Revolutionizing Ketoester Production: A Deep Dive into Vanadium-Catalyzed Oxidation Technology
Revolutionizing Ketoester Production: A Deep Dive into Vanadium-Catalyzed Oxidation Technology
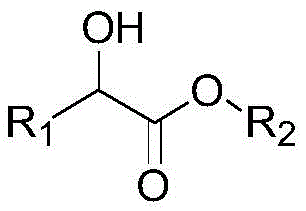
The landscape of fine chemical synthesis is undergoing a transformative shift driven by the urgent need for sustainable, cost-effective, and scalable manufacturing processes, a trend vividly exemplified by the technological breakthroughs detailed in Chinese Patent CN110183327B. This pivotal intellectual property introduces a sophisticated method for preparing ketoesters through the catalytic oxidation of hydroxy esters, utilizing a novel vanadium-based nitrogen-doped carbon material as the heterogeneous catalyst. Unlike traditional methodologies that often rely on prohibitively expensive noble metals or toxic stoichiometric oxidants, this innovation leverages molecular oxygen as a green oxidant to achieve selective oxidative dehydrogenation. For R&D directors and procurement strategists in the pharmaceutical and agrochemical sectors, this represents a critical opportunity to optimize supply chains for high-value intermediates such as pyruvates and benzoylformates. The patent outlines a robust protocol where alpha-hydroxy esters are converted into alpha-keto esters with exceptional conversion rates and selectivity, operating under relatively mild thermal and pressure conditions that facilitate easier scale-up and safer industrial implementation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of ketoesters has been plagued by significant technical and economic bottlenecks that hinder efficient large-scale manufacturing and compromise overall process sustainability. Conventional routes often depend on the esterification of pyruvic acid, which itself is frequently synthesized using harsh oxidants or multi-step sequences that generate substantial quantities of hazardous waste and incur high purification costs. Furthermore, existing catalytic oxidation methods have predominantly utilized noble metal catalysts such as palladium-platinum bimetallic systems or homogeneous catalysts containing toxic elements like selenium, which pose severe challenges regarding catalyst recovery, product contamination, and environmental compliance. For instance, while some prior art reports high yields using Pd-Pt/Bi2O3 systems, the reliance on precious metals drastically inflates the cost of goods sold (COGS) and introduces supply chain vulnerabilities associated with the volatility of noble metal markets. Additionally, methods employing stoichiometric oxidants often suffer from poor atom economy, requiring extensive downstream processing to remove inorganic salts and heavy metal residues, thereby extending production lead times and increasing the burden on waste treatment facilities.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN110183327B offers a paradigm shift by employing a non-noble, vanadium-based nitrogen-doped carbon catalyst that delivers superior performance metrics without the associated economic and environmental penalties. This innovative approach utilizes molecular oxygen or air as the sole oxidant, ensuring that the only byproduct of the reaction is water, which fundamentally aligns with the principles of green chemistry and significantly simplifies the workup procedure. The vanadium-based catalyst exhibits remarkable stability and activity, enabling the selective oxidation of a broad spectrum of alpha-hydroxy esters, including lactates and mandelates, to their corresponding ketoesters with conversion rates reaching up to 99% and selectivity exceeding 95% under optimized conditions. By operating within a moderate temperature range of 40-180°C and oxygen pressures of 0.01-2 MPa, the process mitigates the safety risks associated with high-pressure hydrogenation or extreme thermal regimes, making it highly attractive for commercial adoption. This novel pathway not only eliminates the need for costly noble metals but also streamlines the synthetic route into a single-step oxidative dehydrogenation, thereby reducing capital expenditure on equipment and operational complexity.
Mechanistic Insights into Vanadium-Catalyzed Oxidative Dehydrogenation
The efficacy of this synthesis relies on the unique electronic and structural properties of the vanadium-based nitrogen-doped carbon material, which acts as a highly active heterogeneous catalyst for the activation of molecular oxygen. The nitrogen doping within the carbon matrix plays a crucial role in anchoring the vanadium species, preventing their aggregation and leaching during the reaction, which ensures long-term catalyst stability and reusability essential for continuous flow or batch processing. Mechanistically, the reaction proceeds via an oxidative dehydrogenation pathway where the alpha-hydroxy ester adsorbs onto the active vanadium sites, facilitating the abstraction of hydrogen atoms from the alpha-carbon and the hydroxyl group. The activated molecular oxygen then accepts these hydrogen equivalents to form water, regenerating the active catalytic site and driving the equilibrium towards the formation of the carbonyl functionality in the alpha-keto ester product. This mechanism avoids the formation of radical species that typically lead to over-oxidation or C-C bond cleavage, thereby explaining the high selectivity observed even at elevated conversions. The synergy between the vanadium centers and the nitrogen-doped carbon support creates an electronic environment that lowers the activation energy for the rate-determining step, allowing the reaction to proceed efficiently at lower temperatures compared to uncatalyzed thermal oxidation.
