Advanced Magnetic Core-Shell Catalysts for Scalable Tinib Intermediate Manufacturing
Advanced Magnetic Core-Shell Catalysts for Scalable Tinib Intermediate Manufacturing
The global demand for tyrosine kinase inhibitors (TKIs), commonly known as "tinib" drugs such as Gefitinib, Imatinib, and Nilotinib, continues to surge as frontline treatments for various cancers. However, the industrial synthesis of key intermediates for these potent therapeutics often faces significant bottlenecks, particularly regarding the efficiency of C-N coupling reactions and the removal of toxic heavy metal residues. A groundbreaking technological solution is detailed in Chinese Patent CN108295882B, which introduces a novel magnetic separation core-shell supported palladium catalyst, designated as Pd/Fe3O4@C-N. This innovation represents a paradigm shift in pharmaceutical intermediates manufacturing, offering a robust pathway to high-purity compounds while drastically simplifying downstream processing. By integrating a magnetic Fe3O4 core with a nitrogen-doped carbon shell, this technology not only enhances catalytic performance but also enables effortless catalyst recovery, addressing long-standing pain points in green chemistry and process safety.
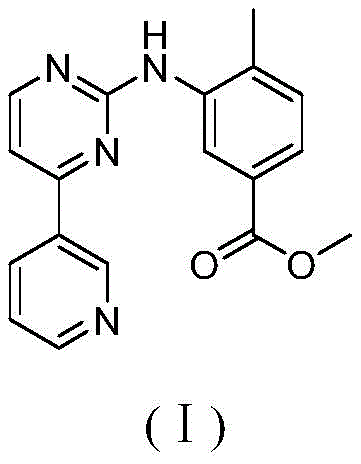
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing tinib drug intermediates typically rely on homogeneous palladium catalysts or standard heterogeneous supports like activated carbon (Pd/C). While these methods can achieve acceptable yields, they suffer from severe drawbacks that hinder cost reduction in API manufacturing. Homogeneous catalysts, though highly active, are notoriously difficult to separate from the reaction mixture, often requiring complex and expensive purification steps to meet stringent regulatory limits on residual metals in pharmaceutical products. Furthermore, heterogeneous Pd/C catalysts often exhibit poor stability, leading to palladium leaching and catalyst deactivation over time. The physical separation of fine Pd/C powders via filtration is also labor-intensive and prone to mechanical losses, resulting in inconsistent batch-to-batch reproducibility and increased operational expenditures. These inefficiencies create a fragile supply chain for high-purity pharmaceutical intermediates, where any delay in purification can jeopardize entire production schedules.
The Novel Approach
The technology disclosed in patent CN108295882B overcomes these hurdles through the engineering of a sophisticated core-shell nanostructure. As illustrated in the reaction scheme below, the Pd/Fe3O4@C-N catalyst facilitates the C-N coupling between 3-bromo-5-(trifluoromethyl)aniline and 4-methyl-1H-imidazole with exceptional efficiency. The unique architecture features a superparamagnetic Fe3O4 core that allows for rapid and complete separation of the catalyst from the reaction broth using a simple external magnet, eliminating the need for tedious filtration or centrifugation. Moreover, the nitrogen-doped carbon shell serves a dual purpose: it acts as a protective barrier that prevents the aggregation of palladium nanoparticles and chemically anchors the active metal sites to minimize leaching. This results in a cleaner reaction profile and a product that requires less rigorous purification, directly translating to higher throughput and reduced environmental impact.
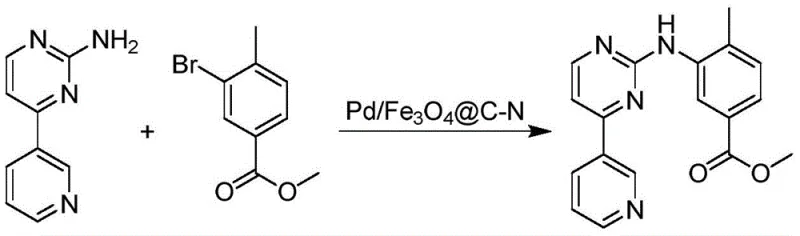
Mechanistic Insights into Magnetic Core-Shell Catalysis
The superior performance of the Pd/Fe3O4@C-N system stems from its precise multi-step synthesis, which creates a synergistic environment for catalysis. The process begins with the hydrothermal synthesis of uniform Fe3O4 nanoparticles, which provide the magnetic foundation. Subsequently, a carbon layer is formed via the hydrothermal carbonization of glucose in the presence of nitrogen sources such as N-acetyl-D-glucosamine or ethylenediamine. This step is critical, as the doping of nitrogen atoms into the carbon lattice creates electron-rich defects that strongly coordinate with palladium ions during the final impregnation step. This strong metal-support interaction ensures that the palladium remains dispersed as ultrafine nanoparticles rather than sintering into inactive bulk metal. The resulting catalyst exhibits high surface area and accessibility, allowing reactants to diffuse easily to the active sites while the robust shell protects the magnetic core from acidic or oxidative degradation during the reaction cycle.

From an impurity control perspective, this mechanism offers distinct advantages for commercial scale-up of complex pharmaceutical intermediates. In conventional systems, metal leaching often leads to the formation of organometallic byproducts that are difficult to remove and can be genotoxic. The nitrogen-doped shell in the Pd/Fe3O4@C-N catalyst effectively traps palladium species, ensuring that the leaching levels remain well below the strict thresholds required by pharmacopeias. Additionally, the magnetic recoverability means that the catalyst can be physically removed before the workup stage begins, preventing any further contact between the catalyst and the product during extraction or crystallization. This inherent design feature minimizes the risk of cross-contamination and simplifies the validation process for regulatory filings, making it an attractive option for contract development and manufacturing organizations (CDMOs) focused on oncology portfolios.
