Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
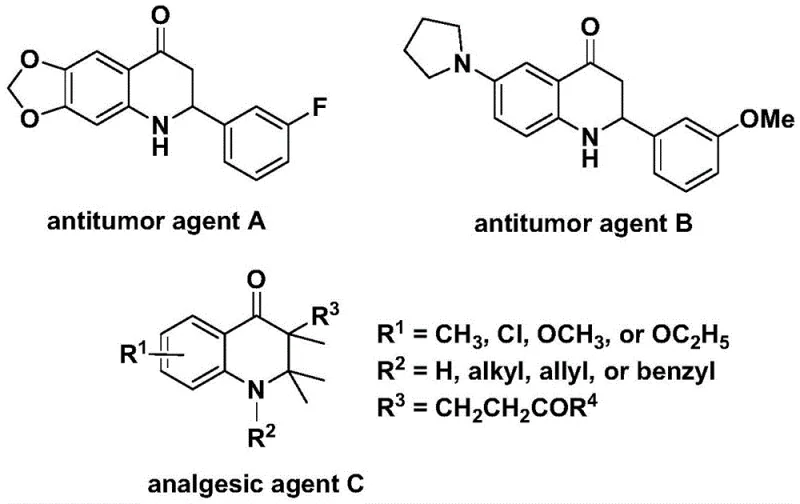
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN112239456B discloses a highly efficient preparation method for substituted 2,3-dihydroquinolone compounds, a privileged scaffold found in numerous therapeutic agents. As illustrated in the structural diversity of known bioactive molecules, these cores are critical for antitumor and analgesic applications, serving as key intermediates in complex drug synthesis. The disclosed technology leverages a transition metal palladium-catalyzed carbonylation strategy, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary building blocks. This approach represents a significant advancement over traditional methods by employing a solid carbon monoxide surrogate, thereby mitigating the safety risks associated with handling toxic CO gas while maintaining high reaction efficiency and substrate tolerance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone skeleton has relied on methodologies that often suffer from significant operational drawbacks. Traditional carbonylation reactions frequently require the use of high-pressure carbon monoxide gas, which necessitates specialized autoclave equipment and rigorous safety protocols, creating bottlenecks in both laboratory optimization and industrial scale-up. Furthermore, many existing synthetic routes exhibit limited substrate compatibility, particularly when attempting to introduce diverse functional groups at the 2-position of the quinolone ring. Harsh reaction conditions, such as extreme temperatures or strong acidic/basic environments, can lead to the decomposition of sensitive functional groups, resulting in complex impurity profiles that are difficult to purify. These factors collectively increase the cost of goods sold (COGS) and extend the lead time for process development, making conventional methods less attractive for the rapid production of high-purity pharmaceutical intermediates required by modern drug discovery pipelines.
The Novel Approach
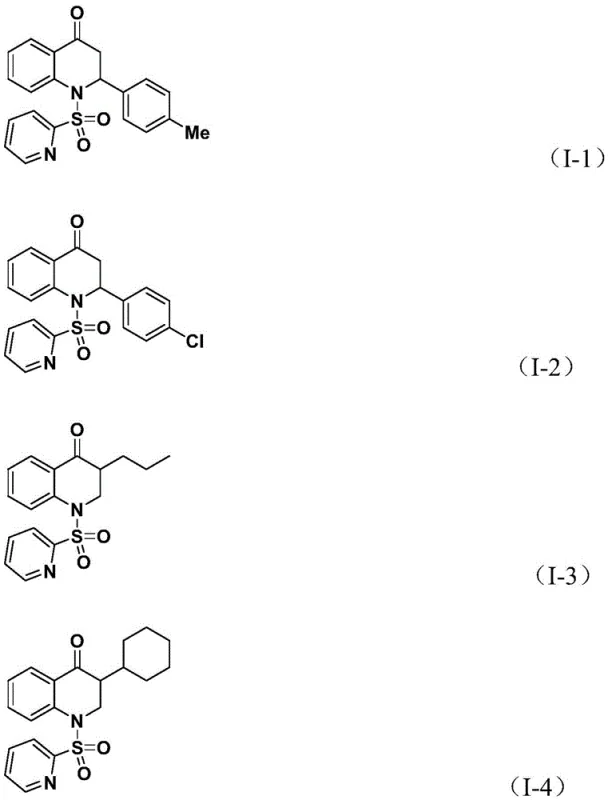
The methodology described in CN112239456B overcomes these hurdles through a cleverly designed palladium-catalyzed cascade reaction. By utilizing 1,3,5-trimethoxybenzene phenol ester (TFBen) as a solid carbon monoxide surrogate, the process eliminates the need for gas cylinders and pressure vessels, allowing the reaction to proceed in standard glassware under atmospheric pressure. The reaction system employs a Pd(acac)2 catalyst with a dppp ligand in 1,4-dioxane at 110 °C, conditions that are mild enough to preserve sensitive functionalities yet energetic enough to drive the cyclization to completion. As demonstrated by the successful synthesis of compounds I-1 through I-5, this route accommodates a wide array of olefins, including aryl, alkyl, and silyl-substituted variants. This versatility allows medicinal chemists to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies without being constrained by the limitations of gas-phase reagents.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The core of this transformation lies in the intricate catalytic cycle driven by the palladium species. The mechanism initiates with the oxidative addition of the palladium(0) catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent functionalization. Following this activation, the carbon monoxide molecule, released in situ from the thermal decomposition of the TFBen additive, inserts into the palladium-carbon bond to form an acyl-palladium species. This insertion step is the defining moment where the carbonyl group of the final quinolone ring is established. The coordination and subsequent migratory insertion of the olefin into the acyl-palladium bond then extends the carbon chain, setting the stage for ring closure. Finally, a reductive elimination step releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium(0) catalyst, completing the cycle. This mechanistic pathway ensures high atom economy and precise control over the regioselectivity of the substitution.
