Revolutionizing Ester Synthesis: Direct Oxidative Cleavage of Secondary Alcohols for Commercial Scale Manufacturing
The landscape of organic synthesis is witnessing a paradigm shift with the emergence of advanced catalytic systems capable of transforming simple precursors into high-value intermediates through unconventional pathways. A groundbreaking development in this field is detailed in patent CN112876355B, which discloses a highly efficient method for preparing carboxylic ester compounds via the oxidative cleavage of carbon-carbon bonds in secondary alcohol compounds. This technology represents a significant departure from classical esterification routes, utilizing a nitrogen-doped mesoporous carbon-supported single-atom catalyst to drive the reaction under relatively mild conditions. For R&D directors and process chemists, this innovation opens new avenues for synthesizing complex ester architectures that were previously difficult or costly to access. The ability to directly convert secondary alcohols into esters bypasses multiple synthetic steps, thereby streamlining the production of key pharmaceutical intermediates and fine chemicals. As we delve deeper into the technical specifics, it becomes evident that this methodology not only enhances reaction efficiency but also aligns with the growing industry demand for sustainable and scalable chemical manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of organic carboxylic esters has long relied on the Fischer esterification process, which typically involves reacting a carboxylic acid with an alcohol in the presence of a strong acid catalyst like concentrated sulfuric acid. While historically significant, this method suffers from severe drawbacks including poor selectivity, extensive side reactions, and the generation of large amounts of acidic waste that necessitates complex neutralization and disposal procedures. Furthermore, the corrosive nature of strong acids imposes stringent requirements on reactor materials, increasing capital expenditure and maintenance costs. Alternative modern approaches often involve activating the carboxylic acid as an acid chloride or anhydride prior to esterification, which introduces additional synthetic steps and generates stoichiometric amounts of hazardous by-products. Even direct oxidative esterification of aldehydes, while useful, frequently relies on stoichiometric oxidants such as manganese dioxide or peroxides, leading to significant atom inefficiency and environmental burden. These conventional limitations create bottlenecks in supply chains, particularly when high purity is required for pharmaceutical applications, as removing trace metal contaminants or acidic residues can be technically challenging and cost-prohibitive.
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes a heterogeneous single-atom catalyst system to achieve direct oxidative cleavage of the C-C bond in secondary alcohols, followed by in-situ esterification. This one-pot transformation eliminates the need for pre-activation of substrates or the use of corrosive mineral acids. The reaction proceeds in an aliphatic primary alcohol solvent under an oxygen atmosphere, where the alcohol serves both as the reactant and the solvent, simplifying the reaction matrix significantly. The use of molecular oxygen as the terminal oxidant is a major green chemistry advantage, as it produces water as the only by-product, drastically reducing the E-factor of the process. Moreover, the catalyst system demonstrates exceptional stability and reusability, addressing the critical issue of catalyst recovery that plagues many homogeneous catalytic systems. By operating at moderate temperatures between 80-150°C and pressures of 0.1-1MPa, the process ensures safety and energy efficiency, making it highly attractive for industrial scale-up.
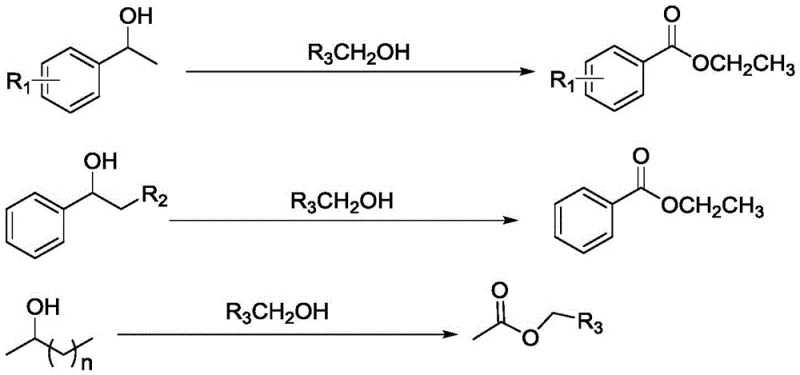
Mechanistic Insights into Co-N-C Catalyzed Oxidative Cleavage
The core of this technological breakthrough lies in the unique structure of the nitrogen-doped mesoporous carbon-supported single-atom catalyst (Co-N-C). Unlike traditional nanoparticle catalysts where active sites are distributed across a metal surface, single-atom catalysts maximize atom utilization by isolating individual metal atoms coordinated with nitrogen atoms within the carbon matrix. This configuration creates highly uniform active sites that facilitate the activation of molecular oxygen and the subsequent abstraction of hydrogen from the secondary alcohol substrate. The mechanistic pathway likely involves the formation of a ketone intermediate via dehydrogenation, followed by a Baeyer-Villiger type oxidation or a radical-mediated C-C bond scission assisted by the cobalt active centers. The nitrogen doping plays a crucial role in stabilizing the cobalt atoms and modulating their electronic properties, thereby enhancing the catalytic turnover frequency. This precise control over the active site geometry minimizes non-selective oxidation pathways, ensuring that the cleavage occurs specifically at the desired carbon-carbon bond adjacent to the hydroxyl group.
Impurity control is another critical aspect where this catalytic system excels. In traditional acid-catalyzed reactions, dehydration of alcohols to alkenes or polymerization of sensitive substrates are common side reactions that complicate downstream purification. The mild basic conditions employed in this novel method, facilitated by additives like potassium carbonate, effectively suppress acid-catalyzed side reactions. The heterogeneous nature of the catalyst also prevents leaching of metal ions into the product stream, a vital consideration for pharmaceutical intermediates where heavy metal limits are strictly regulated. The broad substrate scope encompasses aromatic secondary alcohols with various electronic substituents, heterocyclic alcohols, and even aliphatic chains, demonstrating the robustness of the catalytic cycle against steric and electronic variations.
