Advanced Imatinib Manufacturing: A Scalable Ester Aminolysis Strategy for Global Supply Chains
Introduction to Next-Generation Imatinib Synthesis
The pharmaceutical landscape for kinase inhibitors continues to evolve, driven by the urgent need for safer, more efficient manufacturing processes for critical oncology treatments. Patent CN101735196A introduces a transformative methodology for the synthesis of Imatinib, a pivotal tyrosine kinase inhibitor used globally in the treatment of chronic myeloid leukemia. This innovation addresses longstanding inefficiencies in prior art by replacing hazardous reagents and complex multi-step sequences with a direct, high-yielding ester aminolysis strategy. By leveraging the nucleophilic properties of a pyrimidine-substituted diamine against a functionalized benzoate ester, this process achieves exceptional conversion rates under remarkably mild conditions. The significance of this technical breakthrough extends beyond the laboratory, offering a robust pathway for industrial scale-up that prioritizes operator safety and environmental compliance without compromising on the stringent purity standards required for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
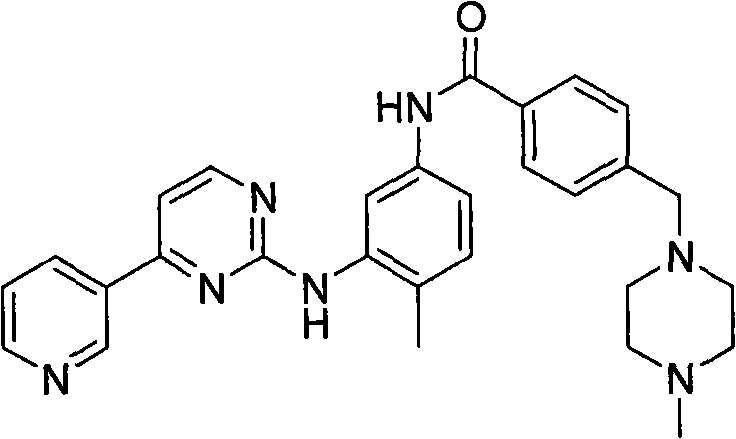
The Limitations of Conventional Methods
Historically, the industrial production of Imatinib has been plagued by significant chemical and operational hurdles that impact both cost and safety profiles. Traditional synthetic routes, such as those disclosed in WO 2004/108699, rely heavily on the use of cyanamide to construct the guanidine moiety, a reagent known for its high volatility and tendency to produce inconsistent yields due to evaporation losses. Furthermore, alternative pathways utilizing trimethylaluminum for aminolysis, as seen in CN1630648A, introduce severe safety risks due to the pyrophoric nature of the reagent, requiring specialized handling infrastructure and inert atmosphere protocols that drive up capital expenditure. Perhaps most critically, these legacy methods often suffer from poor selectivity, generating isomeric impurities at levels as high as 10%, which necessitates complex and yield-eroding purification steps to meet regulatory specifications for human consumption.
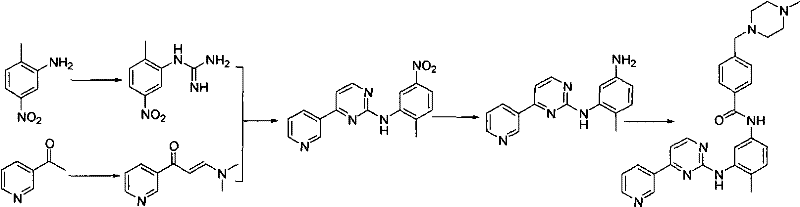
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology outlined in CN101735196A streamlines the final coupling step through a direct aminolysis reaction that is both chemically elegant and operationally simple. By reacting 4-methyl-N-3-(4-pyridin-3-yl-pyrimidine-2-yl)-1,3-phenylenediamine directly with a 4-(4-methylpiperazine-1-methyl)-benzoate ester in the presence of a mild alkali base, the process bypasses the need for dangerous acid chlorides or volatile cyanamide derivatives entirely. This approach not only eliminates the formation of hazardous byproducts but also ensures that the reaction proceeds to completion with high atom economy, as the only byproduct is a benign alcohol. The versatility of this method is further demonstrated by its compatibility with a wide range of solvents and bases, allowing manufacturers to optimize the process for specific facility constraints while maintaining consistently high yields above 90%.
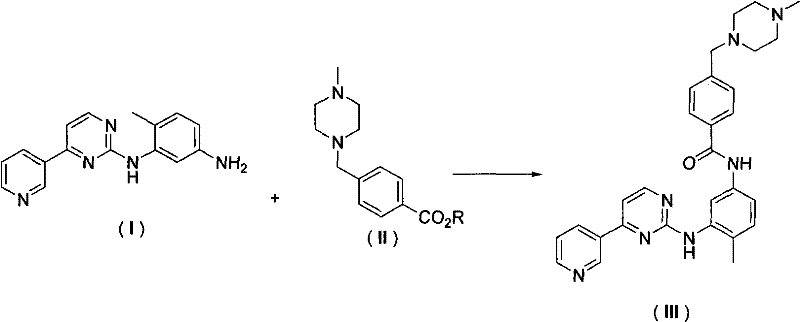
Mechanistic Insights into Base-Catalyzed Ester Aminolysis
The core chemical transformation driving this synthesis is a nucleophilic acyl substitution, where the primary amine of the diamine intermediate acts as a potent nucleophile attacking the carbonyl carbon of the benzoate ester. The presence of an alkali base, such as sodium methylate or potassium tert-butoxide, plays a dual role in this mechanism: it serves to deprotonate the amine slightly, thereby increasing its nucleophilicity, and it helps to stabilize the tetrahedral intermediate formed during the attack. This catalytic environment lowers the activation energy required for the bond formation, allowing the reaction to proceed efficiently at temperatures ranging from 25°C to 50°C, which is significantly milder than the harsh conditions often required for amide bond formation using acid chlorides. The electronic properties of the pyrimidine ring adjacent to the amine do not hinder this reactivity; rather, the specific substitution pattern ensures that the amine remains sufficiently reactive to displace the alkoxide leaving group of the ester effectively.
