Scalable Production of High-Purity BINAP via Novel Lithiation Technology
Scalable Production of High-Purity BINAP via Novel Lithiation Technology
The global demand for chiral ligands in asymmetric synthesis has surged, driven by the pharmaceutical industry's relentless pursuit of enantiomerically pure active ingredients. At the forefront of this sector is 2,2'-bis-diphenylphosphino-1,1'-binaphthyl, universally known as BINAP, a critical ligand for Nobel Prize-winning catalytic reactions. A groundbreaking patent, CN108586528B, disclosed in March 2020, introduces a transformative synthetic methodology that addresses long-standing inefficiencies in BINAP manufacturing. This technical insight report analyzes the patent's novel reductive lithiation strategy, which replaces hazardous high-temperature bromination and expensive noble metal catalysis with a robust, cost-effective ether cleavage protocol. For R&D directors and supply chain leaders, this innovation represents a pivotal shift towards greener, more economically viable production of high-value organophosphine compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of BINAP has been plagued by severe operational constraints and prohibitive costs associated with legacy methodologies. The pioneering Noyori/Takasago route, while chemically elegant, necessitates a brutal bromination step at temperatures reaching 320°C, imposing extreme stress on reactor materials and limiting throughput due to safety concerns. Furthermore, the yield of this high-temperature bromination is notoriously low, often hovering around 45%, which creates significant waste disposal challenges and inflates the cost of goods sold. Alternatively, the Merck and Monsanto processes utilize trifluoromethanesulfonic anhydride to activate the binaphthol core, a reagent that is not only exorbitantly priced but also generates corrosive acidic waste streams. These traditional pathways also frequently rely on air-sensitive reagents like diphenylphosphine hydrogen or expensive nickel and palladium catalysts, complicating the supply chain with stringent storage requirements and necessitating complex downstream purification to remove trace heavy metals from the final API intermediates.
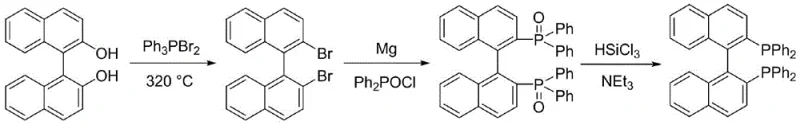
The Novel Approach
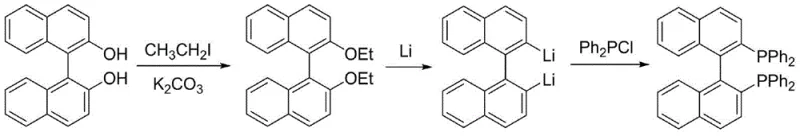
In stark contrast to these cumbersome legacy protocols, the method described in CN108586528B leverages a sophisticated reductive lithiation strategy that fundamentally simplifies the molecular construction of the BINAP skeleton. By converting binaphthol into a 2,2'-bisethoxy-1,1'-binaphthyl intermediate using inexpensive ethyl iodide, the process establishes a stable precursor that can be efficiently activated by metallic lithium. This activation step cleaves the carbon-oxygen bonds to generate a reactive dilithio species in situ, which subsequently undergoes nucleophilic substitution with diphenylphosphine chloride. This approach completely bypasses the need for trifluoromethanesulfonic anhydride and eliminates the reliance on noble metal catalysts, thereby streamlining the reaction workflow. The result is a high-yielding process (up to 97% in optimized examples) that operates under relatively mild thermal conditions, significantly reducing energy consumption and equipment wear while delivering a product of exceptional purity suitable for direct application in sensitive catalytic cycles.
Mechanistic Insights into Reductive Lithiation and Phosphorylation
The core chemical innovation of this patent lies in the efficient generation of the binaphthyl dilithium species through the reduction of the ethoxy-substituted intermediate. Unlike traditional halogen-lithium exchange which requires cryogenic temperatures and pre-halogenated substrates, this method utilizes metallic lithium to directly cleave the C-O bond of the ethyl ether groups. The presence of chelating ligands such as tetramethylethylenediamine (TMEDA) or 12-crown-4 is critical in this mechanism, as they solvate the lithium cations and enhance the reactivity of the metal surface, facilitating electron transfer at moderate temperatures ranging from 60°C to 140°C. This reductive cleavage generates a highly nucleophilic aryl-lithium intermediate at the 2 and 2' positions of the binaphthyl backbone, setting the stage for the subsequent phosphorylation step without the formation of unwanted regioisomers.
From an impurity control perspective, this mechanism offers distinct advantages over transition-metal catalyzed couplings. In nickel or palladium-catalyzed routes, side reactions such as homocoupling of the phosphine reagent or incomplete conversion of the triflate intermediate often lead to difficult-to-remove phosphine oxide impurities or residual metal contaminants. In the lithiation pathway described here, the reaction is driven by stoichiometric equivalents of lithium and diphenylphosphine chloride, ensuring complete consumption of the intermediate. The subsequent quenching with water allows for the easy hydrolysis and removal of excess lithium salts and unreacted phosphine chlorides into the aqueous phase. The final product precipitates or can be crystallized from organic solvents, yielding a solid with purity levels exceeding 99.8% as confirmed by HPLC analysis, effectively mitigating the risk of catalyst poisoning in downstream pharmaceutical applications.
