Advanced Binaphthol Aluminum Catalysis for Commercial Scale Isopulegol Manufacturing
The global demand for high-purity menthol and its precursors continues to drive innovation in fine chemical synthesis, particularly for intermediates like isopulegol. Patent CN108329194B introduces a transformative methodology for the preparation of isopulegol from citronellal, utilizing a specialized binaphthol-based aluminum compound as the primary catalyst. This technology represents a significant leap forward from traditional Lewis acid catalysis, addressing long-standing industry pain points regarding enantioselectivity, catalyst recovery, and process safety. By leveraging the unique steric and electronic properties of binaphthol skeleton ligands, this invention achieves exceptional conversion rates ranging from 95% to 99.9% while maintaining enantiomeric excess levels that meet the rigorous standards of the flavor and pharmaceutical sectors. The process operates under remarkably mild conditions, often near room temperature, which drastically reduces energy consumption compared to cryogenic alternatives. Furthermore, the integration of a novel high-boiling point solvent system facilitates a crystallization-based catalyst recovery method that preserves the structural integrity of the expensive catalytic species, enabling direct reuse without complex regeneration steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial cyclization of citronellal to isopulegol has relied heavily on heterogeneous catalysts like silica-alumina mixtures or homogeneous Lewis acids such as zinc bromide and tin tetrachloride. While zinc bromide has seen commercial application, it presents severe drawbacks, including significant corrosion risks to reactor equipment due to bromide ion generation and relatively modest enantioselectivity capped around 91%. Other organic aluminum-based methods described in prior art, such as those utilizing diaryl phenoxy aluminum compounds, often necessitate the addition of ester inhibitors to control selectivity, which complicates the downstream purification process. Moreover, the recovery of these traditional catalysts frequently involves quenching active species followed by aqueous alkaline washing to separate ligands, a procedure that often leads to ligand degradation, product loss, and substantial wastewater generation. The complexity of these recovery systems often requires capital investment in recycling equipment that exceeds the cost of the reaction vessels themselves, creating a bottleneck for efficient large-scale manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN108329194B utilizes a binaphthol-based aluminum catalyst that eliminates the need for corrosive halides or auxiliary ester inhibitors. The core innovation lies in the specific design of the ligand system, where bulky substituents on the binaphthol backbone create a chiral environment that inherently favors the formation of the desired isopulegol isomer. This structural advantage allows the reaction to proceed with high fidelity at temperatures between 5°C and 25°C, removing the energy burden of deep freezing. Crucially, the process introduces a sophisticated recovery protocol involving high-boiling point ether solvents, such as dicyclohexyl ether, which allows the product to be distilled off while the catalyst remains in the residue. Upon cooling, the catalyst crystallizes out in high purity, ready for immediate reuse in subsequent batches. This closed-loop capability significantly enhances the economic viability of the process by minimizing catalyst consumption and waste disposal costs.
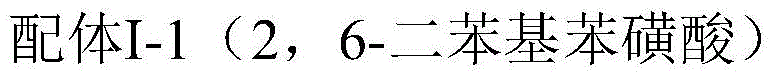
Mechanistic Insights into Binaphthol-Aluminum Catalyzed Cyclization
The exceptional performance of this catalytic system is rooted in the precise molecular architecture of the binaphthol skeleton ligands. As illustrated in the general formula, the ligands feature specific substituents (R1 and R2) that provide substantial steric hindrance around the aluminum center. This steric bulk is critical for discriminating between the transition states leading to different diastereomers, effectively suppressing the formation of unwanted neo-isopulegol and iso-isopulegol byproducts. The aluminum atom, coordinated by the phenolic hydroxyl groups of the binaphthol ligand, acts as a potent yet controlled Lewis acid that activates the carbonyl group of citronellal for intramolecular attack. Unlike simpler Lewis acids that coordinate loosely and promote random cyclization, the chiral binaphthol framework imposes a rigid geometry on the substrate, ensuring that the cyclization occurs with high stereochemical control. This mechanistic precision results in enantioselectivity values consistently exceeding 95%, and in optimized embodiments reaching up to 99.9%, which is paramount for producing high-quality menthol derivatives.