From an impurity control perspective, the heterogeneity of the catalyst system provides a distinct advantage by minimizing the presence of metal residues in the final product, a critical parameter for pharmaceutical intermediates intended for API synthesis. The solid nature of the catalyst allows for straightforward separation via filtration, effectively preventing vanadium contamination that would otherwise require complex chelation or ion-exchange purification steps. Furthermore, the selectivity of the catalyst suppresses side reactions such as ester hydrolysis or decarboxylation, which are common pitfalls in acid-catalyzed or high-temperature oxidative processes. The patent data indicates that by fine-tuning the ratio of nitrogen source to vanadium source during catalyst preparation, one can optimize the dispersion of active sites to further enhance specificity towards the desired ketoester. This level of control over the catalytic microenvironment ensures a clean impurity profile, reducing the burden on downstream crystallization or distillation units and ultimately improving the overall yield of the isolated high-purity product.
How to Synthesize Alpha-Keto Esters Efficiently
The practical implementation of this technology involves a straightforward protocol that integrates seamlessly into existing fine chemical manufacturing infrastructure, requiring only standard high-pressure reactors and heating equipment. The process begins with the precise formulation of the reaction mixture, where the vanadium-based nitrogen-doped carbon catalyst is dispersed in a suitable solvent such as acetonitrile, ethyl acetate, or toluene alongside the alpha-hydroxy ester substrate. Once the reactor is sealed and purged, molecular oxygen is introduced to establish the required partial pressure, creating an oxidizing atmosphere that drives the reaction forward. The mixture is then heated with vigorous stirring to ensure efficient mass transfer between the gas, liquid, and solid phases, maintaining the temperature within the optimal window identified in the patent examples to maximize conversion while preserving selectivity. Throughout the reaction duration, which typically spans several hours, the oxygen pressure is monitored and replenished as necessary to compensate for consumption, ensuring that the oxidant remains in excess to prevent stalling of the catalytic cycle. Upon completion, the reaction is cooled to ambient temperature, depressurized safely, and the catalyst is removed by filtration, yielding a crude solution that can be directly concentrated or purified to obtain the target ketoester.
- Prepare the reaction mixture by loading the vanadium-based nitrogen-doped carbon catalyst, alpha-hydroxy ester substrate, and a suitable liquid-phase solvent such as acetonitrile or ethyl acetate into a high-pressure reactor.
- Seal the reactor and introduce the oxygen source, maintaining an oxygen partial pressure between 0.01 and 2 MPa to ensure sufficient oxidant availability throughout the reaction cycle.
- Heat the mixture with stirring to a temperature range of 40-180°C and maintain reaction for 0-12 hours, replenishing oxygen if consumed, before cooling and depressurizing to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this vanadium-catalyzed oxidation technology presents a compelling value proposition centered around significant cost optimization and enhanced supply security. The elimination of noble metal catalysts such as palladium and platinum removes a major variable cost driver from the manufacturing equation, insulating the production budget from the volatile fluctuations of the precious metals market. Furthermore, the use of molecular oxygen or air as the oxidant replaces expensive and hazardous stoichiometric oxidants, leading to a drastic reduction in raw material expenditures and waste disposal fees associated with heavy metal salts. The simplified downstream processing, enabled by the heterogeneous nature of the catalyst and the clean reaction profile, translates into shorter production cycles and reduced utility consumption, collectively contributing to a leaner and more competitive cost structure. These efficiencies allow suppliers to offer high-purity ketoesters at more aggressive price points while maintaining healthy margins, providing a strategic advantage in negotiations with downstream API manufacturers.
