Scalable One-Pot Synthesis of Imatinib Intermediate for Commercial API Production
Introduction to Advanced Imatinib Intermediate Manufacturing
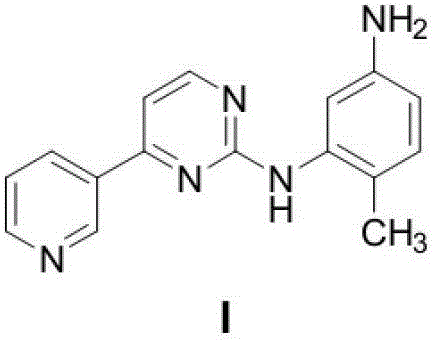
The pharmaceutical landscape for tyrosine kinase inhibitors continues to evolve, driven by the relentless demand for cost-effective and scalable manufacturing processes for critical oncology therapeutics. A pivotal advancement in this domain is detailed in patent CN102796074A, which discloses a highly efficient methodology for preparing N-(2-amino-4-methylphenyl)-4-(3-pyridyl)-2-pyrimidinamine, a key intermediate in the synthesis of Imatinib Mesylate. This innovative approach fundamentally restructures the synthetic pathway by employing a one-pot condensation strategy that merges the coupling and deprotection phases, thereby drastically reducing the operational complexity typically associated with bulk drug production. By leveraging a robust copper-catalyzed system, this technology addresses long-standing bottlenecks regarding step count and atom economy, offering a compelling value proposition for global supply chains seeking reliable pharmaceutical intermediates supplier partnerships. The strategic implementation of this route not only streamlines the workflow but also aligns with modern green chemistry principles by minimizing waste generation and avoiding toxic reagents often found in legacy protocols.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the cumbersome multi-step sequences that have historically plagued Imatinib synthesis. Traditional routes often necessitate the isolation of unstable nitro-intermediates or the use of expensive palladium catalysts, which introduce significant cost volatility and supply chain fragility. In contrast, the disclosed method utilizes readily available starting materials and a cost-effective copper catalytic system to achieve high conversion rates. This shift represents a paradigm change in how we approach the commercial scale-up of complex API intermediates, transforming a potentially hazardous and low-yielding process into a streamlined, industrially viable operation. As we delve deeper into the technical specifics, it becomes evident that this methodology offers a sustainable pathway for meeting the growing global demand for this life-saving medication while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing the Imatinib backbone have been fraught with inefficiencies that hinder large-scale commercial viability. One prevalent conventional method involves the initial linkage of a piperazine fragment with a nitro-segment, followed by a separate nitro-reduction step and subsequent guanidine glycosylation. This linear progression is inherently flawed due to the accumulation of impurities at each stage and the notoriously low total recovery rates associated with multi-step syntheses. Furthermore, alternative routes relying on the chlorination of pyrimidine precursors using phosphorus oxychloride (POCl3) introduce severe environmental and safety liabilities, generating highly toxic byproducts that require specialized and costly waste treatment infrastructure. These legacy methods also frequently employ functional group protection strategies that add unnecessary synthetic steps, inflating both the time-to-market and the overall cost of goods sold (COGS). The reliance on precious metal catalysts in some variations further exacerbates cost concerns, making these processes economically unsustainable for generic manufacturers aiming for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
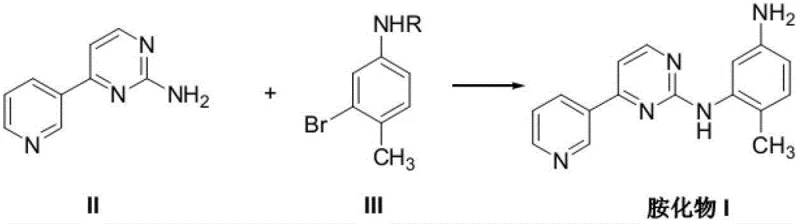
The methodology presented in patent CN102796074A offers a transformative solution by consolidating the synthesis into a concise, high-efficiency sequence. The core innovation involves the direct condensation of 4-(3-pyridyl)pyrimidine-2-amine with a protected 2-bromo-4-aminotoluene derivative in a single reaction vessel. This "one-pot" philosophy eliminates the need for isolating intermediate species, thereby reducing solvent consumption, labor hours, and equipment occupancy time. Crucially, the process employs a versatile protecting group strategy where the amino group on the toluene ring can be masked with various moieties such as acetyl, benzyl, or Boc groups, all of which are compatible with the subsequent coupling conditions. Following the coupling event, the protecting group is removed in situ or in a subsequent simple workup using either strong acids or bases, directly yielding the target amine intermediate. This telescoped approach not only enhances the overall yield but also significantly simplifies the purification profile, resulting in a higher purity product that is essential for downstream pharmaceutical applications.
