purification protocols. By enabling a direct asymmetric olefination, this technology eliminates the need for pre-functionalized reagents or protecting group manipulations that are common in older strategies. The versatility of this system is evident in its ability to accommodate a wide range of substituents on both the indole and isatin cores, providing a flexible platform for generating diverse libraries of chiral intermediates for drug discovery.
Mechanistic Insights into Chiral Copper-Catalyzed Asymmetric Olefination
The exceptional performance of this synthesis method is rooted in the unique architecture of the chiral copper-based catalyst, specifically compounds of Formula (C1) and/or (C2). These catalysts are formed in situ or pre-prepared by coordinating a divalent copper salt with a chiral Schiff base ligand derived from L-phenylalanine and salicylaldehyde derivatives. The ligand design is critical; the bulky aryl groups (Ar1 and Ar2) attached to the quaternary carbon center of the ligand create a highly rigid and sterically demanding chiral pocket around the copper center. This steric environment effectively shields one face of the coordinated substrate, forcing the nucleophilic attack of the 3-alkenyl indole to occur from a specific trajectory. The result is a highly organized transition state that minimizes energy barriers for the desired enantiomer while kinetically suppressing the formation of the opposite enantiomer and unwanted diastereomers.
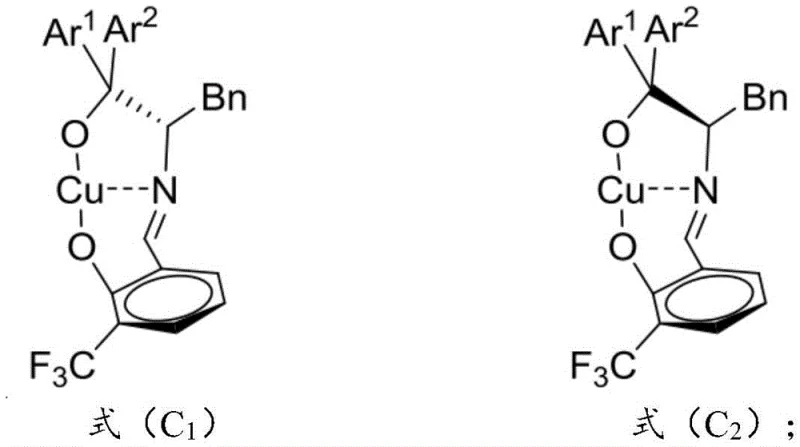
Furthermore, the electronic properties of the ligand play a pivotal role in modulating the Lewis acidity of the copper center, enhancing its ability to activate the carbonyl or imine group of the isatin-like electrophile. The nitrogen-containing organic base present in the catalyst system assists in deprotonating the nucleophile or stabilizing the intermediate species, ensuring a smooth catalytic cycle. This synergistic interaction between the metal center, the chiral ligand, and the base allows the reaction to proceed with enantioselectivity reaching above 91% ee and diastereoselectivity with a dr value greater than 20:1. Such precise control over the stereochemical outcome is crucial for pharmaceutical applications where the biological activity is often dependent on a single enantiomer. The robustness of this mechanistic framework ensures that even with variations in substrate electronics, the catalyst maintains its high fidelity, making it a reliable tool for complex molecule synthesis.
How to Synthesize Optically Active 3-Alkenyl-3-Substituted Oxindoles Efficiently
The practical implementation of this technology involves a straightforward protocol that is amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the chiral ligand, followed by the generation of the active copper catalyst complex, and finally the coupling reaction. The simplicity of the procedure, combined with the use of common solvents like ethanol or dichloromethane, lowers the barrier for adoption by process chemistry teams. The reaction conditions are mild enough to be safe for scale-up while being rigorous enough to ensure consistent quality. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical steps for maximizing yield and purity.
- Prepare the chiral ligand by reacting L-phenylalanine derivatives with salicylaldehyde derivatives to form Schiff base ligands (L1/L2).
- Generate the active chiral copper catalyst complex by mixing the ligand with a divalent copper salt and an organic base in a suitable solvent.
- React 3-alkenyl indoles with isatin-like compounds in the presence of the catalyst at 0-25°C to obtain the optically active product with high ee and dr.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers substantial strategic advantages for procurement and supply chain management within the pharmaceutical sector. The ability to produce high-purity intermediates directly from the reaction mixture significantly simplifies the downstream processing requirements. Traditional methods often require multiple recrystallizations or chromatographic separations to remove closely related impurities and stereoisomers, which are time-consuming and generate significant solvent waste. By achieving purity levels exceeding 99.9% and high stereoselectivity in a single step, this process inherently reduces the consumption of auxiliary materials and energy, leading to a leaner and more cost-effective manufacturing workflow. This efficiency translates directly into improved margin potential and a more competitive pricing structure for the final active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts like cobalt or palladium in favor of a more abundant copper-based system, combined with the high atom economy of the direct olefination reaction, drives down raw material costs. Additionally, the high yield and selectivity minimize the loss of valuable starting materials, ensuring that a greater proportion of input mass is converted into saleable product. The reduction in purification steps further lowers operational expenditures related to solvent recovery and waste disposal, creating a comprehensive cost optimization profile for the entire production lifecycle without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as L-phenylalanine derivatives and substituted isatins, mitigates the risk of supply disruptions often associated with exotic or custom-synthesized reagents. The mild reaction temperatures (0-25°C) reduce the dependency on specialized heating or cooling infrastructure, allowing for production in a wider range of facilities. This flexibility enhances the resilience of the supply chain, ensuring consistent delivery schedules and the ability to respond rapidly to fluctuations in market demand for critical indole-based intermediates used in global drug development pipelines.
- Scalability and Environmental Compliance: The patent data confirms that the stereoselectivity is maintained even when scaling the reaction to the gram level, indicating a strong potential for successful translation to kilogram and ton scales. The use of greener solvents like ethanol for catalyst preparation and the avoidance of hazardous high-temperature conditions align with modern environmental, health, and safety (EHS) regulations. This compliance reduces the regulatory burden and facilitates smoother approval processes for manufacturing sites, supporting sustainable growth and long-term viability in an increasingly regulated global chemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral copper catalysis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers evaluating this synthetic route for their specific projects. Understanding these details is essential for assessing the feasibility of integrating this method into existing production workflows.
Q: What level of stereoselectivity can be achieved with this copper catalyst?
A: The catalyst system described in patent CN114160206B achieves exceptional stereoselectivity, with enantiomeric excess (ee) values exceeding 91% and diastereomeric ratios (dr) greater than 20:1, ensuring the production of single-chirality compounds essential for pharmaceutical applications.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the stereoselectivity of the product is maintained even when the reaction is scaled up to the gram level. The mild reaction conditions (0-25°C) and use of common solvents further support its feasibility for commercial scale-up.
Q: How does this method compare to conventional Lewis acid catalysts?
A: Compared to conventional Lewis acids, this chiral copper-based catalyst offers significant improvements in both selectivity and yield. It operates under milder conditions and provides product purity exceeding 99.9%, reducing the need for extensive downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Indole Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the pharmaceutical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably translated into industrial reality. We are committed to delivering products with stringent purity specifications and utilize our rigorous QC labs to verify every batch, guaranteeing that the high enantioselectivity and diastereoselectivity promised by this copper-catalyzed method are consistently achieved in our commercial outputs.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and optimize your supply chain for high-purity optically active indole compounds.