- Cost Reduction in Manufacturing: The transition from noble metal-based systems to earth-abundant vanadium catalysts fundamentally alters the cost dynamics of ketoester production by removing the dependency on scarce and expensive resources. This substitution not only lowers the direct material cost but also reduces the capital tied up in catalyst inventory and recovery systems, as the robust carbon-supported vanadium catalyst can potentially be regenerated or reused multiple times without significant loss of activity. Additionally, the mild reaction conditions reduce energy consumption for heating and pressurization, while the high selectivity minimizes the loss of valuable starting materials to byproducts, thereby maximizing the effective yield per batch. These cumulative savings create a substantial buffer against market price pressures, enabling more flexible pricing strategies for long-term supply contracts.
- Enhanced Supply Chain Reliability: By utilizing readily available raw materials such as alpha-hydroxy esters and industrial-grade oxygen, this method mitigates the risk of supply disruptions often associated with specialized reagents or imported catalysts. The robustness of the vanadium-based catalyst ensures consistent batch-to-batch quality, reducing the incidence of failed runs or off-spec material that can derail production schedules and delay deliveries to customers. Moreover, the scalability of the process from laboratory to pilot and eventually to commercial tonnage scales is facilitated by the use of standard unit operations, allowing suppliers to rapidly ramp up capacity in response to surging demand from the pharmaceutical or agrochemical sectors. This reliability fosters stronger partnerships with key accounts who prioritize continuity of supply and predictable lead times in their own manufacturing planning.
- Scalability and Environmental Compliance: The green chemistry attributes of this process, specifically the use of oxygen and the generation of water as the primary byproduct, significantly simplify regulatory compliance and environmental permitting for manufacturing facilities. The absence of toxic heavy metal waste streams reduces the liability and cost associated with hazardous waste treatment and disposal, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. This environmental friendliness enhances the marketability of the produced ketoesters to eco-conscious buyers and facilitates easier approval in regulated markets where impurity profiles are strictly scrutinized. The inherent safety of operating at moderate pressures and temperatures further reduces the risk profile of the plant, lowering insurance premiums and ensuring uninterrupted operations free from safety-related shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this catalytic oxidation technology, drawing directly from the experimental data and claims of the underlying patent. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing portfolios or for procurement specialists assessing the quality and consistency of the supply. The answers provided reflect the specific advantages of the vanadium-nitrogen-carbon system over alternative methods, highlighting its versatility across different substrates and its operational robustness under varied conditions. This information serves as a foundational guide for stakeholders seeking to leverage this innovation for competitive advantage in the fine chemicals market.
Q: What are the primary advantages of using vanadium-based nitrogen-doped carbon catalysts over traditional noble metal catalysts?
A: The primary advantage lies in the drastic reduction of raw material costs by eliminating expensive noble metals like palladium or platinum, while simultaneously achieving comparable or superior conversion rates and selectivity under milder reaction conditions.
Q: Can this oxidation method be applied to a wide range of hydroxy ester substrates beyond methyl lactate?
A: Yes, the patent data demonstrates high versatility, successfully converting various alpha-hydroxy esters including ethyl lactate, methyl mandelate, and methyl 3-hydroxybutyrate into their corresponding ketoesters with high efficiency.
Q: How does the use of molecular oxygen impact the environmental profile of this synthesis compared to stoichiometric oxidants?
A: Utilizing molecular oxygen or air as the terminal oxidant significantly improves the atom economy and reduces hazardous waste generation, as the only byproduct is water, avoiding the heavy metal waste associated with chromium or selenium-based oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketoester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to deliver high-performance chemical intermediates that meet the rigorous demands of the global pharmaceutical and fine chemical industries. Our technical team has extensively analyzed the potential of the vanadium-catalyzed oxidation pathway described in CN110183327B and possesses the expertise to translate this laboratory-scale innovation into a robust, commercial-grade manufacturing process. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-purity ketoesters regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications, free from the noble metal residues that plague conventional supplies.
We invite forward-thinking partners to collaborate with us to unlock the full commercial potential of this technology for your specific applications. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to our catalytically optimized ketoesters for your synthesis routes. We encourage you to reach out today to discuss your specific requirements,索取 specific COA data for our current inventory, and review our comprehensive route feasibility assessments to determine how we can best support your supply chain objectives with reliable, cost-effective, and sustainable chemical solutions.
