Advanced Chiral Copper Catalysis for Scalable Production of Optically Active Indole Intermediates
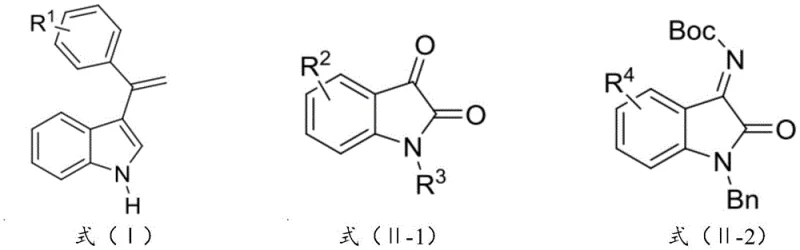
The pharmaceutical industry continuously demands more efficient routes to access complex chiral scaffolds, particularly those found in bioactive natural products and modern therapeutics. Patent CN114160206B introduces a groundbreaking advancement in this domain by disclosing a novel chiral copper-based catalyst system designed for the asymmetric synthesis of optically active indole compounds. This technology specifically targets the construction of 3-alkenyl-3-substituted oxindole motifs, which are prevalent in numerous biologically active molecules such as spirotryprostatin B alkaloids and various receptor antagonists. The core innovation lies in the catalyst's ability to facilitate a direct asymmetric olefination reaction between 3-alkenyl indole compounds and isatin-like substrates with unprecedented levels of stereocontrol. By leveraging this proprietary catalytic system, manufacturers can achieve enantioselectivity exceeding 91% ee and diastereoselectivity greater than 20:1 dr, while maintaining product yields above 81% and purity levels surpassing 99.9%. This represents a significant leap forward compared to prior art methods, offering a reliable pathway for producing high-value pharmaceutical intermediates with reduced impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric alkenylation of isatin and its analogues has presented substantial challenges for synthetic chemists due to the inherent low reactivity of bare alkenyl groups at the reactive sites. Previous methodologies, such as those reported by Zhou's group using cinchona alkaloids or Zhao's group utilizing chiral CoI2-bisphosphine complexes, often required harsh conditions or specific substituents to enhance reactivity, which limited their substrate scope and practical utility. Traditional Lewis acid catalysts frequently struggled to induce sufficient enantioselectivity and diastereoselectivity simultaneously, often resulting in complex mixtures of stereoisomers that necessitated costly and time-consuming purification steps. Furthermore, many existing protocols failed to maintain their stereochemical integrity upon scale-up, creating a bottleneck for the commercial manufacturing of these critical drug precursors. The inability to efficiently construct optically active allyl alcohols and allylamines at the C3 position of oxindoles without compromising yield or purity has remained a persistent pain point in the development of new indole-based therapeutics.
The Novel Approach
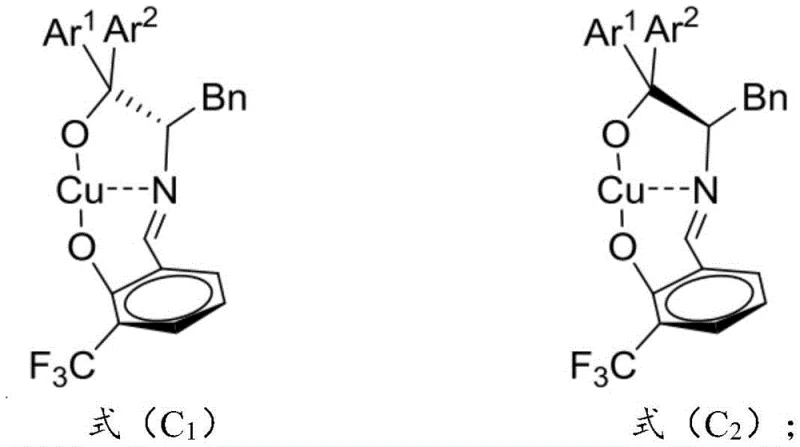
The technology disclosed in CN114160206B overcomes these historical barriers through the deployment of a specially designed chiral copper catalyst, defined by formula (C1) and/or (C2). This new approach enables the direct asymmetric alkenylation of 3-alkenyl indoles to isatin-like compounds under mild conditions, typically between 0°C and 25°C, eliminating the need for extreme temperatures or pressures. The catalyst system utilizes a unique ligand architecture derived from amino acids, which creates a highly defined chiral environment around the copper center, effectively guiding the stereochemical outcome of the reaction. This method not only delivers superior selectivity metrics but also simplifies the operational workflow by allowing for the in situ formation of the catalyst complex, thereby reducing the number of unit operations required. By addressing the reactivity issues of unsubstituted alkenyl groups, this invention opens up a broader chemical space for the synthesis of diverse 3-alkenyl-3-substituted oxindole derivatives, providing a robust solution for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Chiral Copper-Catalyzed Asymmetric Alkenylation
The efficacy of this synthesis relies heavily on the precise molecular architecture of the chiral copper catalyst, which acts as the central engine for stereochemical induction. The catalyst is formed through the coordination of a divalent copper salt, such as copper bromide or copper trifluoromethanesulfonate, with a chiral Schiff base ligand (L1 or L2) in the presence of a nitrogen-containing organic base. The ligand itself is synthesized from L-phenylalanine derivatives and salicylaldehyde derivatives, creating a rigid backbone that locks the copper ion into a specific geometric configuration. This configuration is critical for differentiating the enantiotopic faces of the incoming electrophile and nucleophile during the bond-forming event. The interaction between the copper center and the ligand's donor atoms (oxygen and nitrogen) stabilizes the transition state, lowering the activation energy for the desired pathway while sterically hindering the formation of unwanted stereoisomers.