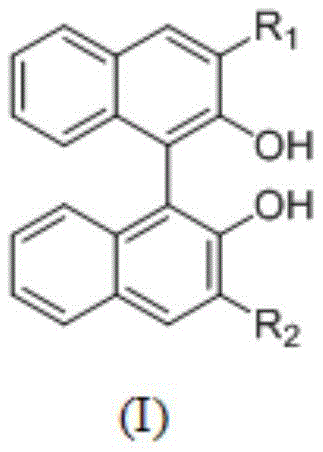
Furthermore, the stability of the catalyst during the reaction and recovery phases is enhanced by the bidentate nature of the binaphthol ligand. The two phenolic hydroxyl groups form a stable chelate with the aluminum, preventing the dissociation of the ligand under the thermal stress of distillation. This stability is the key enabler for the novel recovery method, where the catalyst is not destroyed by hydrolysis or oxidation. In comparative studies involving sulfonic acid ligands or less sterically hindered phenols, the catalysts often decompose or fail to crystallize efficiently, leading to significant losses. The binaphthol-aluminum complex, however, exhibits excellent crystallization behavior in the presence of high-boiling ethers, allowing for a physical separation mechanism that is far gentler than chemical quenching. This ensures that the active catalytic species retains its coordination sphere and activity cycle after cycle, maintaining consistent reaction kinetics throughout prolonged production campaigns.
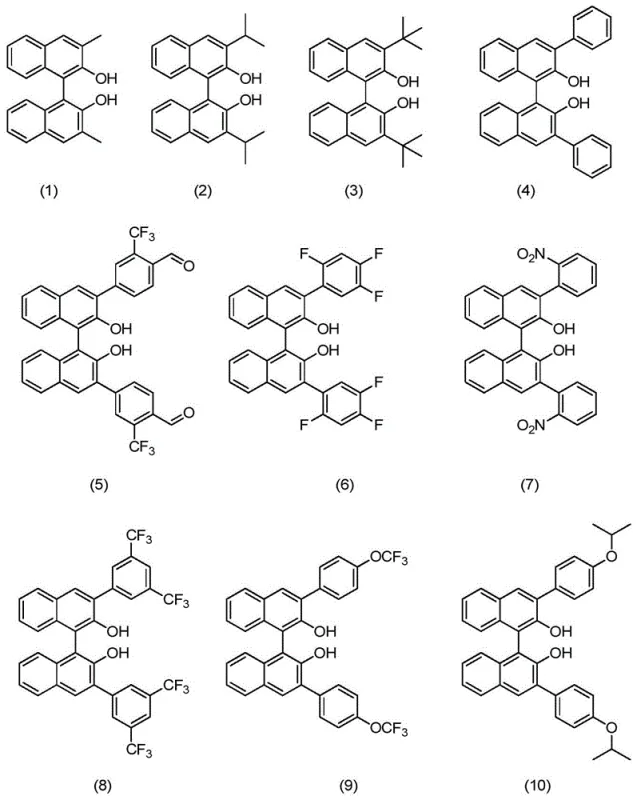
How to Synthesize Isopulegol Efficiently
The synthesis protocol outlined in the patent provides a robust framework for manufacturing isopulegol with minimal operational complexity. The process begins with the in situ generation of the catalyst or the use of a pre-formed solution, which is then introduced to the citronellal feedstock under strictly anhydrous and oxygen-free conditions to prevent catalyst deactivation. The reaction proceeds smoothly at near-ambient temperatures, requiring only standard agitation and temperature control equipment. Following the conversion of citronellal, the workup involves a strategic solvent swap where low-boiling reaction solvents are removed, and a high-boiling ether is introduced to facilitate product distillation. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and crystallization parameters, are provided in the guide below to ensure reproducibility and optimal yield.