Mechanistic Insights into Copper-Catalyzed C-N Coupling
The heart of this synthetic breakthrough is the copper-catalyzed C-N bond formation, a variation of the Ullmann-type coupling that has been optimized for industrial robustness. The reaction mechanism relies on the synergistic interaction between a copper source, specifically Cuprous Iodide (CuI), and a bidentate nitrogen ligand, N,N-dimethylethylenediamine (DMEDA). This catalytic system facilitates the oxidative addition of the aryl bromide to the copper center, followed by coordination of the pyrimidine amine and subsequent reductive elimination to forge the C-N bond. The choice of DMEDA is critical, as it stabilizes the copper species in the active oxidation state and accelerates the turnover frequency, allowing the reaction to proceed efficiently at moderate temperatures ranging from 60°C to 140°C, with 100°C being the optimal setpoint in dioxane solvent. The presence of an inorganic base, such as anhydrous potassium carbonate, serves to neutralize the hydrogen bromide byproduct, driving the equilibrium towards product formation and preventing catalyst deactivation. This mechanistic elegance ensures that the reaction tolerates a wide array of functional groups, making it highly adaptable for process optimization.
From an impurity control perspective, this mechanism offers distinct advantages over palladium-catalyzed alternatives. Copper residues are generally easier to remove to trace levels compared to palladium, which is subject to extremely stringent regulatory limits in final drug substances. Furthermore, the use of protected aniline derivatives prevents potential side reactions such as polymerization or over-alkylation that could occur with free amines under harsh coupling conditions. The patent explicitly details that protecting groups like the acetyl group (where R1 is -CH3) are particularly effective, balancing stability during the coupling phase with ease of removal during the deprotection phase. Whether using trifluoroacetic acid for acidic deprotection or sodium hydroxide for basic hydrolysis, the cleavage of the protecting group proceeds cleanly without compromising the integrity of the sensitive pyrimidine-pyridine scaffold. This level of control over the reaction trajectory is paramount for ensuring a consistent impurity profile, a key metric for any reliable pharmaceutical intermediates supplier aiming to support regulatory filings.
How to Synthesize N-(2-amino-4-methylphenyl)-4-(3-pyridyl)-2-pyrimidinamine Efficiently
Implementing this synthesis requires precise adherence to the optimized parameters outlined in the patent to ensure maximum yield and purity. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically nitrogen, to prevent oxidation of the copper catalyst. Key reagents including the pyrimidine amine, the protected bromo-toluene, CuI, potassium carbonate, and the DMEDA ligand are charged into a reactor containing dioxane. The mixture is then heated to reflux, maintaining vigorous stirring to ensure homogeneous mass transfer throughout the 20-hour reaction period. Upon completion, the system is cooled, and the deprotection agent is introduced directly, streamlining the workflow. For a comprehensive, step-by-step technical guide including exact molar ratios and workup procedures, please refer to the standardized protocol below.
- Combine 4-(3-pyridyl)pyrimidine-2-amine, protected 2-bromo-4-aminotoluene, CuI catalyst, K2CO3 base, and DMEDA ligand in dioxane solvent.
- Heat the reaction mixture to reflux (approximately 100°C) under nitrogen protection and maintain stirring for 20 hours to complete the coupling.