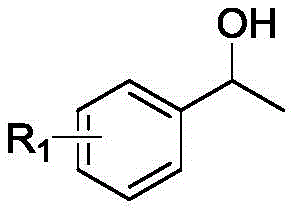
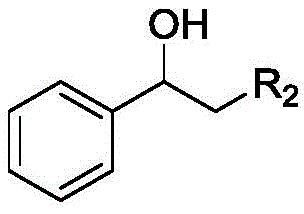
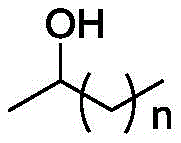
How to Synthesize Carboxylic Ester Compounds Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst and the optimization of reaction parameters to ensure maximum yield and selectivity. The standardized protocol involves mixing the secondary alcohol substrate with the Co-N-C catalyst and an alkaline additive in a primary alcohol solvent, followed by pressurization with oxygen and heating. Detailed operational guidelines regarding catalyst loading, pressure settings, and work-up procedures are essential for reproducibility. For process engineers looking to adopt this technology, understanding the interplay between oxygen pressure and temperature is key to balancing reaction rate and safety. The following section outlines the fundamental steps required to execute this transformation effectively in a laboratory or pilot plant setting.
- Prepare the reaction mixture by adding the secondary alcohol substrate, an alkaline additive (e.g., K2CO3), and the Co-N-C single-atom catalyst into an aliphatic primary alcohol solvent within a pressure vessel.
- Seal the vessel and introduce an oxygen source gas to achieve a pressure between 0.1 MPa and 1 MPa, ensuring a safe and controlled oxidative environment.
- Heat the reaction mixture to a temperature range of 80-150°C and stir for 6-30 hours to facilitate the oxidative cleavage of the carbon-carbon bond and subsequent esterification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this oxidative cleavage technology offers transformative benefits that extend beyond mere chemical efficiency. The shift from stoichiometric reagents to a catalytic process driven by molecular oxygen fundamentally alters the cost structure of ester production. By eliminating the need for expensive activating agents like thionyl chloride or coupling reagents, the raw material costs are significantly reduced. Additionally, the simplicity of the work-up procedure, which primarily involves filtration to remove the solid catalyst, reduces the consumption of solvents and energy associated with complex extraction and distillation steps. This streamlined workflow translates directly into shorter production cycles and lower operational expenditures, providing a competitive edge in the market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic impact of replacing noble metal catalysts or stoichiometric oxidants with a earth-abundant cobalt-based single-atom catalyst cannot be overstated. Cobalt is substantially cheaper than gold or palladium, and the single-atom dispersion ensures that every metal atom contributes to the reaction, maximizing cost-efficiency. Furthermore, the ability to recycle the heterogeneous catalyst multiple times without significant loss of activity amortizes the initial catalyst cost over a larger production volume. The reduction in waste treatment costs, due to the absence of heavy metal sludge or acidic wastewater, further contributes to the overall financial viability of the process, making it an ideal solution for cost-sensitive bulk manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials. Secondary alcohols are commodity chemicals available from diverse global sources, reducing dependency on specialized or scarce reagents. The robustness of the catalyst also means that production schedules are less likely to be disrupted by catalyst degradation or the need for frequent replenishment. This reliability is crucial for maintaining consistent supply to downstream customers in the pharmaceutical and agrochemical sectors, where interruptions can have cascading effects on drug development timelines. The simplified logistics of handling gaseous oxygen and solid catalysts, as opposed to liquid corrosive acids, also enhances workplace safety and reduces regulatory compliance burdens.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the use of standard pressure reactors and the absence of exothermic hazards associated with strong acid additions. The green chemistry credentials of the process, characterized by high atom economy and minimal waste generation, align perfectly with increasingly stringent environmental regulations globally. Companies adopting this technology can leverage its sustainability profile to meet corporate social responsibility goals and satisfy the eco-conscious demands of end-users. The ease of meeting discharge standards due to the benign nature of the by-products simplifies the permitting process for new manufacturing facilities, accelerating time-to-market for new products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cleavage technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios.
Q: What are the advantages of using the Co-N-C single-atom catalyst over traditional noble metal catalysts?
A: The Co-N-C catalyst offers high activity comparable to noble metals like Au or Pd but at a significantly lower cost. Furthermore, being a heterogeneous catalyst, it allows for easy separation and recycling, reducing metal contamination in the final pharmaceutical product.
Q: Can this oxidative cleavage method handle diverse substrate structures?
A: Yes, the method demonstrates broad substrate tolerance, successfully converting aromatic secondary alcohols with various substituents (electron-donating or withdrawing), heterocyclic alcohols, and aliphatic secondary alcohols into their corresponding esters with high selectivity.
Q: How does this process improve environmental compliance compared to traditional esterification?
A: Unlike traditional methods requiring concentrated sulfuric acid or stoichiometric toxic oxidants, this process uses molecular oxygen as the oxidant and produces water as the primary byproduct, significantly reducing hazardous waste generation and equipment corrosion.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboxylic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this oxidative cleavage technology to redefine the production of high-purity carboxylic esters. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped to handle the specific pressure and temperature requirements of this catalytic process, while our rigorous QC labs guarantee that every batch meets stringent purity specifications required by the global pharmaceutical industry. We are committed to delivering not just a product, but a comprehensive solution that optimizes your supply chain and reduces your total cost of ownership.
We invite you to explore how this innovative synthesis route can benefit your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating the tangible economic advantages of switching to this catalytic method. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your path to market with superior quality and efficiency.