How to Synthesize Tinib Intermediate Efficiently
The implementation of this catalytic system in a production setting follows a streamlined protocol that balances reaction kinetics with ease of operation. The synthesis involves charging the aryl halide and heterocyclic amine substrates into a reactor containing the Pd/Fe3O4@C-N catalyst and a suitable base, such as potassium hydroxide, in a mixed solvent system of DMSO and water. The reaction is typically conducted at elevated temperatures ranging from 100°C to 130°C to ensure complete conversion. Once the reaction reaches completion, the unique magnetic properties of the catalyst allow for immediate separation, after which the product is isolated through standard liquid-liquid extraction and recrystallization. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures validated in the patent literature, please refer to the standardized synthesis guide below.
- Combine 3-bromo-5-(trifluoromethyl)aniline, 4-methyl-1H-imidazole, solvent (DMSO/Water), base (KOH), and the Pd/Fe3O4@C-N catalyst in a reaction vessel.
- Heat the mixture to 100-130°C and stir for 8-15 hours to facilitate the coupling reaction.
- Separate the catalyst using an external magnet, extract the product with ethyl acetate, dry the organic phase, and recrystallize from toluene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the Pd/Fe3O4@C-N catalyst technology offers compelling strategic benefits that extend beyond mere technical performance. The primary value driver is the drastic simplification of the downstream processing workflow. By replacing filtration-intensive methods with magnetic decantation, manufacturers can significantly reduce cycle times and labor costs associated with catalyst removal. This efficiency gain is particularly valuable in the production of high-value oncology intermediates, where time-to-market is a critical competitive factor. Furthermore, the ability to recycle the catalyst multiple times without significant loss of activity means that the effective consumption of precious palladium metal is minimized, providing a hedge against the volatility of noble metal prices and contributing to substantial long-term cost savings.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive purification steps required to remove trace palladium residues. Traditional methods often necessitate the use of scavenger resins or repeated recrystallizations to meet metal specifications, which adds material costs and reduces overall yield. With the Pd/Fe3O4@C-N system, the low leaching profile inherently produces a cleaner crude product, thereby reducing the load on purification units. Additionally, the reusability of the catalyst across multiple batches amortizes the initial cost of the palladium loading over a much larger volume of product, effectively lowering the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and stability of the catalyst formulation. Unlike sensitive homogeneous catalysts that require inert atmosphere handling and have short shelf lives, this core-shell material is stable under ambient conditions and easy to transport. The simplified synthesis of the catalyst itself, utilizing commodity chemicals like iron salts and glucose, ensures that the supply of the catalytic material is not dependent on obscure or single-source specialty reagents. This decentralization of raw material risk ensures a more reliable and continuous supply of the critical tinib intermediate, safeguarding production schedules against upstream disruptions.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology aligns perfectly with modern green chemistry mandates. The reduction in solvent usage for catalyst washing and the elimination of filter aid wastes (such as diatomaceous earth) significantly lower the facility's E-factor (mass of waste per mass of product). The magnetic separation process generates virtually no solid waste compared to the disposal of spent Pd/C filters, which are often classified as hazardous waste due to their metal content. This reduction in hazardous waste streams simplifies regulatory compliance and lowers waste disposal costs, making the process more sustainable and socially responsible for large-scale pharmaceutical intermediates production.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this magnetic catalysis platform into their existing workflows, we have compiled answers to common inquiries regarding the technology's performance and operational parameters. These insights are derived directly from the experimental data and technical specifications outlined in the underlying patent documentation, ensuring accuracy and relevance for process development scientists. Understanding these nuances is essential for optimizing reaction conditions and maximizing the economic potential of this innovative approach.
Q: How does the Pd/Fe3O4@C-N catalyst improve metal residue control compared to traditional methods?
A: The catalyst utilizes a core-shell structure where nitrogen-doped carbon shells stabilize palladium active sites. This design, combined with magnetic separation, prevents catalyst fragmentation and significantly reduces palladium leaching into the final API intermediate, addressing critical purity concerns in oncology drug synthesis.
Q: Can this magnetic catalyst be reused for multiple batches without losing activity?
A: Yes, experimental data from patent CN108295882B demonstrates that the catalyst maintains high catalytic activity over at least eight consecutive recycling runs. The magnetic core allows for easy recovery without filtration losses, ensuring consistent yield and reducing the need for fresh catalyst procurement.
Q: What are the scalability advantages of using hydrothermal synthesis for this catalyst?
A: The hydrothermal method used to prepare the Fe3O4 core and the subsequent carbonization steps are highly scalable industrial processes. They utilize common reagents like glucose and iron salts, avoiding complex organometallic ligands, which simplifies the supply chain and lowers the overall production cost for large-scale pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tinib Intermediate Supplier
The technological advancements described in patent CN108295882B highlight the immense potential for optimizing the production of critical oncology building blocks. At NINGBO INNO PHARMCHEM, we pride ourselves on staying at the forefront of such process innovations to deliver superior value to our global partners. As a premier CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch of high-purity Tinib Intermediate meets the most stringent purity specifications required by international regulatory bodies.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage these advanced catalytic technologies for their pipeline projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation can drive efficiency and reliability in your supply chain.