From an impurity control perspective, this mechanism offers distinct advantages over transition metal-catalyzed alternatives. By avoiding the use of palladium catalysts and halogenated precursors, the process inherently prevents the formation of metal-associated impurities and halogenated side products that are notoriously difficult to remove to trace levels. The chemoselectivity of the aminolysis is high, as the reaction conditions are tuned to favor the attack of the specific aniline nitrogen over other potential nucleophilic sites within the complex molecular architecture. This precision minimizes the generation of regioisomers and over-acylation byproducts, resulting in a crude reaction mixture that is significantly cleaner and requires less intensive chromatographic purification, thereby preserving overall process yield and reducing solvent consumption.
How to Synthesize Imatinib Efficiently
The practical execution of this synthesis involves dissolving the key diamine intermediate and the functionalized benzoate ester in a suitable aprotic solvent such as tetrahydrofuran or toluene, followed by the addition of a stoichiometric amount of base. The reaction mixture is then heated gently to facilitate the aminolysis, monitored until the starting materials are fully consumed, typically indicated by HPLC analysis. Following the reaction, the workup procedure is straightforward, involving the removal of the solvent and washing of the resulting solid to remove the alcohol byproduct and residual salts, yielding the final API intermediate in high purity. For detailed standardized operating procedures and specific parameter optimization, please refer to the technical guide below.
- Dissolve 4-methyl-N-3-(4-pyridin-3-yl-pyrimidine-2-yl)-1,3-phenylenediamine and the corresponding benzoate ester in an aprotic organic solvent such as THF or Toluene.
- Add a suitable alkali base, such as sodium methylate or potassium tert-butoxide, to the reaction mixture to initiate nucleophilic attack.
- Heat the mixture to a mild temperature range of 25-50°C until reaction completion, followed by concentration and purification to isolate high-purity Imatinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ester aminolysis technology represents a strategic opportunity to de-risk the supply of critical oncology intermediates while simultaneously driving down manufacturing costs. The elimination of pyrophoric reagents like trimethylaluminum removes the need for specialized storage and handling equipment, significantly lowering the barrier to entry for contract manufacturing organizations and reducing the overhead costs associated with safety compliance. Furthermore, the reliance on commercially available esters and diamines ensures a stable supply of raw materials, mitigating the risk of bottlenecks that often occur with proprietary or highly regulated reagents. This stability allows for more accurate forecasting and inventory management, essential for maintaining continuous production schedules in a high-demand therapeutic area.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the reaction workflow and the reduction in waste disposal costs. By replacing expensive and hazardous reagents with common alkali bases and esters, the direct material costs are substantially lowered, while the high yield minimizes the loss of valuable intermediates. Additionally, the benign nature of the alcohol byproduct simplifies waste treatment protocols, avoiding the high fees associated with disposing of heavy metal catalysts or halogenated waste streams, leading to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates directly into improved supply chain resilience, as the mild reaction conditions are less prone to failure due to minor fluctuations in temperature or mixing efficiency. The use of standard organic solvents and bases means that sourcing is not dependent on single-source suppliers of exotic chemicals, providing procurement teams with greater flexibility and negotiating power. This redundancy in the supply base ensures that production can be maintained even during periods of global raw material scarcity, securing the continuity of supply for downstream drug formulation.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is facilitated by the absence of exothermic hazards and the use of conventional reactor materials, allowing for rapid capacity expansion without major infrastructure upgrades. The environmentally friendly profile of the reaction, characterized by low toxicity and minimal hazardous waste generation, aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against regulatory changes. This sustainability advantage not only reduces compliance risks but also enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this novel Imatinib synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms traditional techniques in terms of safety, purity, and efficiency. Understanding these distinctions is crucial for technical teams evaluating process transfers or seeking to optimize their current manufacturing workflows for better economic and environmental outcomes.
Q: How does this new method improve upon traditional cyanamide-based routes?
A: Traditional routes often utilize cyanamide for guanidine formation, which is highly volatile and leads to unstable yields. This novel ester aminolysis method eliminates the need for cyanamide, resulting in a more stable reaction profile and significantly higher overall yields without the safety hazards associated with volatile reagents.
Q: Does this process avoid the formation of difficult-to-remove isomers?
A: Yes. Unlike methods employing trimethylaluminum which can generate up to 10% isomeric impurities that are challenging to purify, this direct aminolysis approach offers superior chemoselectivity. The mild basic conditions ensure clean conversion to the target amide, simplifying downstream purification and enhancing final product purity.
Q: What are the environmental benefits of this synthesis route?
A: The primary byproduct of this aminolysis reaction is the corresponding alcohol derived from the ester, which is generally less toxic and easier to handle than the halogenated waste or aluminum residues produced in alternative methods. This aligns with green chemistry principles by reducing hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imatinib Supplier
As the demand for high-quality kinase inhibitors continues to grow, partnering with a technically proficient manufacturer is essential for ensuring product integrity and market availability. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver Imatinib intermediates that meet the most rigorous industry standards. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch conforms to the precise chemical profile required for final API synthesis, minimizing the risk of downstream processing failures. We are committed to supporting our partners with a supply chain that is both resilient and responsive to the dynamic needs of the global pharmaceutical market.
We invite you to engage with our technical team to explore how this advanced synthesis route can be integrated into your supply chain to drive value and efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific excellence and commercial reliability.