How to Synthesize 2,2'-bis-diphenylphosphino-1,1'-binaphthyl Efficiently
The implementation of this synthetic route involves a straightforward two-stage sequence that begins with the O-alkylation of commercially available binaphthol. The first stage produces the bisethoxy intermediate in high yield using potassium carbonate as a base, a reagent that is both inexpensive and easy to handle on a multi-ton scale. The second stage involves the critical lithiation and phosphorylation steps, where precise control of temperature and reagent addition rates ensures optimal conversion. While the general procedure is robust, adherence to the specific solvent systems and ligand ratios outlined in the patent examples is essential for maximizing yield and minimizing side products. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are provided in the technical guide below.
- Synthesize 2,2'-bisethoxy-1,1'-binaphthyl by reacting binaphthol with ethyl iodide and potassium carbonate in acetonitrile at 80°C.
- Perform reductive lithiation of the bisethoxy intermediate using metallic lithium and a ligand (e.g., TMEDA) in an ether solvent at 60-140°C.
- Quench the dilithio intermediate with diphenylphosphine chloride at 0°C, followed by aqueous workup and purification to obtain BINAP.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this lithiation-based synthesis offers a compelling value proposition centered on raw material stability and process simplification. By shifting away from the volatile and costly trifluoromethanesulfonic anhydride to commodity chemicals like ethyl iodide and metallic lithium, the manufacturing cost structure is drastically improved. Furthermore, the elimination of noble metal catalysts removes the necessity for expensive metal scavenging resins and complex analytical testing for residual heavy metals, which are significant cost drivers in the production of pharmaceutical intermediates. This route also enhances supply chain resilience by relying on widely available bulk chemicals rather than specialized, single-source reagents that are prone to market fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the substitution of high-cost activators and catalysts with low-cost alternatives. The replacement of trifluoromethanesulfonic anhydride with ethyl iodide represents a massive reduction in raw material expenditure, as the latter is a mass-produced alkylating agent with a stable global supply. Additionally, the absence of nickel or palladium catalysts eliminates the capital expense associated with catalyst recovery systems and the operational cost of rigorous metal removal protocols. The high reaction yield reported in the patent further contributes to cost efficiency by maximizing the output per batch, reducing the overall solvent and energy usage per kilogram of finished BINAP.
- Enhanced Supply Chain Reliability: Operational simplicity translates directly into supply chain reliability, as this method avoids the use of air-sensitive diphenylphosphine hydrogen, which requires specialized handling and storage infrastructure. The use of diphenylphosphine chloride, a stable liquid, simplifies logistics and reduces the risk of shipment delays due to hazardous material restrictions. Moreover, the reaction conditions do not demand the extreme temperatures of the Noyori route or the strictly anhydrous, inert atmospheres required for sensitive transition metal catalysis, allowing for production in standard glass-lined or stainless steel reactors without specialized modifications. This flexibility ensures consistent production schedules and minimizes the risk of batch failures due to environmental excursions.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns with modern green chemistry principles by reducing the generation of hazardous waste. The avoidance of heavy metal catalysts significantly lowers the toxicity profile of the waste stream, simplifying wastewater treatment and disposal compliance. The solvent systems employed, such as tetrahydrofuran and dibutyl ether, are well-understood in industrial settings and can be efficiently recovered and recycled via distillation, as demonstrated in the patent examples. The ability to scale this reaction from laboratory glassware to 50L reactors with maintained yields indicates a smooth path to commercial scale-up, enabling manufacturers to rapidly increase capacity to meet surging market demand for chiral ligands without extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and comparative analysis provided in CN108586528B, offering clarity on the practical benefits of switching to this lithiation-based protocol. Understanding these nuances is crucial for technical teams evaluating the feasibility of technology transfer and for procurement specialists negotiating supply agreements based on process efficiency.
Q: How does the new lithiation method reduce production costs compared to the Merck process?
A: The new method eliminates the need for expensive trifluoromethanesulfonic anhydride and noble metal catalysts (Nickel/Palladium), replacing them with cost-effective ethyl iodide and metallic lithium.
Q: What are the purity levels achievable with this synthetic route?
A: According to patent data, the process yields product with purity up to 99.8% as analyzed by liquid chromatography, meeting stringent requirements for asymmetric catalysis.
Q: Does this process require inert atmosphere protection?
A: Unlike previous catalytic coupling methods requiring strict inert gas protection for sensitive catalysts, this lithiation route simplifies operational requirements, enhancing industrial feasibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BINAP Supplier
The technological advancements detailed in patent CN108586528B underscore the immense potential for optimizing the production of critical chiral ligands like BINAP. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this lithiation method are fully realized in large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chromatography and spectroscopy tools to guarantee that every batch of BINAP meets the exacting standards required for asymmetric catalysis in drug discovery and development.
We invite pharmaceutical and agrochemical companies to collaborate with us to leverage this cost-effective synthesis route for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic advantages of adopting this method for your projects. We encourage you to contact us today to discuss your specific requirements,索取 specific COA data for our current inventory, and review our comprehensive route feasibility assessments to ensure a seamless integration of high-quality BINAP into your manufacturing pipeline.