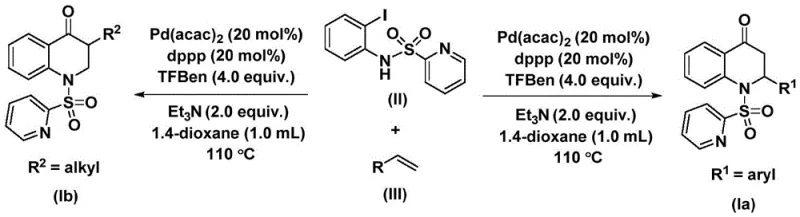
From an impurity control perspective, the choice of the pyridine sulfonyl protecting group on the aniline nitrogen plays a pivotal role. This group not only directs the initial oxidative addition but also stabilizes the intermediate species, preventing unwanted side reactions such as homocoupling of the aryl iodide or polymerization of the olefin. The use of triethylamine as an additive further assists in neutralizing any acidic byproducts generated during the reaction, maintaining a neutral pH environment that protects the integrity of the product. The high yields observed, ranging from 59% to 88% across various substrates, indicate that the catalytic system effectively suppresses the formation of non-productive palladium black and other decomposition pathways. For R&D teams, understanding this mechanism allows for fine-tuning of ligand electronics and steric properties to further optimize yields for specific, challenging substrates, ensuring a clean impurity profile that simplifies downstream purification.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it suitable for both small-scale discovery and larger pilot production. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and substrates in an organic solvent, followed by heating. The simplicity of the workup procedure, involving filtration and standard column chromatography, minimizes the technical burden on the production team. For detailed operational parameters and stoichiometry, please refer to the standardized guide below which outlines the precise steps for implementation.
- Combine bis(acetylacetone)palladium, dppp ligand, triethylamine, TFBen (CO surrogate), N-pyridine sulfonyl-o-iodoaniline, and olefin in 1,4-dioxane.
- Heat the reaction mixture to 110 °C and stir for 48 hours under inert atmosphere to facilitate the carbonylation cyclization.
- Filter the reaction mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 2,3-dihydroquinolone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The shift from gaseous reagents to solid surrogates fundamentally changes the logistics of raw material handling, reducing the regulatory burden and infrastructure costs associated with hazardous gas storage. This transition facilitates a more agile supply chain capable of responding quickly to fluctuating demand without the delays inherent in managing specialized gas supplies. Furthermore, the broad substrate scope means that a single platform technology can be used to produce a wide variety of intermediates, consolidating supplier relationships and simplifying inventory management. The robustness of the reaction conditions ensures consistent quality output, reducing the risk of batch failures that can disrupt production schedules and delay time-to-market for critical drug candidates.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and toxic gas handling systems leads to a substantial reduction in capital expenditure (CAPEX) and operational expenditure (OPEX). By using commercially available and inexpensive starting materials like olefins and iodoanilines, the direct material costs are kept low. Additionally, the high conversion rates and clean reaction profiles minimize the consumption of solvents and silica gel during purification, further driving down the overall cost of production. The ability to run the reaction in standard reactors rather than specialized autoclaves also lowers maintenance and inspection costs, contributing to a more economically viable manufacturing process for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents like TFBen and common organic solvents like dioxane ensures a secure supply chain less susceptible to the logistical disruptions often associated with compressed gases. The starting materials, including various substituted styrenes and alkyl olefins, are widely available from multiple global chemical suppliers, mitigating the risk of single-source dependency. This diversification of the supply base enhances resilience against market volatility and geopolitical instability. Moreover, the scalability of the process from gram to kilogram levels without significant re-optimization ensures that supply can be ramped up seamlessly as clinical trials progress, guaranteeing continuity of supply for long-term commercial projects.
- Scalability and Environmental Compliance: The process aligns well with green chemistry principles by avoiding the use of toxic carbon monoxide gas, thereby reducing the environmental footprint and improving workplace safety. The waste stream is primarily composed of organic solvents and benign byproducts, which are easier to treat and dispose of compared to heavy metal-laden or highly toxic waste streams from alternative methods. The high atom efficiency of the carbonylation step ensures minimal waste generation per unit of product. This environmental compliance simplifies the permitting process for new manufacturing facilities and reduces the liability associated with hazardous waste disposal, making it an attractive option for companies aiming to meet stringent sustainability goals while scaling up complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters. Understanding these details is crucial for assessing the feasibility of integrating this route into existing manufacturing workflows.
Q: What is the advantage of using TFBen over gaseous CO in this synthesis?
A: Using 1,3,5-trimethoxybenzene phenol ester (TFBen) as a solid carbon monoxide surrogate eliminates the need for high-pressure gas equipment, significantly enhancing operational safety and simplifying the reaction setup for scale-up.
Q: What is the substrate scope for the olefin component?
A: The method demonstrates excellent compatibility with various olefins, including substituted styrenes (aryl), linear and branched alkenes (alkyl), and even silyl-substituted alkenes, allowing for diverse functionalization at the 2-position.
Q: What are the typical reaction yields for this protocol?
A: Experimental data indicates robust yields ranging from 59% to 88% across different substrates, with aryl-substituted olefins generally providing higher conversion rates compared to alkyl variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We are committed to delivering high-purity 2,3-dihydroquinolone intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to adapt the patented palladium-catalyzed carbonylation process allows us to offer a reliable supply of these valuable scaffolds, tailored to the specific needs of your R&D and commercial programs.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis specific to your target molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall manufacturing costs. Let us be your partner in turning complex chemical challenges into commercial successes.