Furthermore, the mechanism ensures exceptional impurity control by minimizing side reactions that typically plague transition metal catalysis, such as polymerization of the alkenyl species or non-selective background reactions. The high diastereoselectivity (dr > 20:1) indicates that the catalyst effectively controls both the newly formed stereocenters simultaneously, preventing the generation of diastereomeric impurities that are notoriously difficult to separate. The use of mild reaction conditions (10-20°C) further contributes to product stability, preventing thermal degradation or racemization that could occur at higher temperatures. This level of mechanistic precision translates directly into a cleaner crude reaction profile, which significantly reduces the burden on downstream purification processes like column chromatography or recrystallization, ultimately enhancing the overall process efficiency and sustainability.
How to Synthesize Optically Active 3-Alkenyl-3-Substituted Oxindoles Efficiently
The synthesis protocol outlined in the patent offers a straightforward and reproducible method for accessing these valuable chiral building blocks, suitable for both laboratory research and pilot-scale production. The process begins with the preparation of the chiral ligand, followed by the in situ generation of the active copper catalyst, and concludes with the coupling of the indole and isatin substrates. This modular approach allows for flexibility in substrate selection, accommodating various substituents on both the indole and isatin rings to generate a diverse library of compounds. The detailed standardized synthesis steps below provide a clear roadmap for implementing this technology, ensuring consistent results and high-quality output for your specific application needs.
- Prepare the chiral ligand (L1 or L2) by reacting L-phenylalanine derivatives with salicylaldehyde derivatives under mild conditions.
- Form the active chiral copper catalyst complex in situ by mixing a divalent copper salt, the chiral ligand, and a nitrogen-containing organic base in a solvent like ethanol.
- Conduct the asymmetric direct alkenylation reaction by adding 3-alkenyl indole and isatin-like compounds to the catalyst system at 0-25°C to achieve high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this catalytic technology offers transformative benefits that extend beyond mere chemical yield. The ability to produce optically active indole compounds with purity exceeding 99.9% drastically simplifies the quality control landscape, reducing the risk of batch rejection due to impurity specifications. High stereochemical purity is paramount in pharmaceutical manufacturing, as even trace amounts of the wrong enantiomer can have significant regulatory and safety implications. By securing a supply of intermediates produced via this high-selectivity route, procurement teams can mitigate the risks associated with complex purification workflows and ensure a more stable supply of critical raw materials for API synthesis. Additionally, the robustness of the catalyst system supports consistent batch-to-batch reproducibility, which is essential for maintaining long-term supply continuity.
- Cost Reduction in Manufacturing: The implementation of this catalyst eliminates the need for expensive and toxic heavy metal scavengers often required with traditional transition metal catalysts, leading to substantial cost savings in waste treatment and material consumption. The high yield (>81%) and selectivity mean that less starting material is wasted on byproducts, optimizing the atom economy of the process. Furthermore, the simplified workup procedure, which avoids complex separation techniques for diastereomers, reduces labor hours and solvent usage, contributing to a leaner and more cost-effective manufacturing operation overall.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including substituted indoles and isatins, are commercially available and structurally diverse, reducing dependency on single-source custom synthons. The mild reaction conditions (0-25°C) allow the process to be run in standard glass-lined or stainless steel reactors without requiring specialized cryogenic or high-pressure equipment, making it easier for contract manufacturing organizations (CMOs) to adopt. This accessibility ensures that production schedules are less likely to be disrupted by equipment limitations or raw material shortages, fostering a more resilient supply chain.
- Scalability and Environmental Compliance: The patent explicitly notes that stereoselectivity is maintained when scaling the reaction to gram levels, indicating a smooth path towards kilogram and ton-scale commercial production. The use of common solvents like ethanol and ethyl acetate aligns with green chemistry principles, facilitating easier regulatory approval and waste disposal compared to processes relying on chlorinated or hazardous solvents. This environmental compatibility not only reduces the ecological footprint but also minimizes compliance costs related to hazardous waste management, making the process sustainable for long-term industrial application.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic system, derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production pipelines. The answers reflect the specific performance metrics and operational parameters validated in the patent examples, providing a factual basis for decision-making.
Q: What are the selectivity metrics for this new catalytic method?
A: The method achieves exceptional stereocontrol with enantioselectivity exceeding 91% ee and diastereoselectivity greater than 20:1 dr, ensuring high optical purity for drug candidates.
Q: Can this synthesis process be scaled for industrial production?
A: Yes, the patent demonstrates that stereoselectivity is maintained even when scaling the reaction to gram-level quantities, indicating robust potential for commercial manufacturing.
Q: How does this catalyst compare to traditional Lewis acids?
A: Unlike conventional Lewis acids which often suffer from poor reactivity or selectivity with bare alkenyl groups, this chiral copper catalyst provides significantly improved yields (>81%) and purity (>99.9%).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Indole Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is fully equipped to handle the nuances of chiral copper catalysis, ensuring that the stringent purity specifications and rigorous QC labs required for pharmaceutical intermediates are met with precision. We understand that the transition from bench-scale discovery to industrial manufacturing requires not just chemical expertise but also a deep commitment to quality assurance and process safety, which are the cornerstones of our CDMO services.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project requirements. Whether you need specific COA data for the 3-alkenyl-3-substituted oxindole derivatives or comprehensive route feasibility assessments for your API pipeline, our experts are ready to provide the support you need. Partner with us to leverage this cutting-edge catalytic technology and secure a competitive advantage in the global market for high-purity pharmaceutical intermediates.