- Prepare the catalyst solution by reacting a binaphthol skeleton ligand with alkyl aluminum or aluminum hydride in an anhydrous solvent under inert atmosphere.
- Add the catalyst solution dropwise to a citronellal solution maintained at mild temperatures (5-25°C) to initiate the cyclization reaction.
- Recover the catalyst by adding a high-boiling point solvent, distilling off the product and low-boiling solvents, and crystallizing the catalyst from the residue.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this binaphthol-aluminum catalytic process offers compelling economic and logistical benefits that extend beyond mere chemical yield. The elimination of corrosive zinc bromide removes the necessity for specialized corrosion-resistant reactors, thereby reducing capital expenditure (CAPEX) for new production lines and extending the lifespan of existing assets. Additionally, the removal of ester additives from the reaction mixture simplifies the purification train, reducing the number of distillation columns or extraction stages required to meet purity specifications. This streamlining of the process flow directly translates to lower utility costs and reduced labor intensity, as operators are not required to manage complex aqueous waste streams or perform intricate ligand regeneration chemistries. The ability to recover and reuse the catalyst directly without regeneration creates a circular economy within the reactor, significantly lowering the cost of goods sold (COGS) associated with expensive chiral ligands.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by eliminating the need for expensive ester inhibitors and reducing energy consumption through mild reaction temperatures. The direct reuse of the catalyst without chemical regeneration minimizes the recurring cost of ligand purchase, which is often a major expense in asymmetric synthesis. Furthermore, the simplified downstream processing reduces solvent usage and waste treatment fees, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: By utilizing a catalyst system that is stable and easily recoverable, manufacturers can ensure consistent production schedules without the downtime associated with catalyst replenishment or equipment maintenance due to corrosion. The high conversion rates minimize the accumulation of unreacted raw materials, optimizing inventory turnover and reducing the risk of supply bottlenecks. The robustness of the process against minor fluctuations in operating conditions also enhances overall plant reliability, ensuring a steady supply of high-purity isopulegol to downstream customers.
- Scalability and Environmental Compliance: The absence of heavy metal contaminants and corrosive halides makes this process inherently safer and more environmentally friendly, aligning with increasingly stringent global regulatory standards. The crystallization-based recovery method generates significantly less wastewater compared to aqueous washing techniques, simplifying effluent treatment and reducing the environmental footprint of the facility. This green chemistry profile facilitates easier permitting for capacity expansion and strengthens the sustainability credentials of the final product in the eyes of eco-conscious consumers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced catalytic technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the operational nuances of the binaphthol-aluminum system. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production workflows or designing new facilities dedicated to high-value terpene synthesis.
Q: What are the advantages of the binaphthol aluminum catalyst over traditional zinc bromide?
A: Unlike zinc bromide, which suffers from equipment corrosion and lower enantioselectivity (around 91%), the binaphthol aluminum catalyst achieves 95-99.9% enantioselectivity under milder conditions without corrosive byproducts.
Q: How is the catalyst recovered in this new process?
A: The catalyst is recovered via a simple crystallization method using high-boiling point ether solvents, allowing direct reuse without regeneration, unlike previous methods requiring complex aqueous washing.
Q: Does this process require ester additives as inhibitors?
A: No, the steric hindrance provided by the specific binaphthol ligand structure eliminates the need for ester additives, simplifying the downstream purification and reducing waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isopulegol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes for high-value intermediates like isopulegol. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patented technologies are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of isopulegol meets the exacting standards required for flavor, fragrance, and pharmaceutical applications. Our commitment to process excellence allows us to deliver products with consistent quality profiles, supporting our partners in maintaining their own high standards of production.
We invite potential partners to engage with our technical procurement team to discuss how this innovative catalytic process can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to this advanced methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive global market.