- Perform deprotection using strong acid (e.g., TFA or HCl) or strong base (e.g., NaOH), followed by pH adjustment and extraction to isolate the final amine intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this synthetic route translates into tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the manufacturing process, which inherently reduces the risk of batch failures and production delays. By collapsing multiple synthetic steps into a streamlined sequence, manufacturers can significantly lower their operational expenditures related to labor, energy, and solvent usage. This efficiency gain is critical in a competitive market where cost reduction in pharmaceutical intermediates manufacturing is a primary driver of profitability. Moreover, the avoidance of hazardous reagents like POCl3 and the elimination of high-pressure hydrogenation steps mitigate safety risks, leading to lower insurance premiums and reduced regulatory scrutiny. The robustness of the copper-catalyzed system also implies a more stable supply of the intermediate, as the raw materials are commodity chemicals with established global supply chains, reducing the likelihood of raw material shortages disrupting production schedules.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts in favor of abundant copper salts results in substantial raw material cost savings. Additionally, the one-pot nature of the reaction reduces solvent consumption and waste disposal costs, as fewer isolation and purification steps are required. The high atom economy of the coupling reaction ensures that a greater proportion of input materials are converted into valuable product, minimizing waste generation and maximizing resource utilization. These factors collectively contribute to a leaner, more cost-effective production model that enhances the overall margin profile of the final API.
- Enhanced Supply Chain Reliability: The versatility of the protecting group strategy allows manufacturers to source the most economically viable aniline derivative available at any given time, whether it be the acetyl, Boc, or Cbz protected variant. This flexibility acts as a buffer against price volatility in specific raw material markets, ensuring continuous production capability. Furthermore, the mild reaction conditions and standard equipment requirements mean that this process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without the need for specialized high-pressure or corrosion-resistant reactors. This adaptability strengthens the resilience of the supply chain against geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and reagents that are manageable on a multi-ton scale. The absence of toxic heavy metals like palladium simplifies the environmental compliance landscape, reducing the burden of heavy metal testing and remediation in wastewater streams. The use of dioxane, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols. By adhering to greener chemistry principles through improved atom economy and reduced step count, this method supports corporate sustainability goals and facilitates smoother regulatory approvals in environmentally conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within patent CN102796074A, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines or for assessing the quality of intermediates sourced from external suppliers. The answers reflect a balance between theoretical potential and practical execution, highlighting the robustness of the described methodology.
Q: What are the advantages of this one-pot synthesis over traditional nitro-reduction routes?
A: This method eliminates the need for hazardous nitro-reduction steps involving tin/hydrochloric acid or high-pressure hydrogenation, significantly improving safety profiles and reducing heavy metal waste disposal costs.
Q: Which protecting groups are compatible with this copper-catalyzed coupling?
A: The process demonstrates high versatility, successfully utilizing acetyl, benzyl, trityl, tosyl, Fmoc, Boc, and Cbz protecting groups, with acetyl being the preferred cost-effective option.
Q: What yields can be expected from this optimized protocol?
A: Experimental data indicates robust yields ranging from 72.5% to 81.5% depending on the specific deprotection conditions employed, ensuring high material throughput for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imatinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape. Our technical team has extensively analyzed the methodology described in CN102796074A and possesses the expertise to implement this advanced copper-catalyzed coupling strategy at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of Imatinib intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch adheres to the highest international standards. By leveraging our deep understanding of process chemistry and our commitment to operational excellence, we serve as a strategic partner for companies looking to optimize their Imatinib supply chain.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. Whether you are looking to reduce costs, improve supply security, or accelerate your development timeline, we are prepared to offer a Customized Cost-Saving Analysis tailored to your needs. We encourage potential partners to request specific COA data and route feasibility assessments to verify our capabilities firsthand. Let us collaborate to drive efficiency and innovation in the production of this vital oncology therapeutic, ensuring that life-saving medications reach patients faster and more affordably.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
